Vendor-neutral IMS for OCT
ROU AI Models for OCT Analysis of 30+ Retinal Pathologies & 40+ Biomarkers
Free Trial without CallsExplore Altris IMS
Explore the platform in a virtual demo or let us show it to you during the call
Free Trial

Video Guide

Setting Higher Standards in Eye Care
For Ophthalmology
For Pharma
For Optometry
-
Our clients on Altris
What do professionals think?
-
“FinnGen and the University of Helsinki collaborated with Altris on a large-scale research project. We’re very pleased with the partnership. Altris delivered highly valuable data, enabling the analysis of 40,000 OCT scans and providing statistical insights that significantly accelerated our research.”

-
 Dr. Marko Lukic
Dr. Marko LukicMedical Director at Ascento CDA
"I was impressed with the robustness of the data of Altris we used for our reserach projects at Ascento CDA."

-
 Dr.Samuel Minaker
Dr.Samuel MinakerDirector of Clinical Research at Tyler Retina Consultants
"I am really impressed by Altris that was used as a reserach support tool.Not many companies perform as well in GA. I do a lot of trials and have found it a valuable tool for research imaging review."

-
 Dr. Sundeep Kheterpal
Dr. Sundeep KheterpalSurgeon at Windsor Eye Clinic
"We were using Altris for more than half a year already, and the results are positive.We are very happy with the outstanding customer service and support."







For Innovative Eye Care Specialists
Support research site eligibility workflows using AI ROU models for 40+ retinal biomarkers & for 30+ retinal conditions
Gather analytics on biomarkers, patient imaging data, and treatment patterns for your RW research
Analyze large volumes of historical OCT data to gather analytics for your reserach
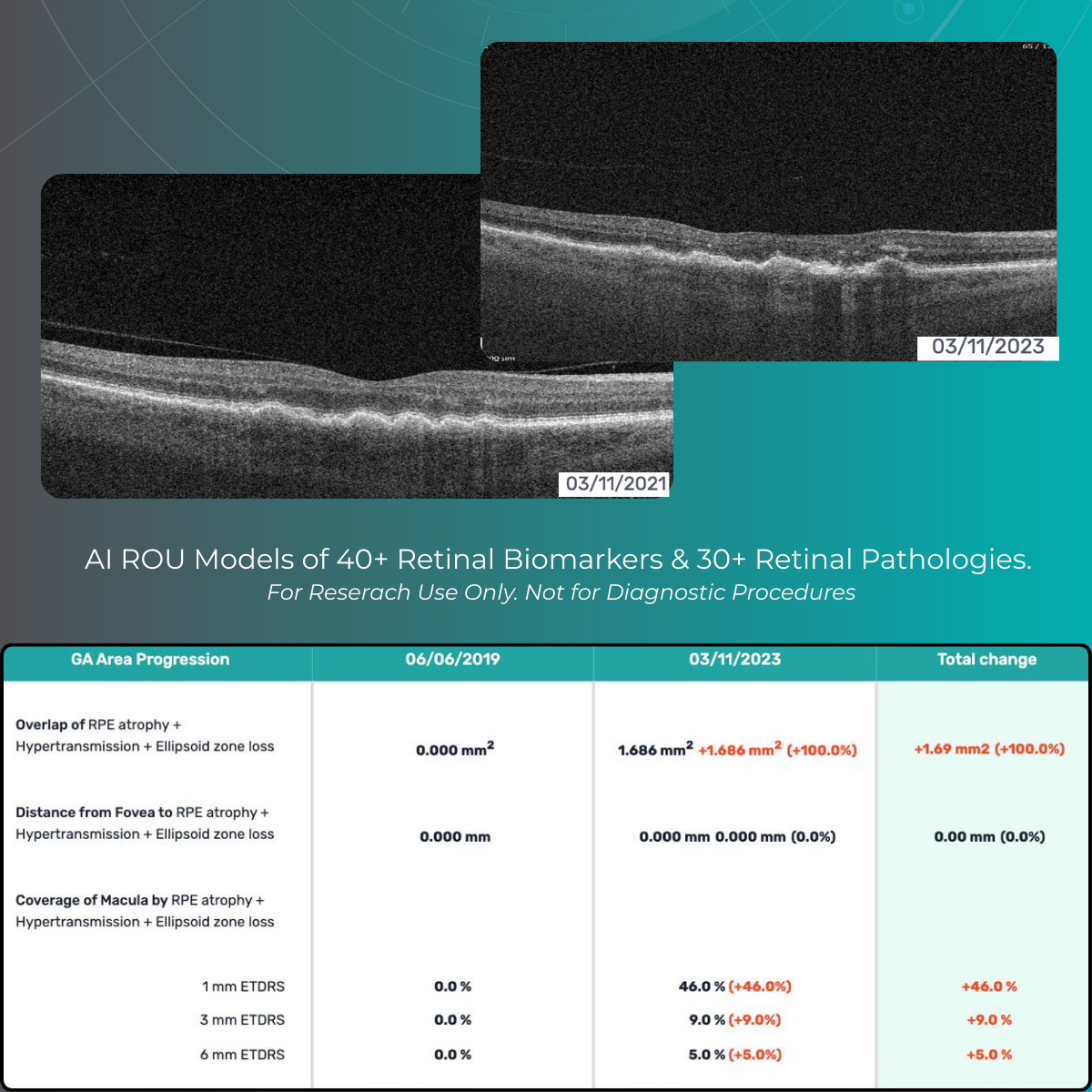
Quantify and analyze 40+ retinal biomarkers relevant to reserach of 30+ retinal conditions
Explore 40+ retinal biomarkers studied across major retinal research areas (Dry and Wet AMD, DR, DME, RVO).
For Research Use Only.Not for use in diagnostic procedures.
Support research site eligibility workflows using AI ROU models for 40+ retinal biomarkers & for 30+ retinal conditions
Gather analytics on biomarkers, patient imaging data, and treatment patterns for your RW research
Power your research on 40+ retinal biomarkers in relation to 30+ retina conditions
Quantify and analyze 40+ retinal biomarkers relevant to reserach of 30+ retinal conditions
Explore 40+ retinal biomarkers studied across major retinal research areas (Dry & Wet AMD, GA, DR, DME, RVO).
For Research Use Only.Not for use in diagnostic procedures.
Unlock vendor-neutrality: work with data from 9 OCT manufacturers in Altris IMS
Organize historical OCT data
Power your research on 40+ retinal biomarkers in relation to 30+ retina conditions
Analyze 40+ retina biomarkers — supporting research on 30+ retinal pathologies
Explore 40+ retinal biomarkers studied across major retinal research areas (e.g., AMD subtypes and DR).
For Research Use Only.Not for use in diagnostic procedures.
Results in numbers
Our company was created in 2017 and we achieved many milestones since then
Let's talk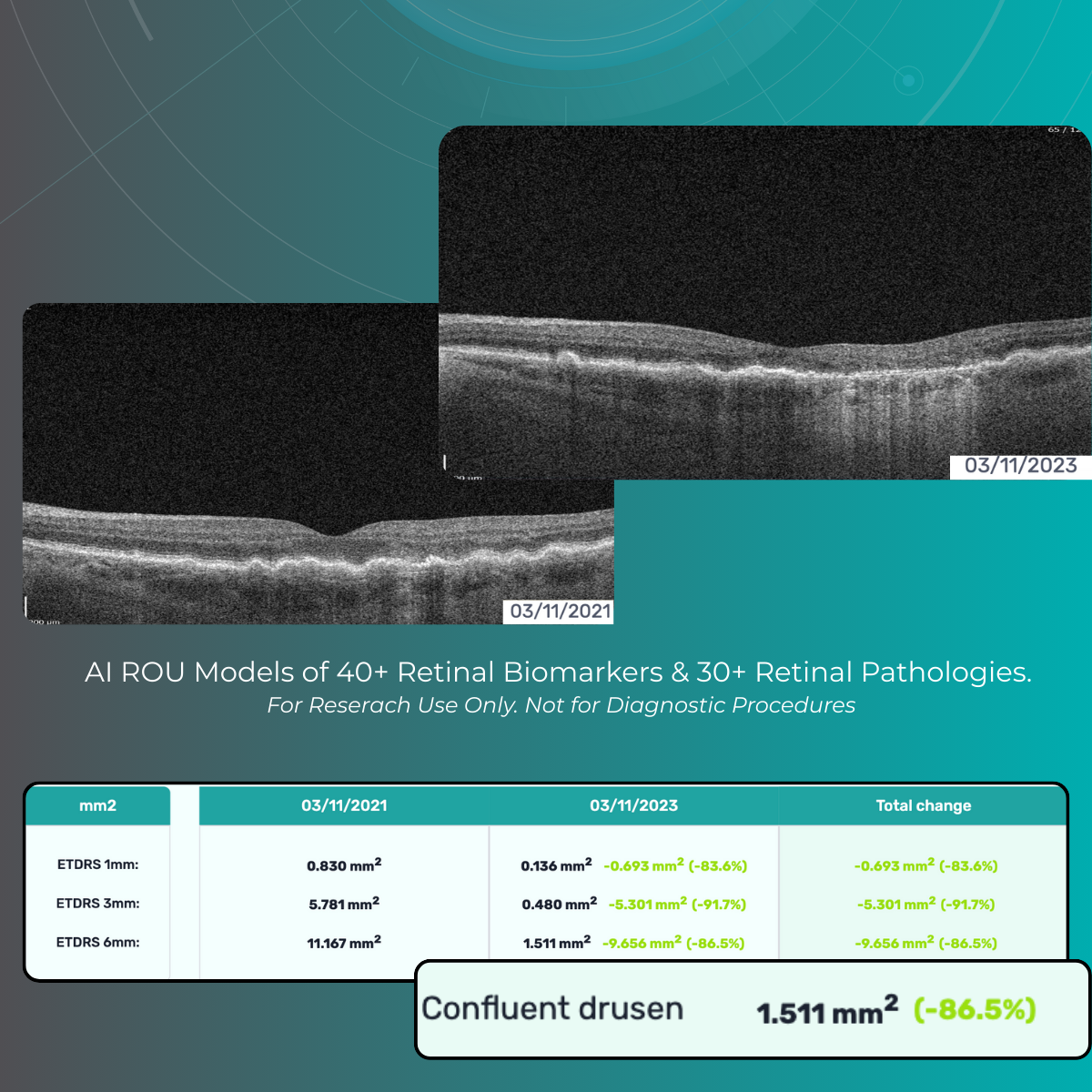
Organize your OCT data for Research
Explore 40+ retinal biomarkers studied across 30 major retinal research areas (e.g., Dry & Wet AMD, GA, DR, DME, etc.).For Research Use Only. Not for use in diagnostic procedures.
Analyze large data volumes of OCT, generating research-ready data
Quantify and analyze 40+ retinal biomarkers relevant to research on 30+ retinal conditions with the help of maps, graphs, and percentages.
For Research Use Only. Not for use in diagnostic procedures.
Import, store, and analyze OCT data from all major manufacturers in one secure, vendor-neutral environment
Transform raw OCT scans into research-ready data
Use formats convenient for your reserach goals
Aggregate OCT data across devices using Altris IMS to support multi-center or institutional research
Create custom pdf reports comprehensible for patients and convenient for eye care specialists
Enable multiple researchers to access and annotate OCT datasets simultaneously
For clinical research
Running clinical studies shouldn’t be slowed down by manual processes or fragmented data.
Contact Us- Centralized OCT Data Management
- Historical Data Analysis
- Research-Ready Ecosystem
- Vendor-neutral analysis of OCT scans ( 9 manufacturers)
- Data Security and Compliance
- Additional capabilities for research
- 40+ retinal biomarkers studied in research across 30+ retinal conditions. For Research Use Only. Not for diagnostic procedures.
- Quantitative exploration of 40+ biomarkers for Research Use Only. Not for diagnostic procedures.
- Centralized OCT Data Management
- Vendor-Neutral OCT Compatibility ( 9 manufacturers)
- Secure and Compliant Data Environment
- Seamless Clinical Workflow Integration
- Historical data analysis
- 40+ retinal biomarkers studied in research across 30+ retinal conditions. For Research Use Only. Not for diagnostic procedures.
- Quantitative exploration of 40+ biomarkers for Research Use Only. Not for diagnostic procedures.
- Data Security and Compliance






Project Manager at FinGenn