Recently Posted
-

Increasing Referral Efficiency in Eye Care: Addressing Data Gaps, Wait Times, and more
 Maria Martynova
04.07 20237 min read
Maria Martynova
04.07 20237 min readOphthalmology has the highest average number of patients waiting, but up to 75% of patients make preventable trips to eye hospitals and general practitioners. Some of these patients are referred by optometrists who, more often than not, receive no feedback on the quality of their referrals, perpetuating this cycle. Optometry referral is puzzling for both primary and secondary education. This article examines the referral procedure and potential solutions for increasing referral efficiency in eye care that practitioners can implement.
More than 25% of U.S. counties lack a single practicing eye care provider, and the situation isn’t unique to the U.S. In the UK, ophthalmology has been the most overburdened healthcare sector for some time. With a globally aging population and an increasing prevalence of age-related diseases, ensuring accessible eye care is crucial. Unfortunately, the reality is quite the opposite. One contributing factor is the high number of failures in the referral process.
How did we arrive at this point, and what can be done to improve it?
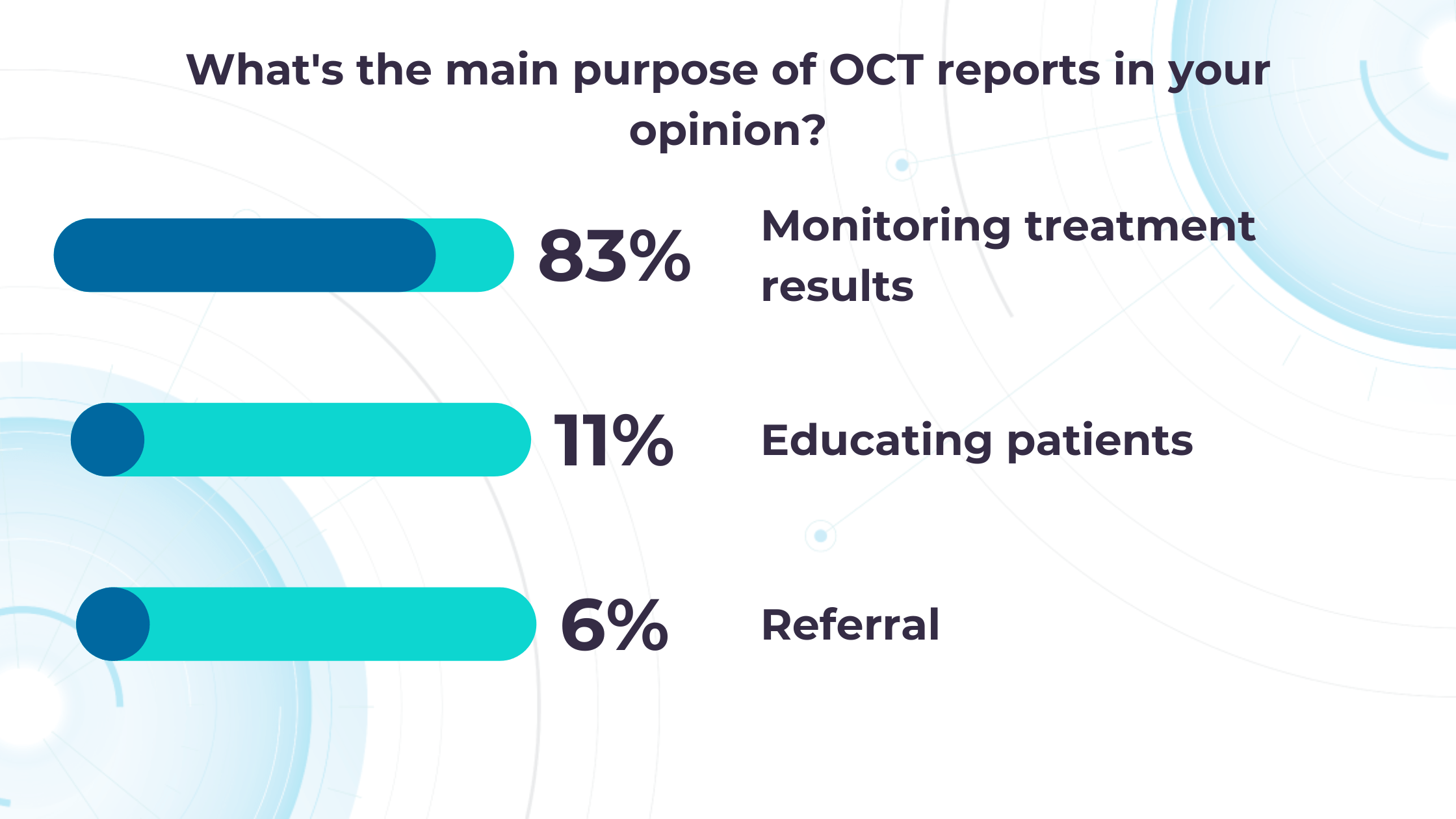
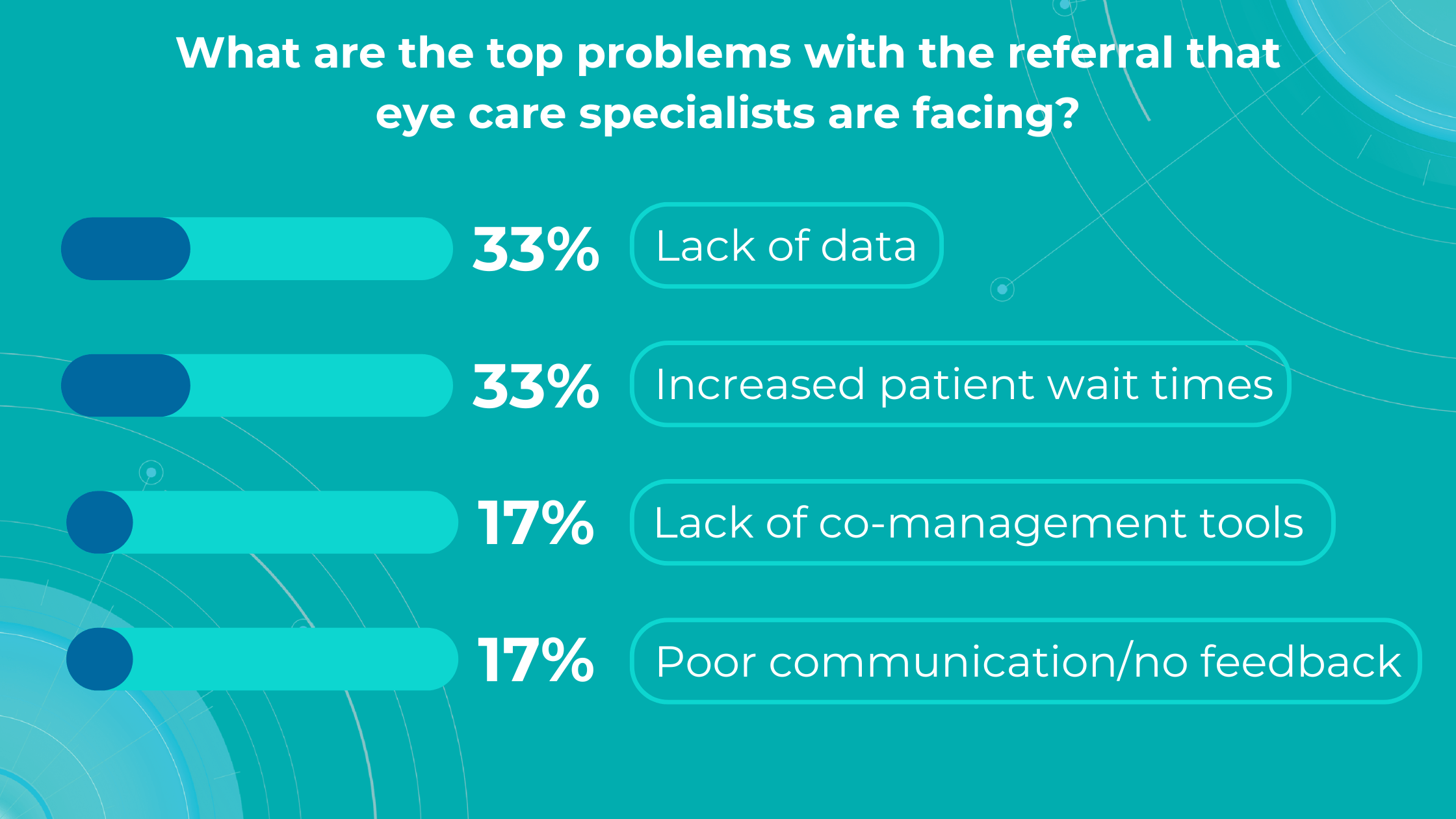
Altris AI’s survey identified a lack of data and increased patient wait times as the top problems with referrals for practitioners, while lack of co-management tools and poor communication/feedback ranked lower.

Let’s dive into more details:
Optometry referral: top problems
-
Lack of diagnostic data
The ultimate goal of optometry referral is to ensure patients receive appropriate treatment for their specific pathology or confirmation of its absence. The receiving specialist’s first step is to review the referral report, making its completeness and clarity paramount. While there is a clear need for specialised assessment and treatment, almost 80% of those attending eye casualty do not require urgent ophthalmic attention following triage, and up to 60% of patients are seen and discharged on their first visit.
In eye care, both text information and accompanying images are crucial in ensuring efficient and accurate diagnoses.
However, handwritten and fragmented data continue to pose significant challenges in the patient referral process. Despite the prevalence of electronic health records (EHRs), over half of referrals are still handled through less efficient channels like fax, paper, or verbal communication. This can lead to fragmented or doubled patient data, potential gaps in care, and delays in treatment.
The study on the Impact of direct electronic optometric referral with ocular imaging to a hospital eye service showed that, given some limitations, electronic optometric referral with images to a Hospital Eye Service (HES) is safe, speedy, efficient, and clinically accurate, and it avoids unnecessary HES consultations.

Direct electronic referrals with images reduced the need for hospital eye service appointments by 37% compared to traditional paper referrals. Additionally, while 63% of electronic referrals led to HES appointments, this figure was 85% for paper referrals.
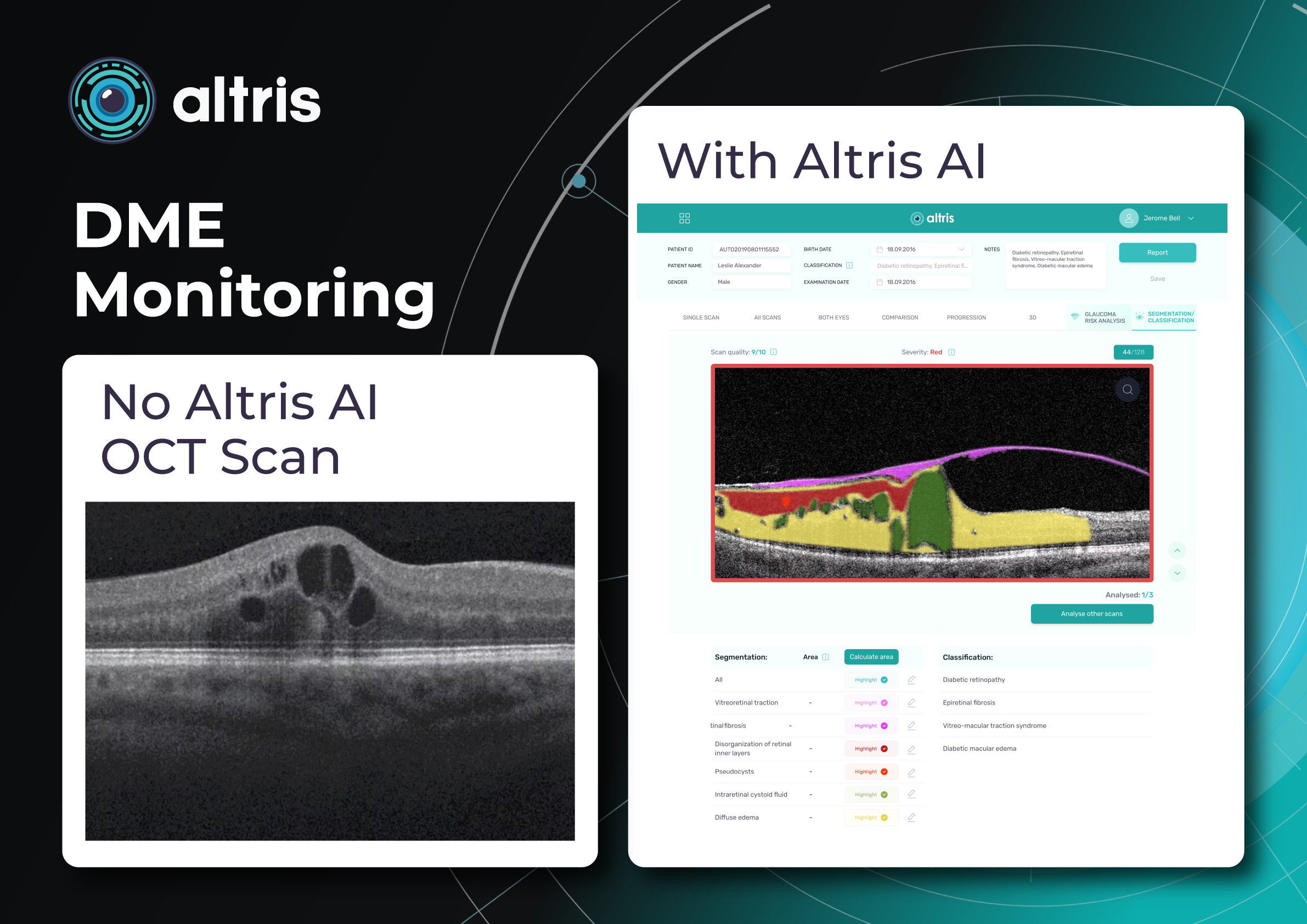
While incorporating images like OCT scans can significantly enhance understanding, some subtle or early-stage pathologies might still be overlooked. This is where detailed and customized reports become invaluable.
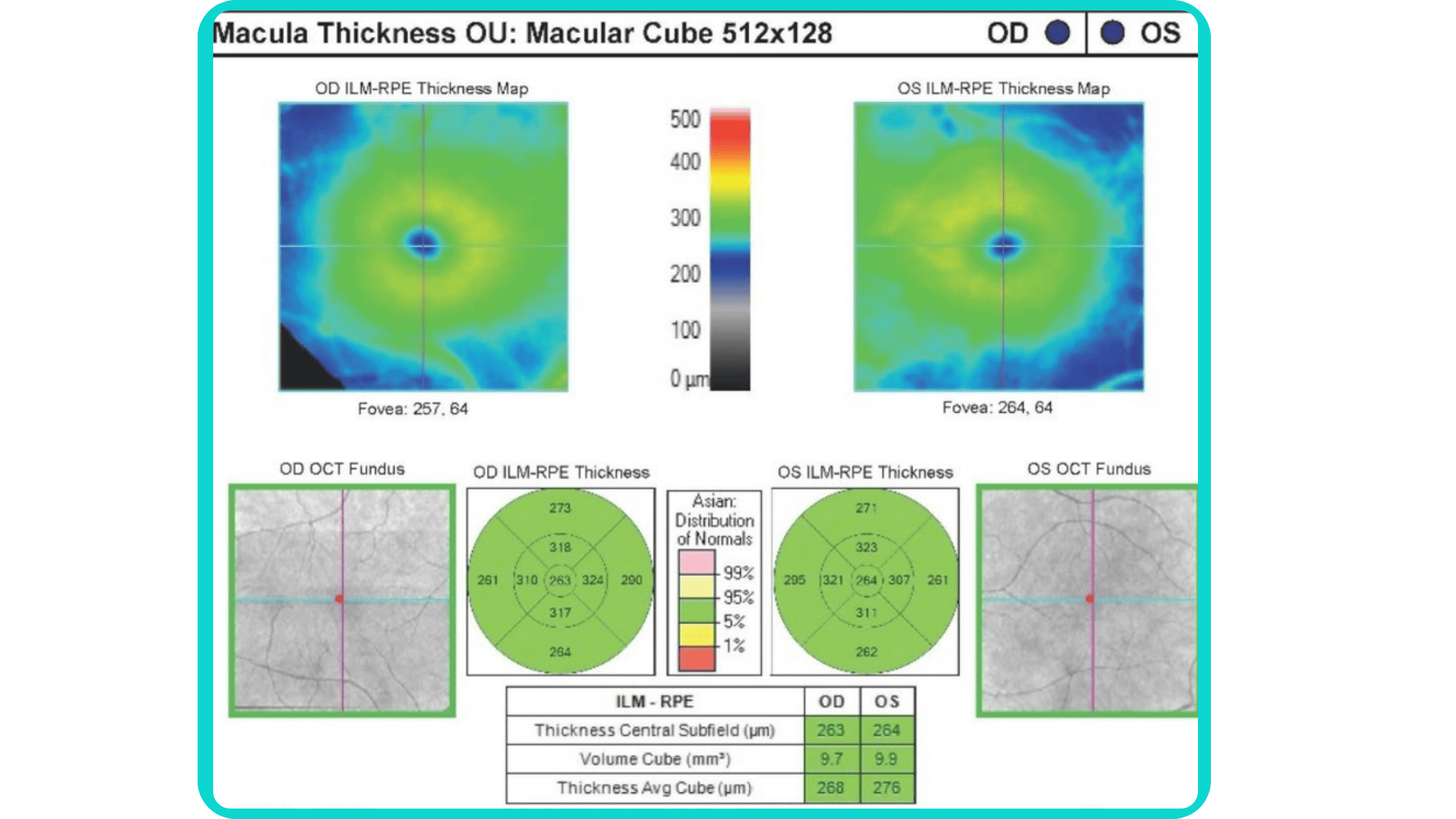
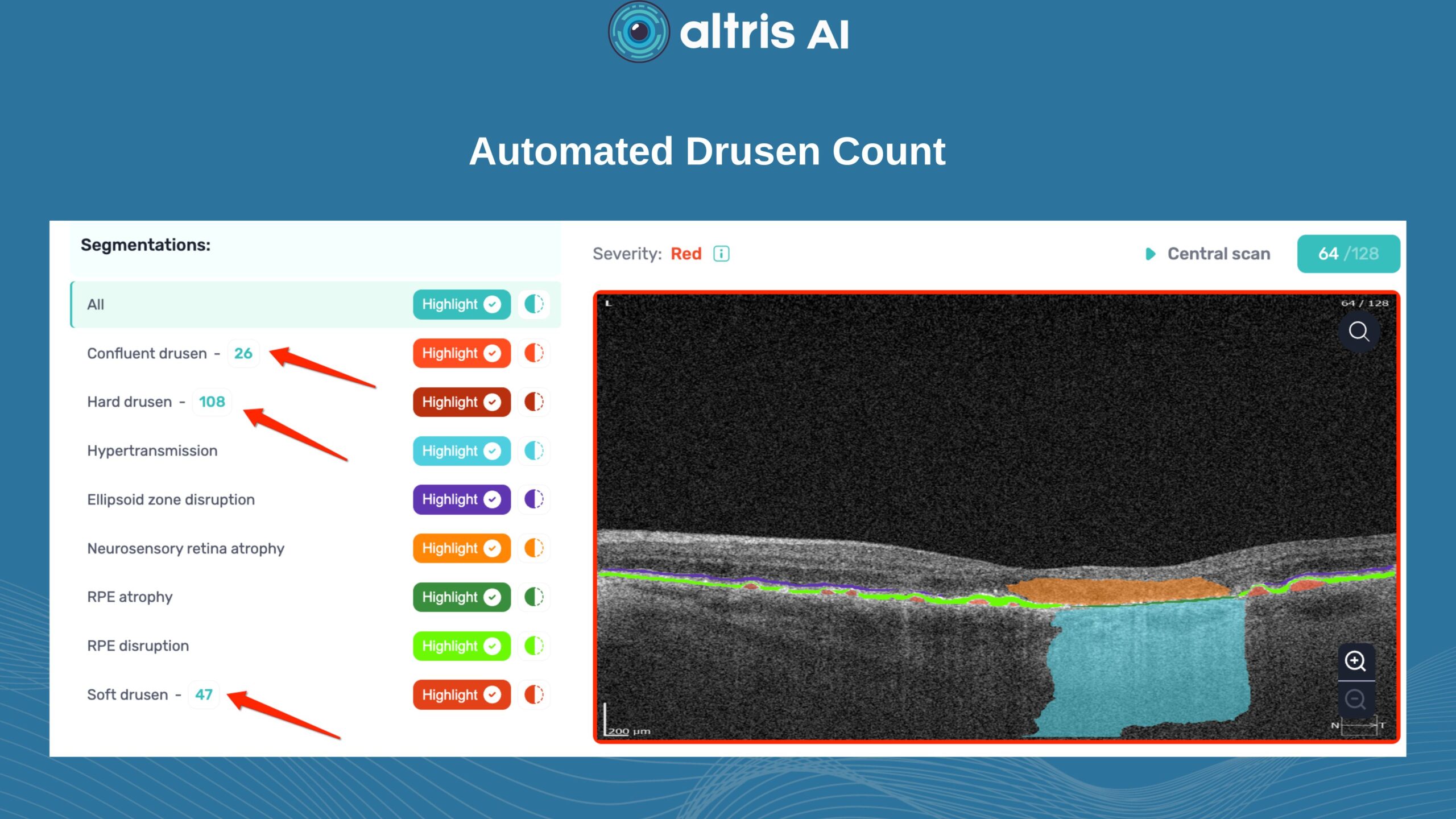
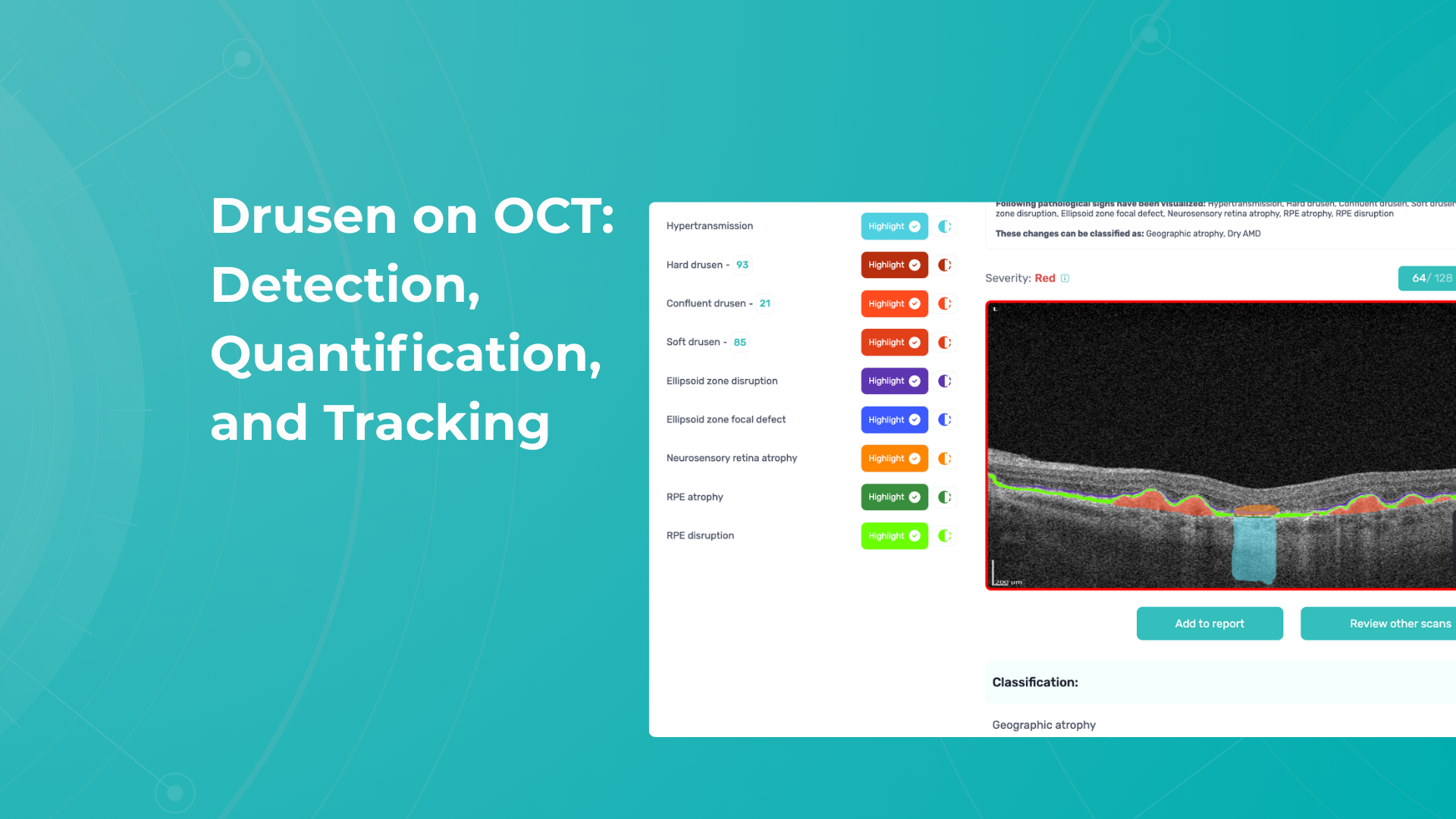
To illustrate the point, here is a handwritten referral compared to one of the types of customised OCT report from the Altris AI system, a platform that automates AI-powered OCT scan analysis for 70+ pathologies and biomarkers. This screenshot, in particular, shows segmented retina layers and highlights biomarkers of Dry AMD alongside a comparison of the patient’s macular thickness over visits.
-
Lack of experience and access to a second opinion
Research reveals a notable inverse relationship between clinician experience and the frequency of false-positive referrals in optometry, echoing findings in other medical fields where diagnostic proficiency typically improves with experience. This highlights the importance of recognizing the learning curve inherent in optometric practice and supporting less experienced practitioners.
The challenge is amplified by the fact that optometrists often practice in isolation, lacking the immediate professional support network available to their hospital-based counterparts. Unlike colleagues in hospital settings who have ready access to peer consultation for other opinions or guidance, optometrists often face limited opportunities for collaborative decision-making and skill development.
Another problem specialists often face is a lack of confidence in diagnosing, which may or may not be linked to experience. Knowing that their patients could potentially suffer irreversible vision loss from a pathology not yet detected during an exam, they often err on the side of caution and refer to a hospital. While this “better safe than sorry” approach is understandable, it places a significant burden on hospitals, extending wait times for those already at risk of blindness.
These concerns primarily revolve around glaucoma, age-related macular degeneration (AMD), and diabetic retinopathy (DR). AI can help identify these and other eye diseases at their earliest stages during routine visits. Some retinal changes are so minute that they escape detection by the human eye, making the program’s ability to detect tiny retinal changes invaluable.
Another significant benefit of AI systems lies in their approach to OCT analysis for glaucoma. Traditional methods rely on normative databases to assess retinal normality, but these databases are often limited in size and represent a select group of individuals. This can result in missed diagnoses of early glaucoma in those who deviate from the “norm” or unnecessary referral from optometry to ophthalmology for those who don’t fit the “normal” profile but have healthy eyes. AI can overcome this limitation by providing more personalized and comprehensive analysis.
-
Increased wait times for patients with eye doctor referral

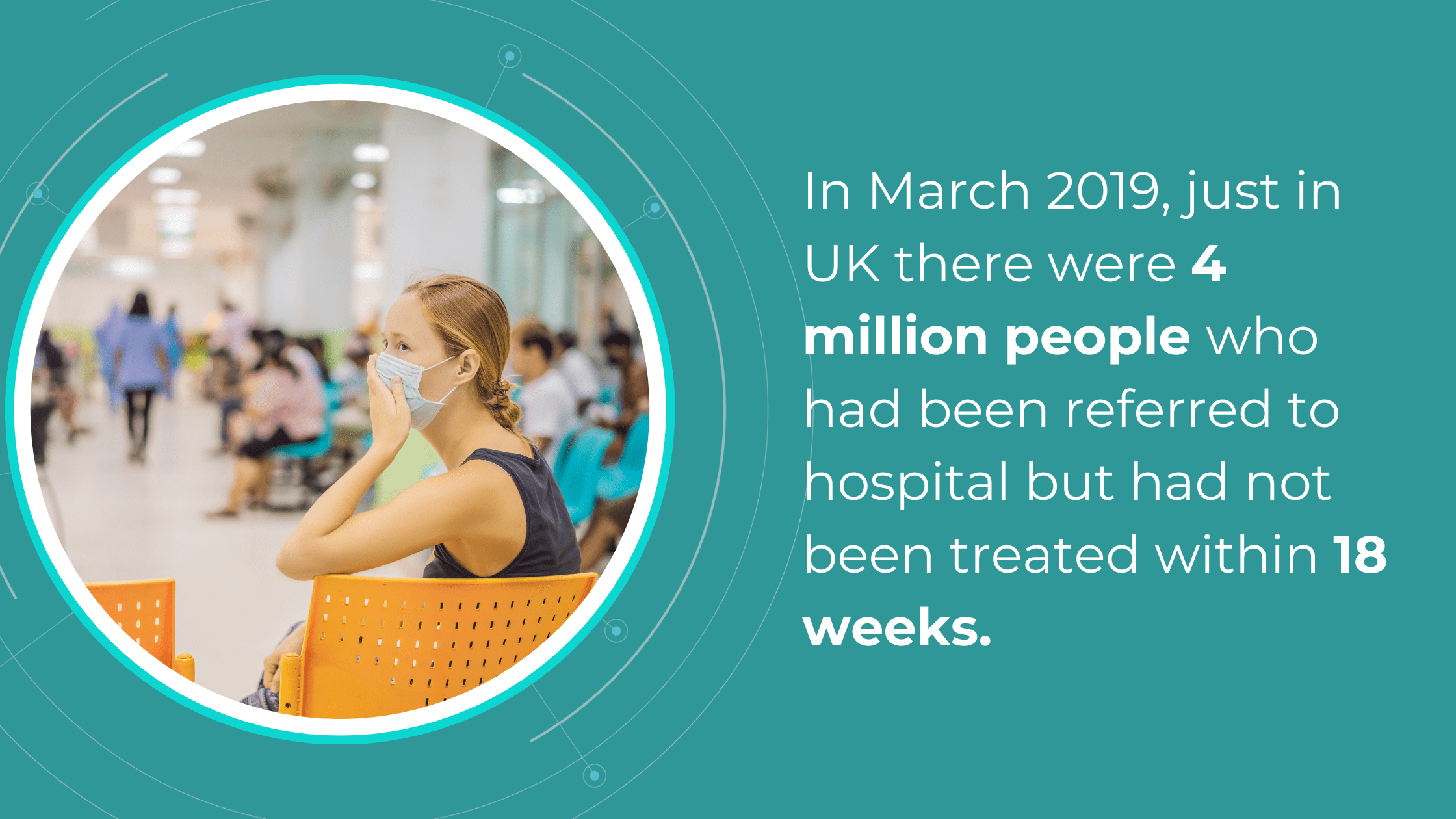
The National Health Service (NHS) is grappling with significant backlogs in ophthalmology services, which account for nearly 10% of the 7.8 million patients awaiting treatment.
The consistently high average number of patients waiting per trust in Ophthalmology, with high follow-up waitlists, delays care that poses substantial risks. The Royal College of Ophthalmologists reported that the risk of permanent visual loss is nine times higher in follow-up patients than in new patients. With 30% more patients on ophthalmology waitlists than pre-pandemic, the number of people at risk of sight loss may have increased.

Community Eyecare (CHEC), a provider of community-based ophthalmology services, received around 1000 referrals per week before the pandemic, further highlighting the strain on the system.
An analysis of electronic waitlists revealed that administrative issues, such as deceased patients or those already under care remaining on the list, artificially inflate wait times by up to 15%.
Improving administrative processes and reassessing referrals for appropriateness could help address this problem. Additionally, interim optometric examinations could revise referral information or determine the necessity of hospital visits, further reducing wait times.
Artificial intelligence can significantly speed up the screening process while reducing the controversy around diagnoses. This faster and more accurate diagnostic tool will enable more patients to be seen, allow for quicker responses to pathologies that pose a risk to eyesight, and reduce the burden on strained hospitals with needless patient referrals, as well as free up patients from unnecessary stress and wasted time.
International studies have shown that collaborative care also can increase screening and detection rates of eye disease.
-
Lack of comanagement tools for optometry referral
The increasing demand for Hospital Eye Services, projected to grow by 40% in the next two decades and currently accounting for 8% of outpatient appointments, necessitates a re-evaluation of referral pathways and comanagement strategies between optometrists and ophthalmologists.
The lack of digital connectivity between primary, community, and secondary care creates a significant barrier to effective collaboration. In many cases, optometrists cannot make direct digital referrals to Hospital Eye Service, often relying on general practitioners as intermediaries, causing delays in diagnosis and treatment.
The COVID-19 pandemic highlighted the vital role of optometrists as first-contact providers for eye health, relieving pressure on hospitals. However, better integration between primary and secondary care is essential to build upon this and create a more sustainable eye care system. The current lack of digital connectivity hinders efficient communication and impedes the timely transfer of patient records, potentially leading to unnecessary referrals and delays in care.
 As David Parkins, the ex-president of the College of Optometrists, emphasizes, the solution lies in increased integration and streamlined communication between primary and secondary eye care services. Implementing integrated digital platforms for referrals and feedback can enhance collaboration, improve patient outcomes, and reduce the burden on hospitals.
As David Parkins, the ex-president of the College of Optometrists, emphasizes, the solution lies in increased integration and streamlined communication between primary and secondary eye care services. Implementing integrated digital platforms for referrals and feedback can enhance collaboration, improve patient outcomes, and reduce the burden on hospitals.Leveraging optometrists’ expertise through shared care programs and direct digital referral pathways can alleviate the strain on eye hospitals and ensure timely access to care for patients with eye conditions.
-
Referral to Ophthalmology: Poor communication/lack of feedback
A recent study published in Ophthalmic and Physiological Optics revealed that in 73% of cases, the referring optometrist was unaware of the outcome of their referral.
This lack of closure can lead to unnecessary re-referrals, patient anxiety, and potential treatment delays that could result in preventable vision loss, especially considering the extended waiting times for hospital eye service appointments.
Effective referral in eye care requires a closed feedback loop, where referring providers receive timely updates and reports from specialists. However, studies have shown that up to 50% of primary care providers (PCPs) are unsure whether their patients have even been seen by the referred specialists. This disconnect necessitates time-consuming follow-up calls and manual data integration, increasing the risk of errors and jeopardizing patient care.
The absence of consistent feedback also impacts optometrists’ professional development. Without knowing the accuracy of their referrals, optometrists cannot identify areas for improvement or refine their diagnostic skills. This is particularly relevant for newly qualified practitioners who may benefit from feedback to enhance their clinical judgment.
Implementing electronic referral systems that include feedback mechanisms can significantly improve communication and close the feedback loop. This would enable optometrists to track the progress of their referrals, receive timely updates on patient outcomes, and make informed decisions about future referrals.
Technology is also bridging the gap in specialist communication by enabling secure online consultations, such as live chat with dedicated ophthalmologists. A notable example in the UK is Pocket Eye, a platform designed to empower eye care professionals with clinical advice, diagnostic and image support, and AI-powered OCT analysis.

Summing up
Implementing digital platforms that foster collaboration between eye care providers, increasing confidence in complex cases, and utilizing AI technologies to expedite diagnostics is crucial in a world where an aging population will increasingly rely on healthcare. Referral to ophthalmology from optometry should be effective, fast, and painless to eye care specialists and patients.
Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.
-
-

OCT Reports: Enhancing Diagnostic Accuracy
 Maria Martynova
07.06. 20238 min read
Maria Martynova
07.06. 20238 min readThe average OCT device is a significant investment, costing upwards of $40,000. As eye care specialists, we recognize the revolutionary power of OCT. However, patients often receive only a standard OCT report from this investment. Unfortunately, many patients are unaware of OCT’s true value and may not even know what it is. This raises a crucial question: are these standard reports truly reflecting the full diagnostic potential of such an expensive and sophisticated device? Are we, as professionals, maximizing the capabilities of this technology to ensure optimal patient care?
This article explores how OCT Reports address these shortcomings, enhancing diagnostic accuracy, treatment monitoring, referral efficiency, patient education, and audit readiness.
Common OCT reports and their limitations
Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.
How does the standard report look?

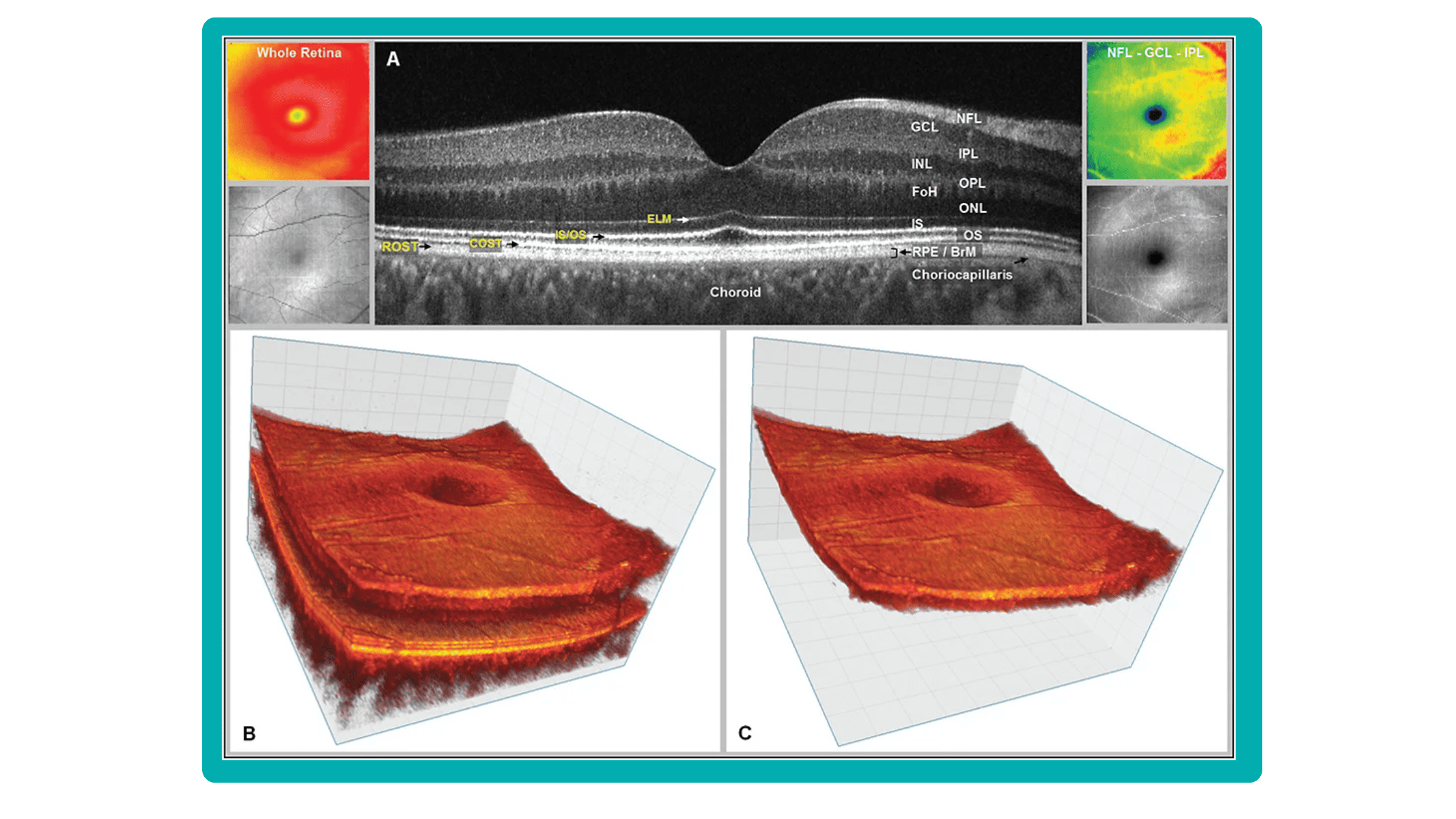
OCT has become a golden standard for diagnosing and monitoring many ocular pathologies, thanks to its unparalleled level of detail in ophthalmic imaging.
While retinal reports vary among OCT models, they typically include:- a foveally centered B-scan,
- a quantitative thickness map,
- and a semi-quantitative thickness map.
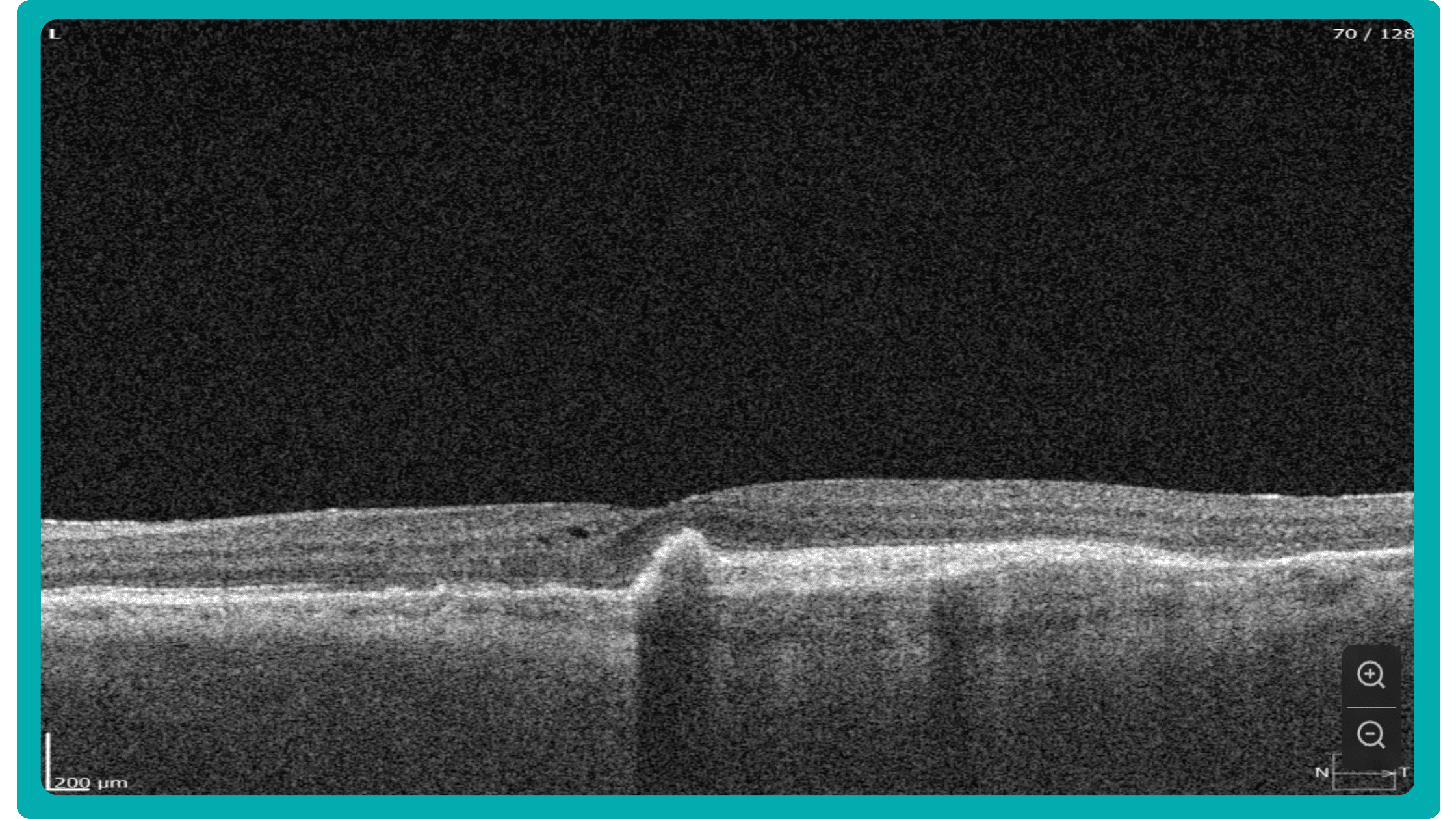
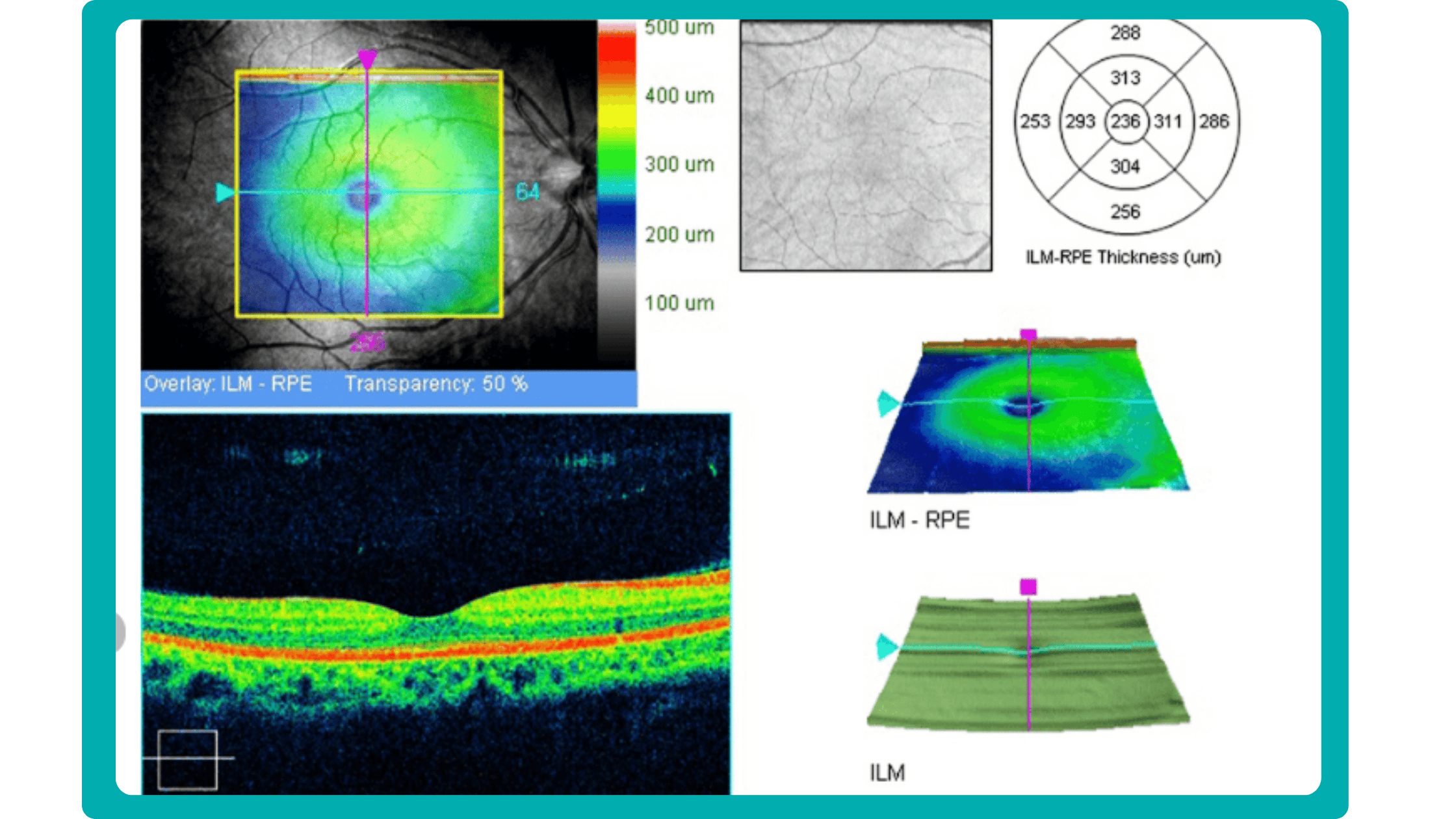
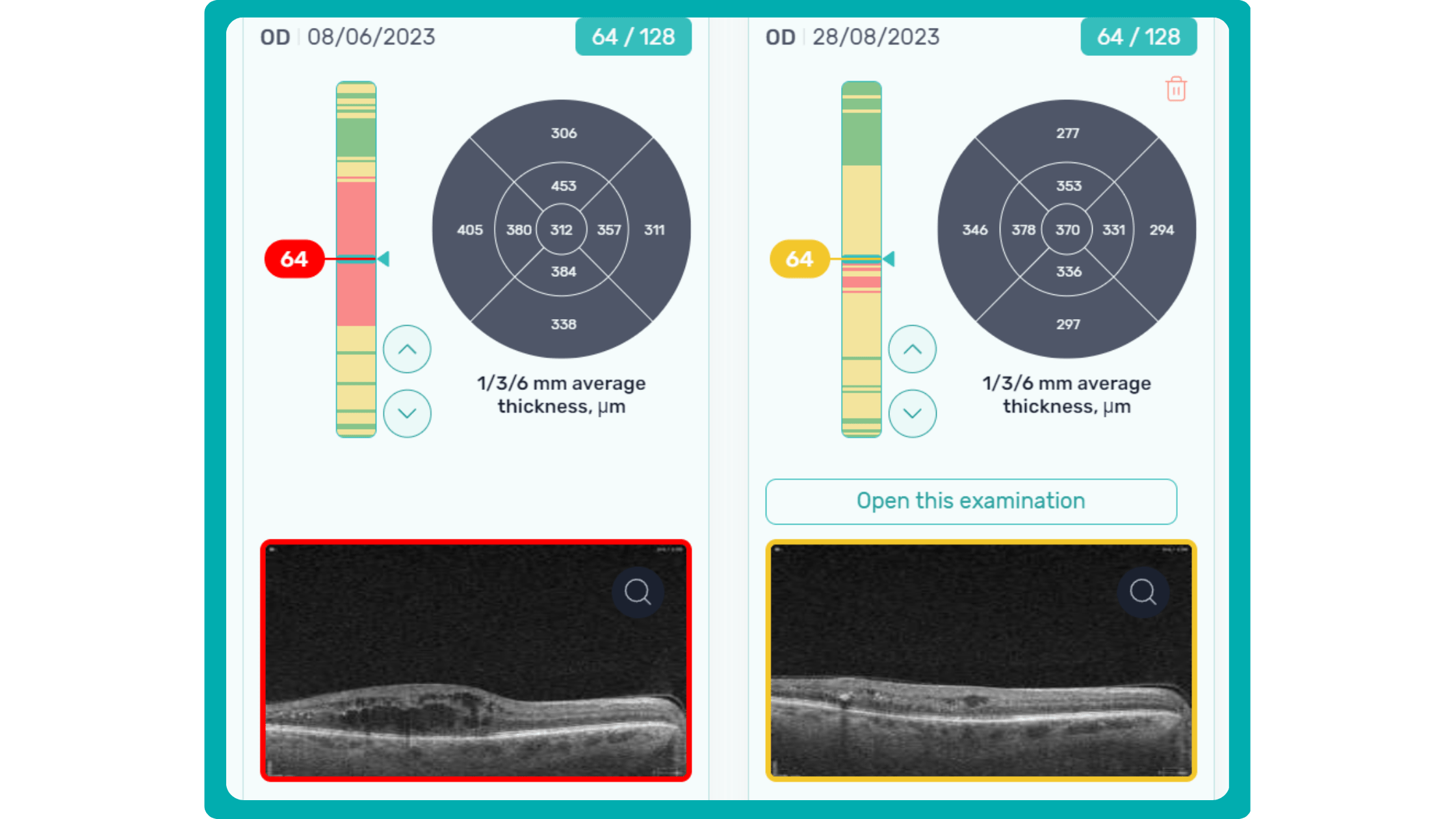
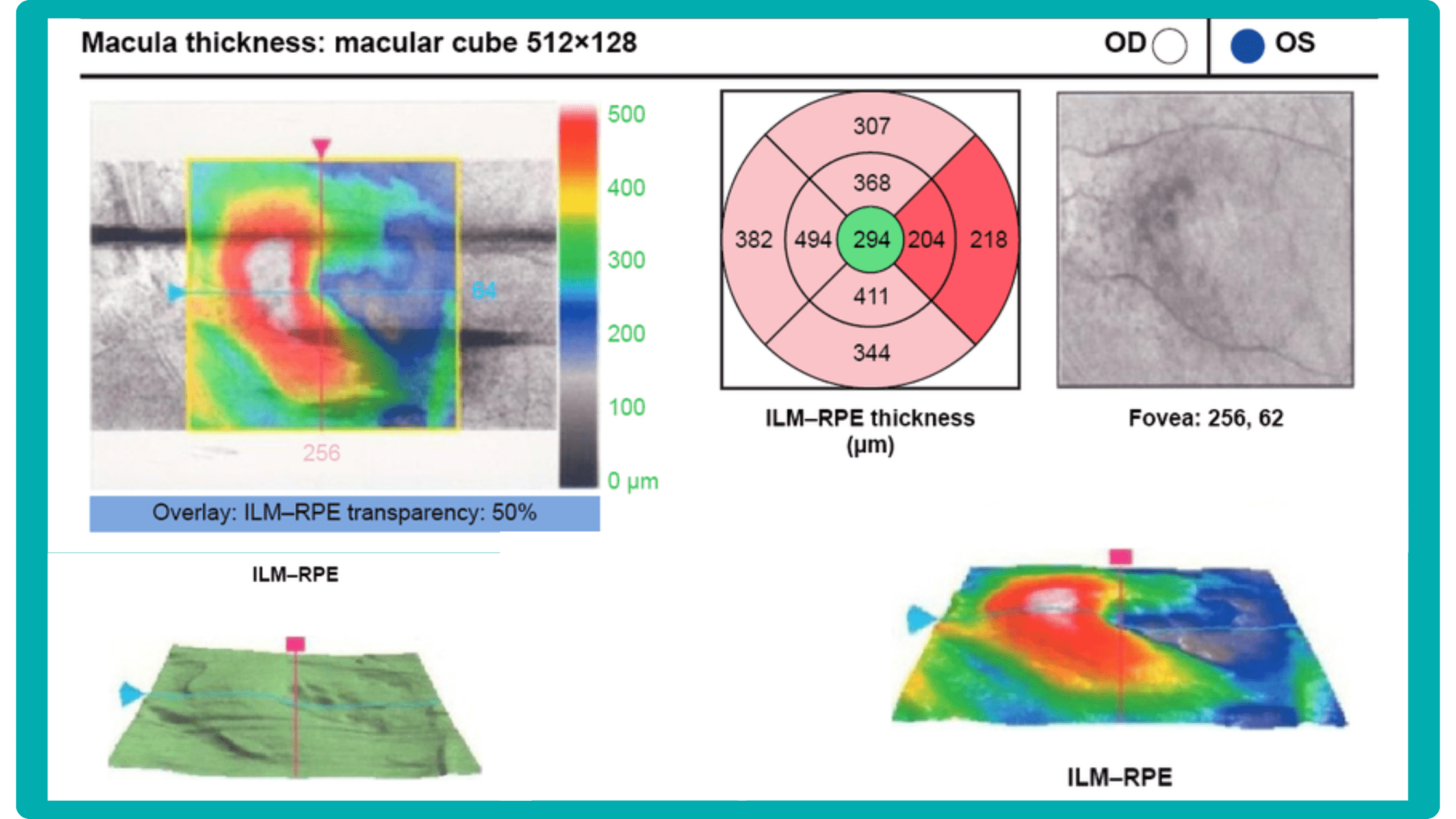
The B-scan offers a visual snapshot of foveal architecture and confirms proper scan centering. The quantitative thickness map employs the ETDRS sector map to measure retinal thickness within a 6mm circle around the fovea, with specific measurements for the foveal sector (1mm), inner macular ring (3mm), and outer macular ring (6mm).
Progression analytics enable comparison of serial macular scans, which is invaluable for managing vitreomacular interface disorders and macular edema. The semi-quantitative thickness map provides a broader overview of retinal thickness throughout the scan.
Given this amount of data, it is challenging to identify subtle and localized retinal pathological changes. As a result, entire OCT datasets are represented by few aggregated values, and the standard OCT reports generated by most devices often rely on significant data reduction to simplify interpretation, which you can usually not customize.
OCT report interpretation: 3 methods exist for displaying OCT data
Firstly, acquired 2D image slices are presented individually. This allows for detailed examination, but navigating through numerous images can be cumbersome, particularly with large datasets.

Secondly, a fundus image is displayed with superimposed retinal layers. This facilitates linking layers to the fundus, but only one layer can be examined at a time, hindering the analysis of multiple layers simultaneously.

Thirdly, the OCT tomogram is visualized in 3D, providing a comprehensive overview, but adjusting the visual representation often has limitations. Additionally, combined 3D visualizations of the tomogram and layers are typically unavailable, potentially obscuring spatial relationships.

While existing reports offer diverse approaches to managing, analyzing, and presenting OCT data, each solution focuses on specific aspects and lacks customization. The situation becomes even more complex if scans come from different OCT devices, as manufacturers only provide software for the data for proprietary OCT scanners. Consequently, no approved way of viewing, analyzing, or comparing data from different manufacturers exists.
Furthermore, there are limited possibilities for implementing prototypes to perform such tasks since software libraries are provided with exclusive licenses and incomplete data specifications. Hence, managing and analyzing OCT data and relating them to other information are challenging and time-consuming tasks.
Often, supplementary software is utilized to overcome these limitations by providing additional information, visualizing and emphasizing data differently, and enabling the selection of relevant subsets.
How can customized reports for OCT help?

Altris AI’s recent survey has revealed that the key benefits of OCT technology for eye care specialists lie in treatment monitoring, patient education, and referral optimization.

-
Measuring treatment progress: biomarkers tracking, pathology progression
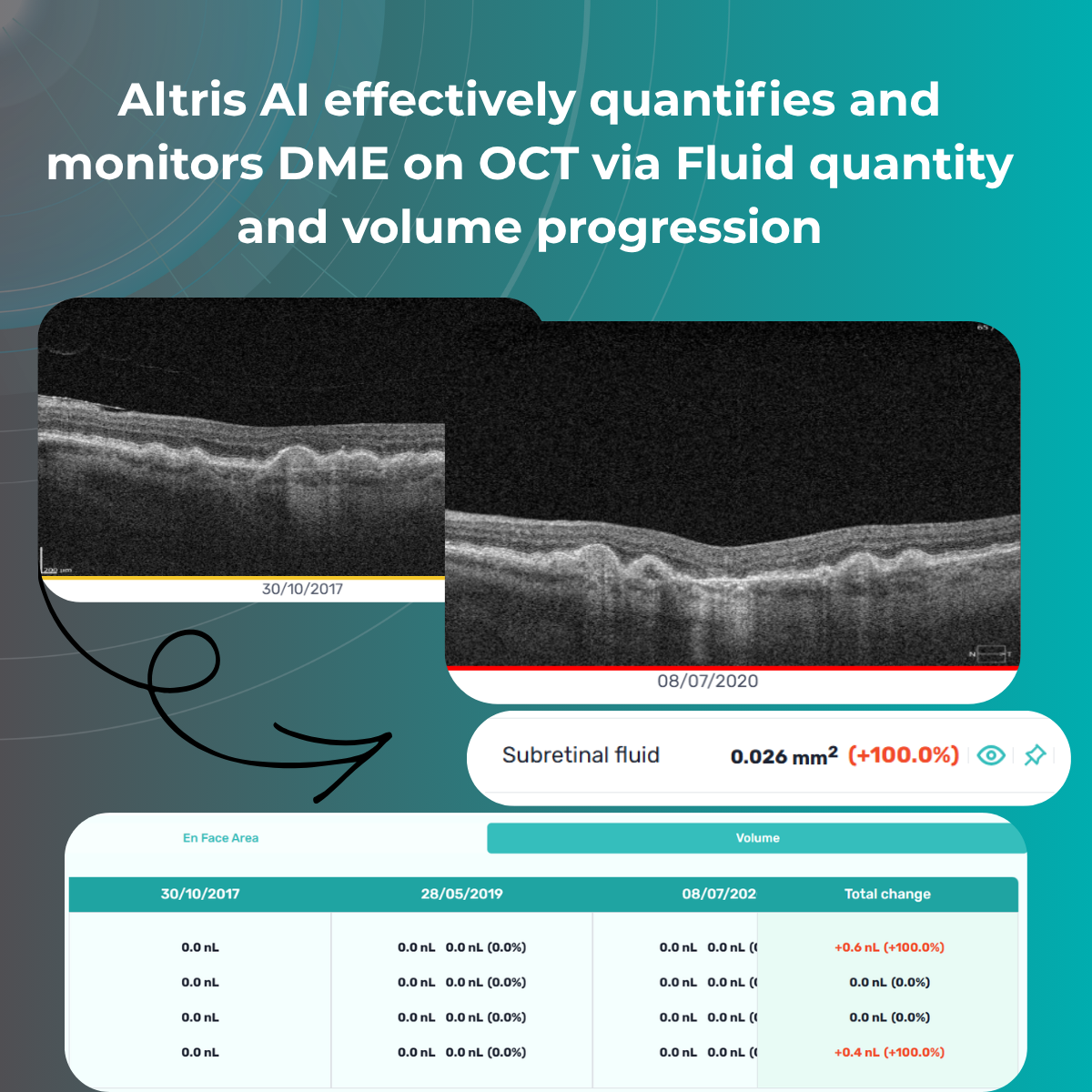
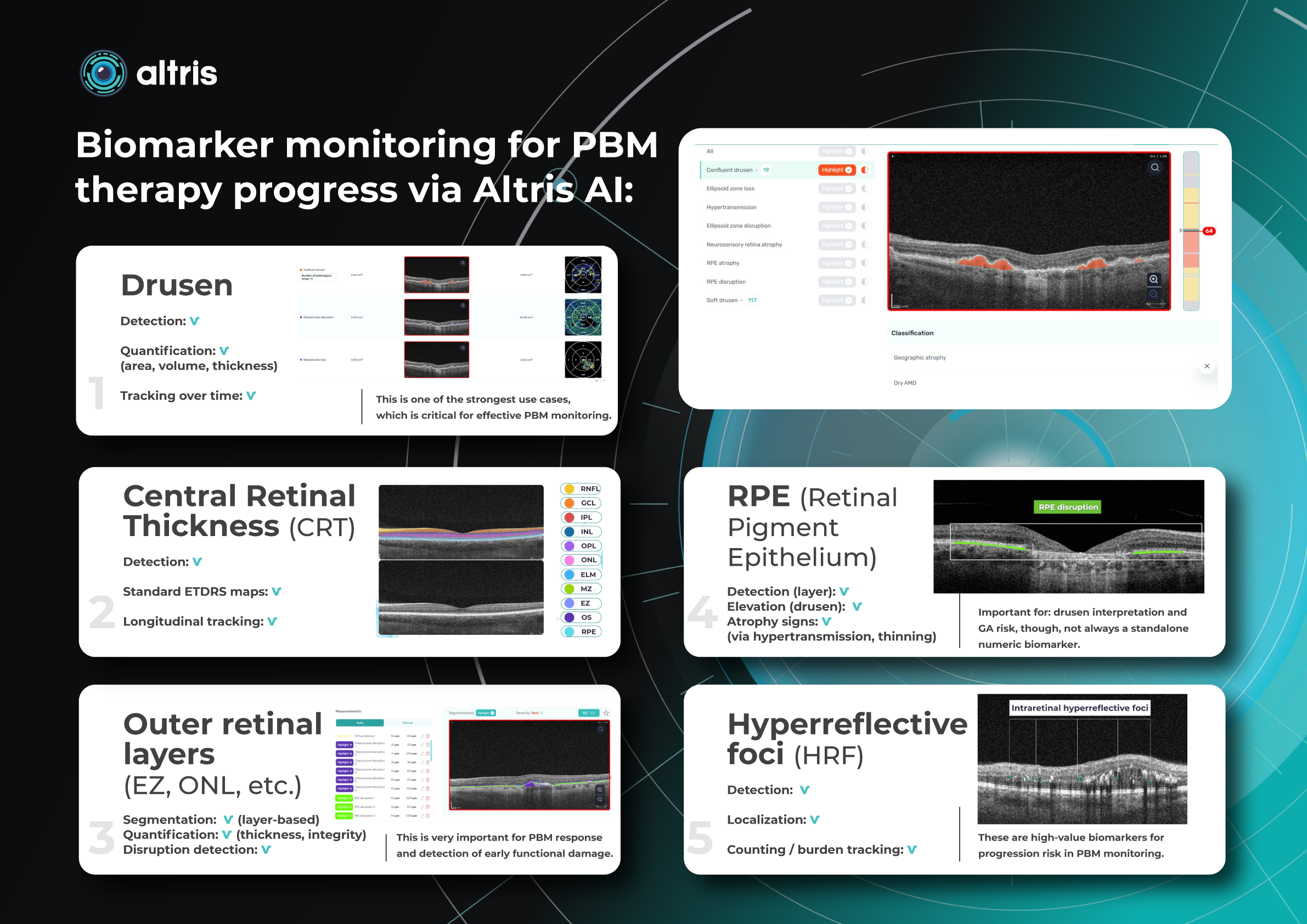
Imaging biomarkers are a particularly attractive option for clinical practice due to their non-invasive and real-time nature. Quantitative measurements of retinal thickness, fluid volume, and other biomarkers relevant to diseases like diabetic retinopathy and age-related macular degeneration aid in treatment monitoring.

OCT reports with customized measurements and selected biomarkers, retinal layers, or segments allow for precise focus on treatment monitoring and patient response to therapy. This personalized approach enhances clinical decision-making by highlighting each case’s most relevant information.

In current clinical practice, macular damage assessment typically involves measuring the distance between the ILM and RPE layers, summarized in a post-scan report.

However, these reports often fall short of visualization best practices, employing ineffective or inconsistent color schemes. Additionally, they lack flexibility, with static visuals preventing in-depth examination of specific details. Despite these limitations, these reports remain valuable for many clinicians by distilling complex data into a manageable format.
Enhanced OCT data visualization offers a promising solution to these challenges. It enhances report clarity and comprehensibility while preserving the richness of the underlying data.
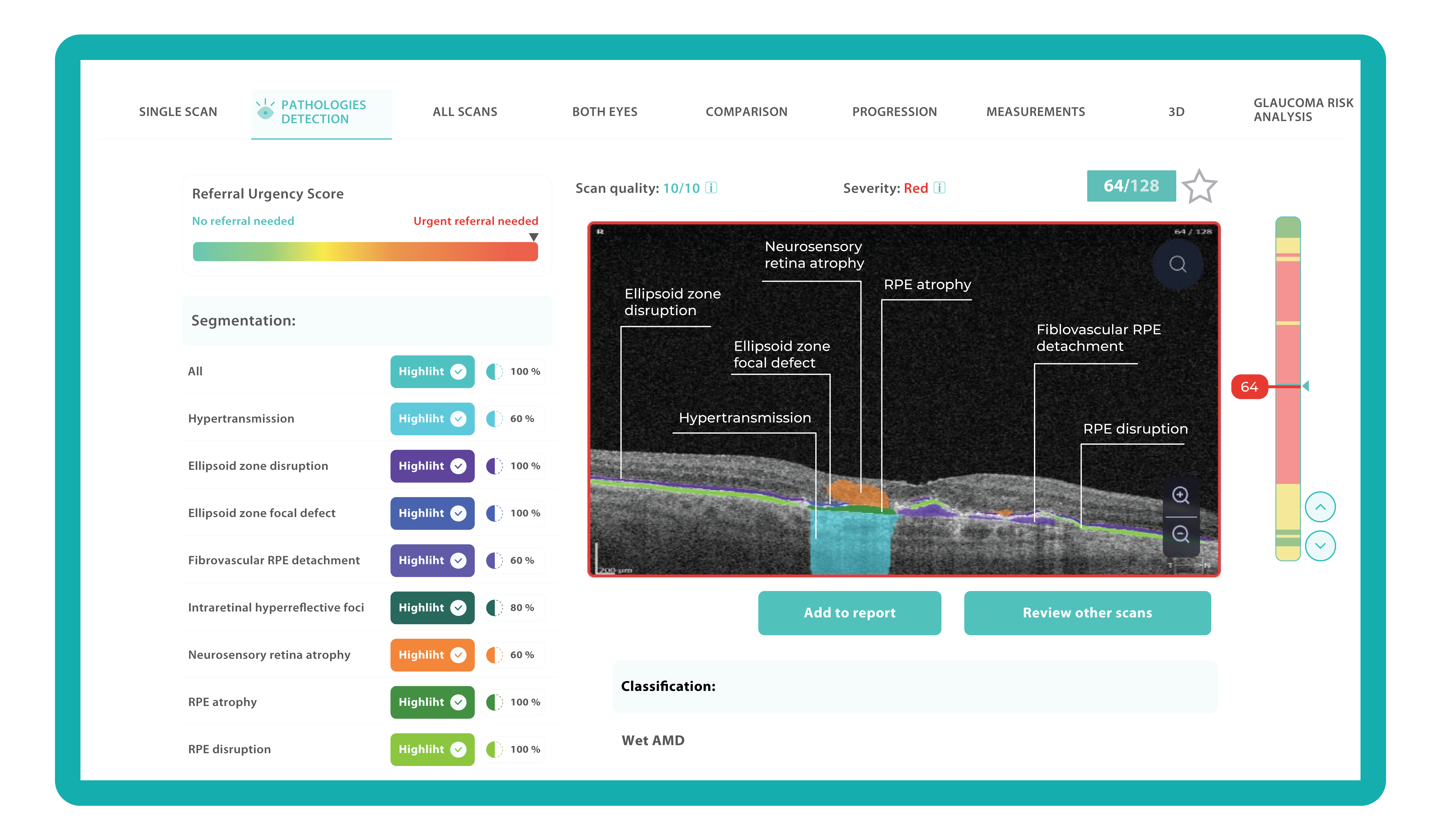
Let’s explore how this applies to a clinical case, such as monitoring a patient with Wet AMD during follow-up visits.

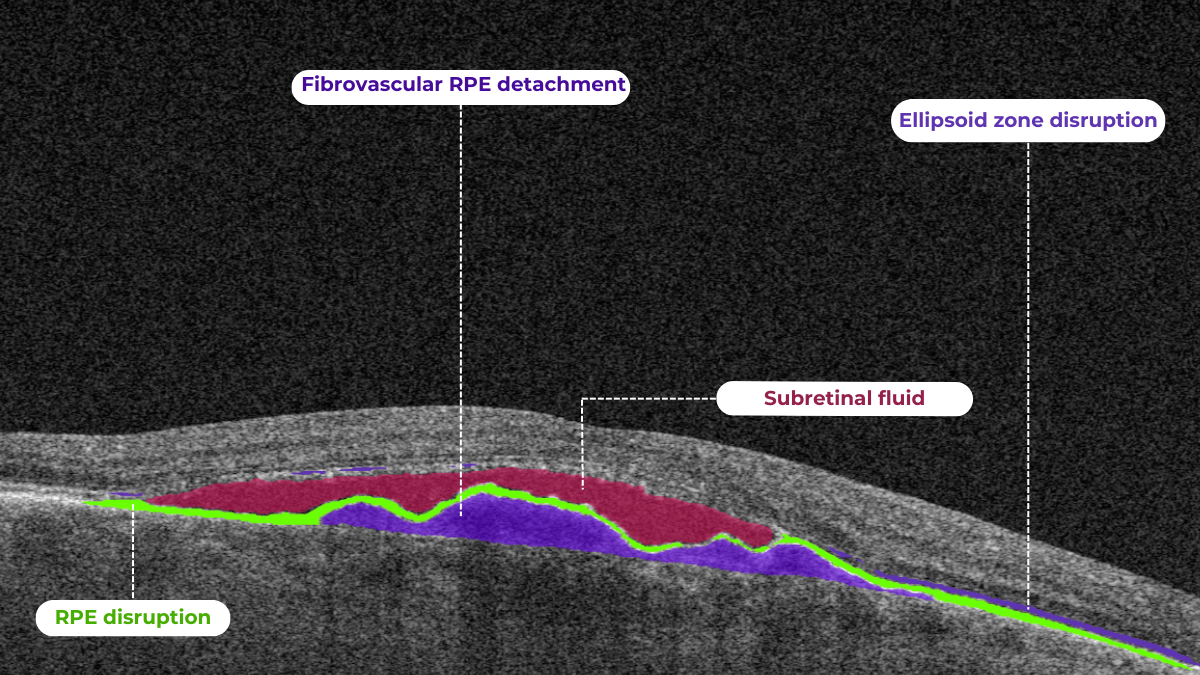
Data demonstrates that OCT findings can reveal the onset or progression of neovascular AMD before a patient reports new symptoms or changes in visual acuity. In fact, OCT images are reported to have the best diagnostic accuracy in monitoring nAMD disease states. This underscores the importance of key OCT findings or biomarkers in personalizing anti-VEGF treatment, achieving disease control, and reducing monitoring burdens.

Central Retinal Thickness emerged as one of the earliest OCT biomarkers used as an outcome measure in clinical trials for nAMD.
However, due to confounding factors, CRT’s use in outcome-based assessments of nAMD varies. Thus, it is essential to evaluate additional morphological changes alongside retinal thickness and their relationships with functional outcomes.
It has been reported that OCT images have the best diagnostic accuracy in monitoring nAMD disease states.
Another finding that is correlated with a worsening VA due to the associated photoreceptor defects is any damage to the four outer retina layers, including the RPE, interdigitation zone (IZ), ellipsoid zone (EZ), and external limiting membrane band (ELM).

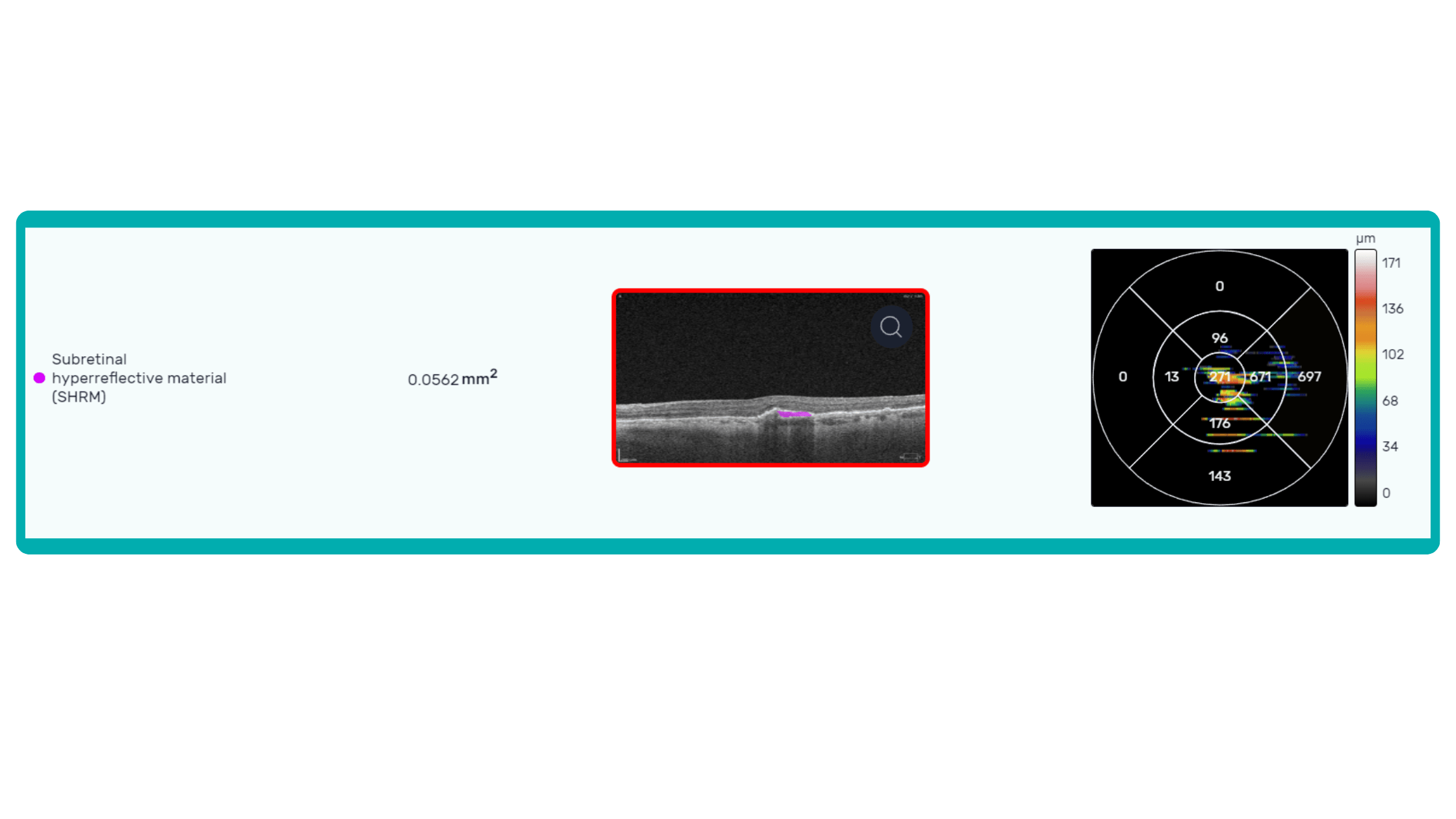
OCT is a valuable imaging tool for visualizing subretinal hyperreflective material (SHRM). It can automatically identify and quantify SHRM and fluid and pigment epithelial detachment to calculate the overall risk of worsening visual outcomes associated with SHRM.

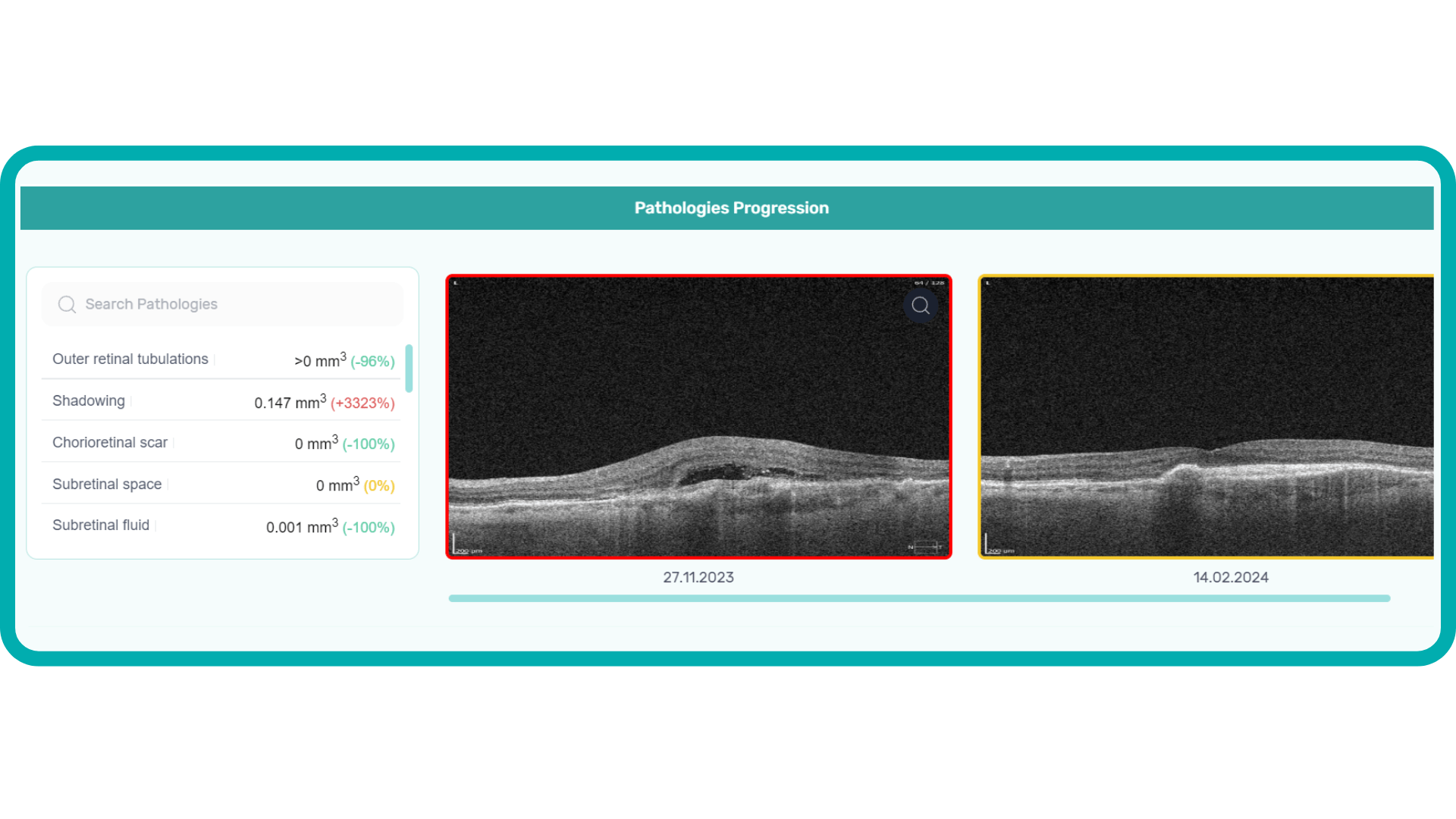
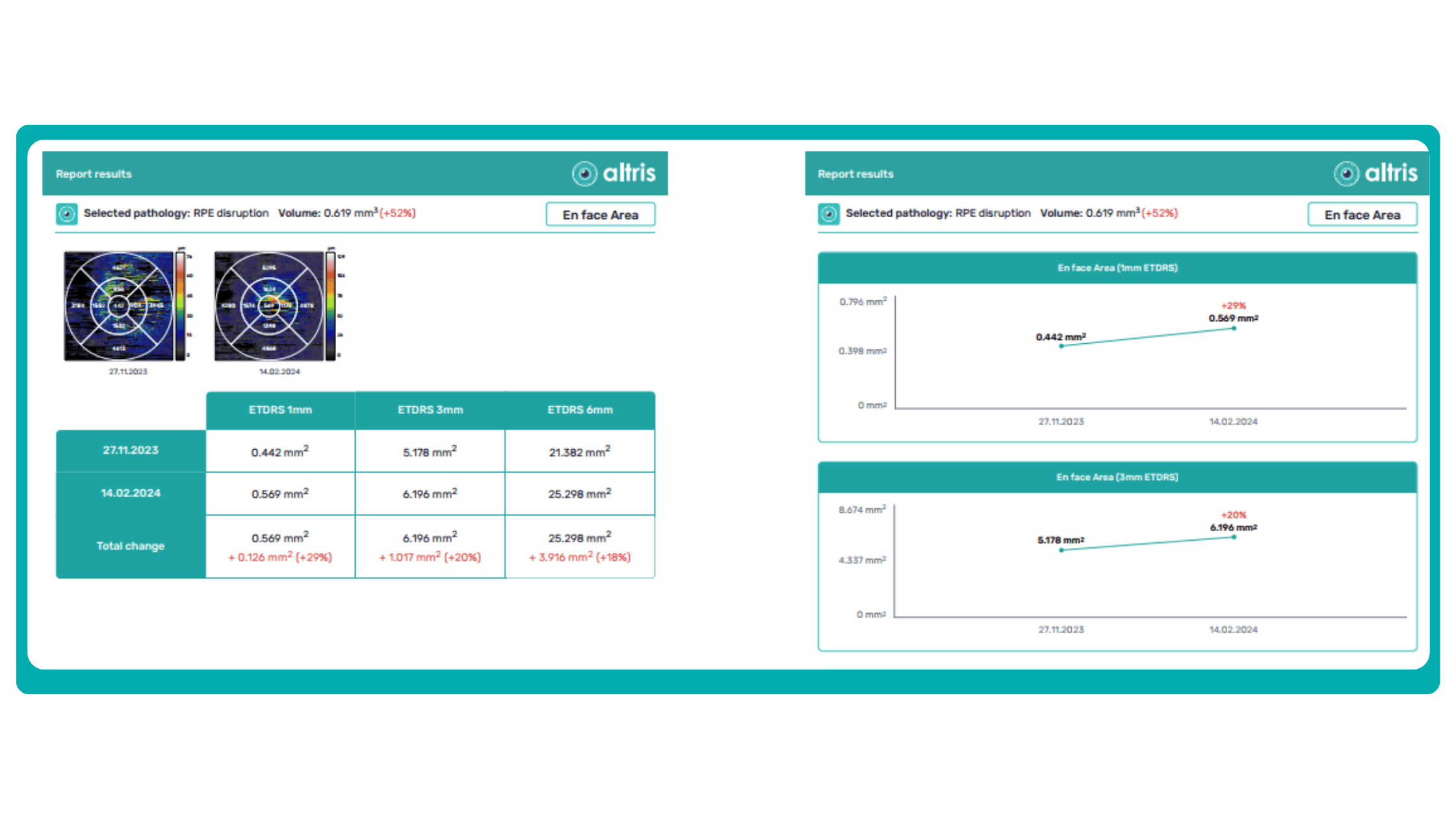
Subsequent follow-up visits will then display the most relevant picture, highlighting the most pertinent biomarkers for tracking a particular pathology (wet AMD in our example) and comparing their volume, progression, or regression through visits.

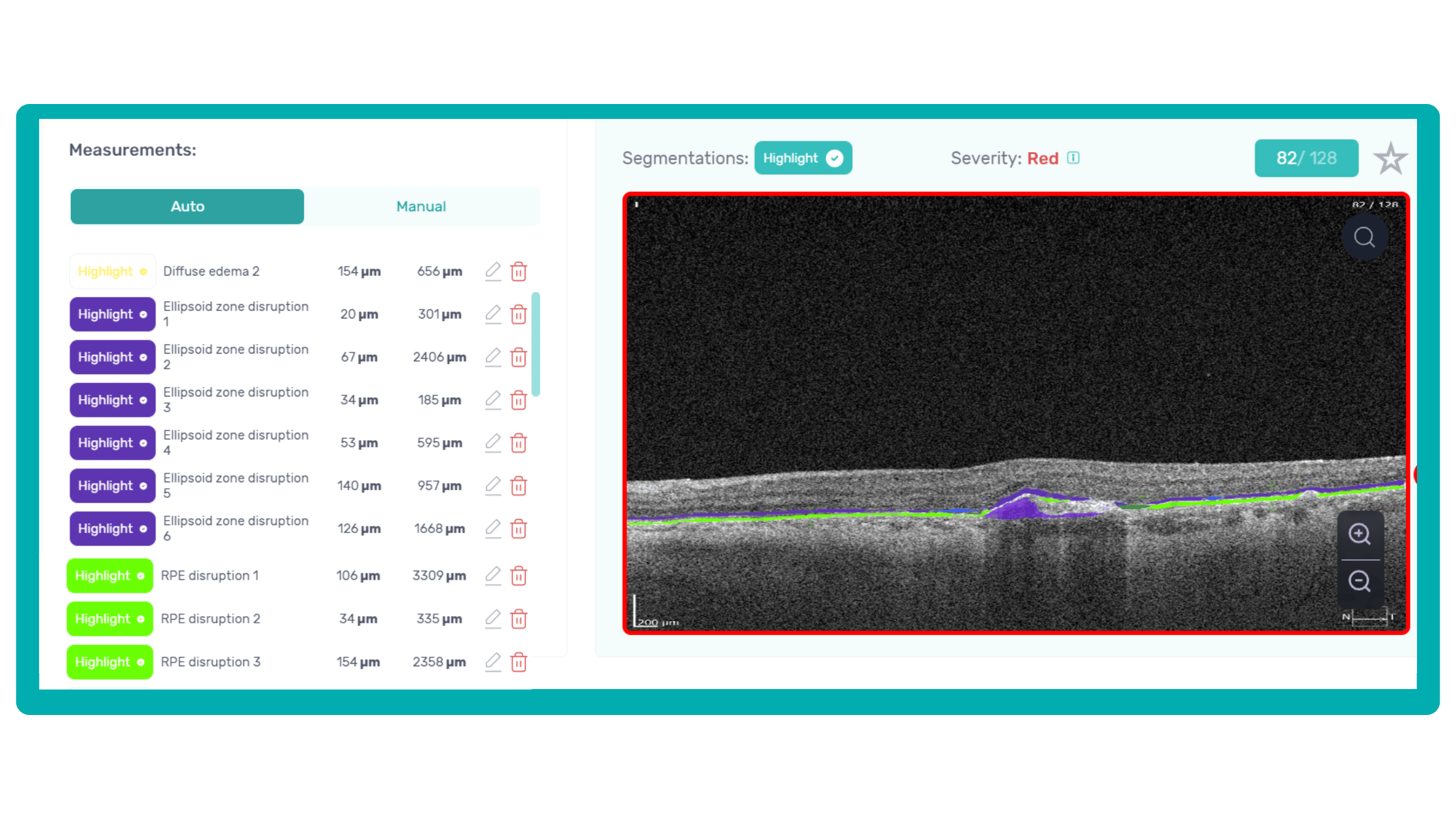
Another helpful option is retinal layer segmentation, which focuses solely on the retinal layers of interest for the specific case.
This level of customization empowers clinicians with a comprehensive yet targeted view of the patient’s condition. It saves time from manually detecting anomalies on scans and facilitates informed decision-making and personalized treatment plans.
-
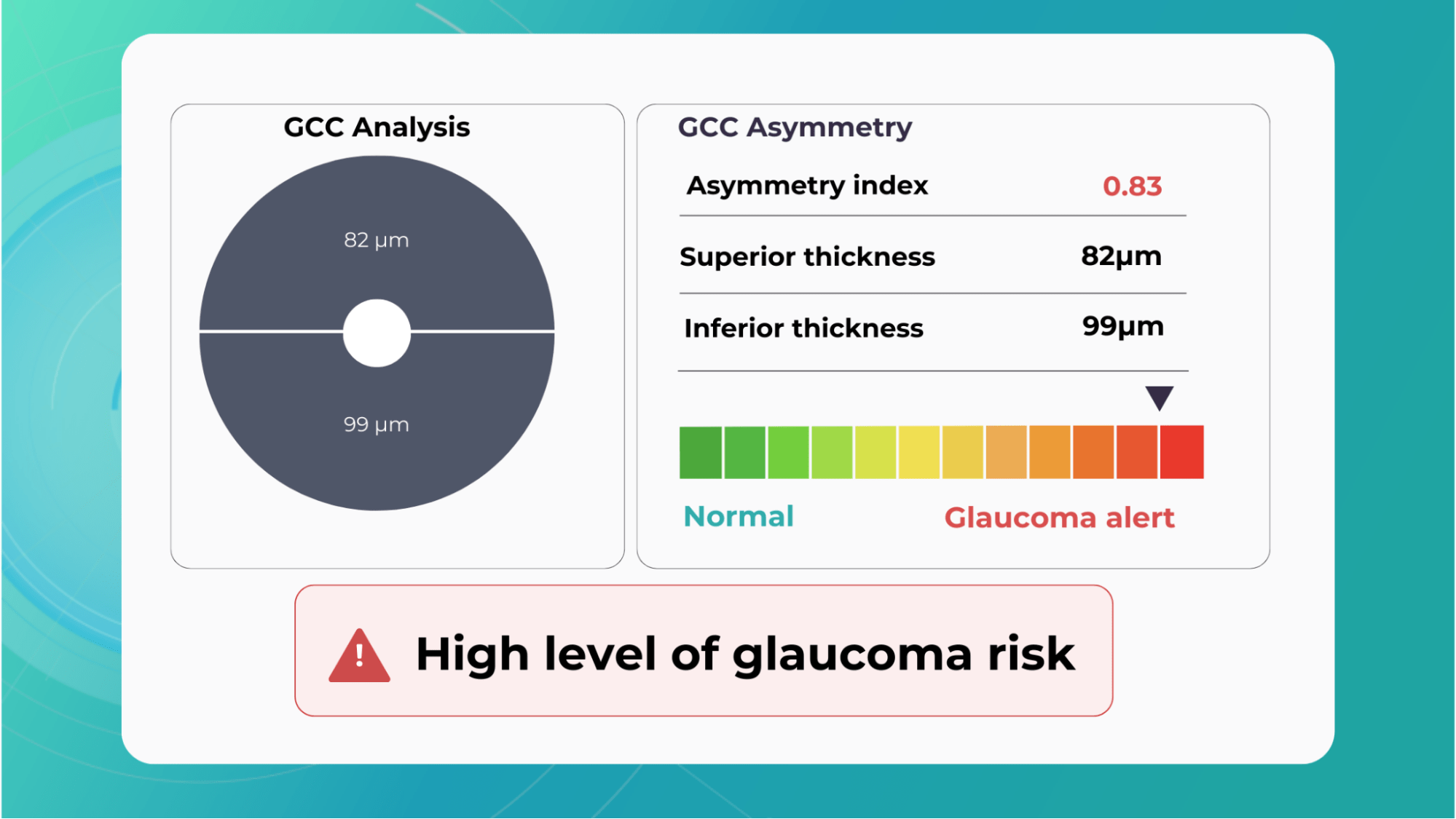
Glaucoma risk evaluation
Millions risk irreversible vision loss due to undiagnosed glaucoma, underscoring the need for improved early detection. Current tests often rely on observing changes over time, delaying treatment assessment and hindering early identification of rapid disease progression. OCT frequently detects microscopic damage to ganglion cells and thinning across these layers before changes are noticeable through other tests. However, the earliest signs on the scan can still be invisible to the human eye.
AI algorithms offer insights into glaucoma detection by routinely analyzing the ganglion cell complex, measuring its thickness, and identifying any thinning or asymmetry to determine a patient’s glaucoma risk without additional clinician effort.

Another significant benefit of AI systems is that OCT for glaucoma usually utilizes a normative database to assess retinal normality. However, these databases are limited in size and represent an average of a select group of people, potentially missing early glaucoma development in those who deviate from the “norm.” Conversely, individuals may be unnecessarily referred for treatment due to not fitting the “normal” profile, even if their eyes are healthy.
-
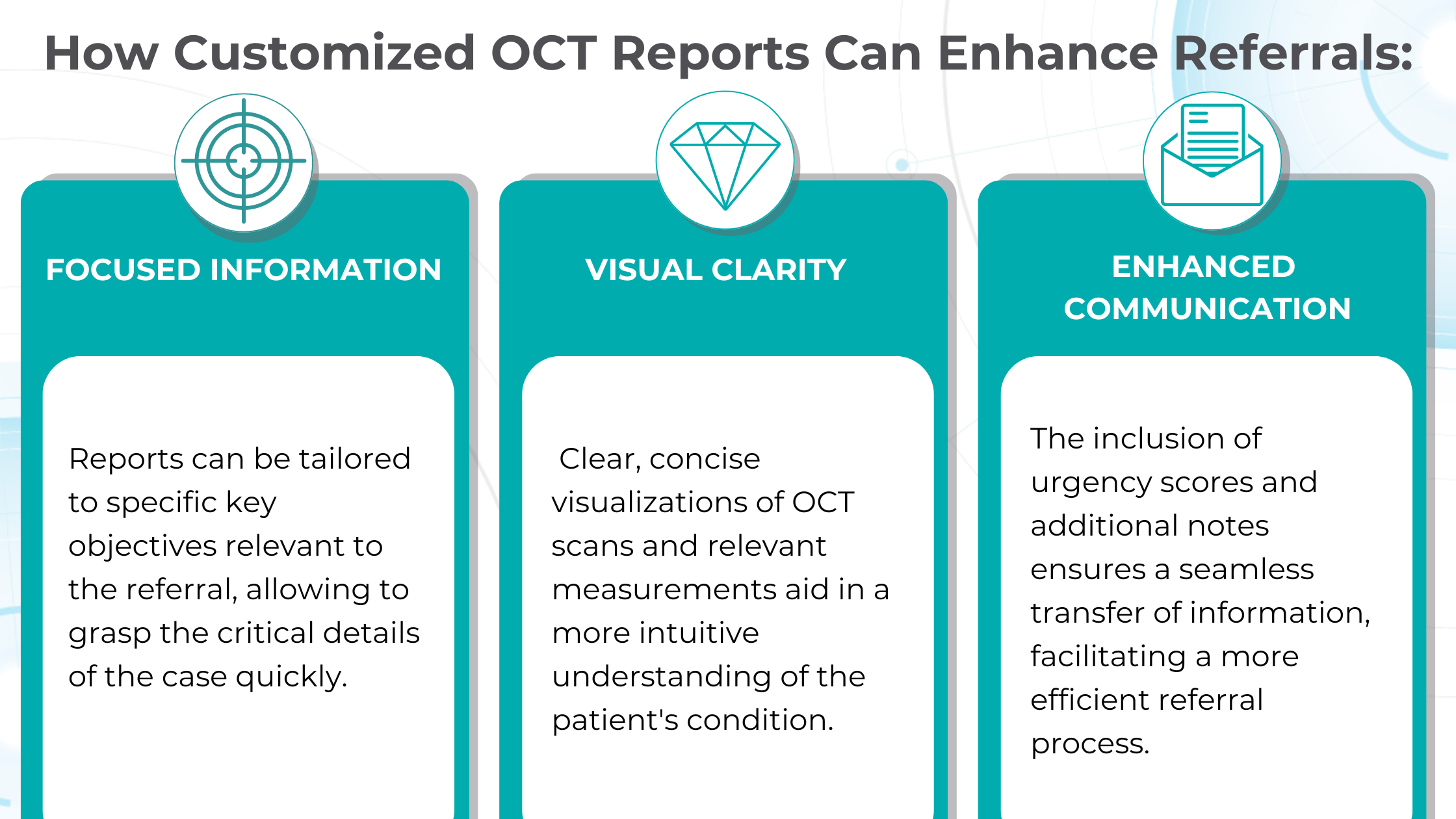
Crafting effective referral
In the UK, optometrists are crucial in initiating referrals to hospital eye services (HES), with 72% originating from primary care optometric examinations. While optometrists generally demonstrate proficiency in identifying conditions like cataracts and glaucoma, discrepancies in referral thresholds and unfamiliarity with less common pathologies can lead to unnecessary or delayed referrals.

At the same time, an evaluation of incoming letters from optometrists in a glaucoma service found that 43% of the letters were considered “failures” because they did not convey the necessity and urgency of the referral.
So, having an elaborate record of the entire clinical examination in addition to a referral letter is crucial.

Customized OCT reports solve this challenge by streamlining the referral process and improving communication between optometrists and ophthalmologists. These reports can significantly reduce delays and ensure patients receive timely care by providing comprehensive and relevant information upfront.
-
Patient Education

Patient education and involvement in decision-making are vital for every medical field and crucial for ophthalmology, where insufficient patient engagement can lead to irreversible blindness.

Research specifically targeting the ophthalmology patient population, which often includes older and potentially visually impaired individuals, reveals a clear preference for materials their eye care provider endorsed.

Providing explicit visual representations of diagnoses can significantly improve patient understanding and compliance. Seeing photos of their condition, like glaucoma progression, builds trust and reinforces the importance of treatment recommendations.
Surveying eye care professionals specializing in dry eye disease revealed a strong emphasis on visual aids during patient education.
Photodocumentation is a favored tool for demonstrating the condition to asymptomatic patients, tracking progress, and highlighting treatment’s positive outcomes.
The visual approach provides tangible evidence of the benefits of their treatment investment, allowing for a deeper understanding of the “why” behind treatment recommendations and paving the way for ongoing collaboration with the patient.

Color-coded OCT reports for pathologies and their signs, severity grading, and pathology progression over time within its OCT analysis highlight the littlest bits that a patient’s unprepared eye would miss otherwise. With follow-up visits, patients can see what’s happening within their eyes and track the progress of any conditions during treatment.

-
Updating EMR and Audit readiness
OCT reports are crucial components of a patient’s medical history and are essential for accurate diagnosis, personalized treatment, and ongoing monitoring. The streamlined process of integrating OCT data into EMR ensures that every eye scan, with its corresponding measurements, biomarkers, and visualizations, becomes an easily accessible part of the patient’s medical history.
This is crucial for continuity of care and simplifies the audit process, providing a clear and comprehensive record of the patient’s eye health over time. Just optometry chains alone can perform an imposing volume of OCT scans, with some reaching upwards of 40,000 per week. While this demonstrates the widespread adoption of this valuable diagnostic tool, it also presents a challenge: the increased risk of missing subtle or early-stage pathologies amidst the sheer volume of data.
Enhanced OCT reports offer a solution by providing a crucial “second look” at scan results. While not foolproof, this double-check significantly reduces the risk of overlooking abnormalities, ultimately improving patient outcomes and safeguarding the clinic’s reputation.
In audits, comprehensive OCT reports are critical in ensuring regulatory compliance. As the Fundamentals of Ophthalmic Coding states, “It is the responsibility of each physician to document the interpretations as promptly as possible and then communicate the findings with the patient… to develop a fail-safe way to ensure that your interpretations are completed promptly.”
Auditors typically look for several key elements in OCT reports:
- Physician’s Order: Document the test order, indicating which eye(s) and the medical necessity.
- Interpretation and Report: The physician analyzes the scan results, including any identified abnormalities or concerns.
- Timely Completion: Prompt documentation and communication of findings to the patient.
Customisable OCT reports can streamline this process by generating comprehensive reports that meet these requirements. These reports include detailed measurements, biomarker analysis, and clear visualizations, making it easier for physicians to review, interpret, and document their findings efficiently.
Summing up
Standard OCT reports, while valuable, often need more customization due to data reduction and lack of customization. The inability to visualize multiple scans simultaneously or compare data from different devices hinders comprehensive analysis. Enhanced OCT reports address these limitations by offering detailed visualizations, customizable measurements, and biomarker tracking.
Customisable OCT reports aid in the early detection and monitoring of diseases like wet AMD and glaucoma, empowering clinicians with accurate diagnoses and personalized treatment plans. Additionally, they streamline referrals by providing focused information and clear visualizations, reducing delays and improving communication between optometrists and ophthalmologists.
These comprehensive reports also enhance patient education by offering clear visual representations of their conditions and treatment progress, fostering better understanding and compliance. Moreover, with detailed documentation and analysis, detailed reports ensure audit readiness for eye care professionals, mitigating the risk of missed pathologies and upholding regulatory compliance.
-

AI for Ophthalmic Drug Development: Enhancing Biomarkers Detection
 Maria Martynova
20.05.20238 min read
Maria Martynova
20.05.20238 min readDisclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.
Despite increased research and development spending, fewer novel drugs and biologics are reaching the market today.
Large pharmaceutical companies invest an average of over $5 billion and 12+ years in research and development for each new drug approval.
The high failure rate of drug candidates (only 15% of Phase I drugs reach approval) further exacerbates the issue. This risk often leads pharmaceutical companies to favor lower-risk investments like biosimilars or generic drugs over novel therapies.
Due to the eye’s specialized anatomy and physiology, ophthalmic drug development faces unique challenges. Ocular barriers like the tear film and blood-ocular barrier can hinder drug efficacy. Many therapeutic endpoints in ophthalmology are subjective, making controlled trials difficult. The imprecise nature of some measurements further complicates trial design. Rare ophthalmic diseases pose additional challenges, as clinical trials may group diverse conditions, like multiple types of uveitic, together despite their distinct underlying mechanisms and therapeutic needs.
Here is where AI enters the game. With its ability to rapidly analyze vast amounts of data and detect subtle patterns, AI is revolutionizing how we approach clinical trials for ophthalmic drugs.
In this article, we will explore how AI for ophthalmic drug development transforms the landscape by accelerating the identification of biomarkers for conditions like diabetic retinopathy and age-related macular degeneration, ensuring the right patients are enrolled in trials, and providing quantitative metrics for evaluating treatment efficacy.
How AI for ophthalmic drug development can accelerate the search for biomarkers in clinical trials
-
Biomarkers for quantitative analysis before and after treatment
A biomarker, as defined by the BEST Resource FDA-NIH Biomarker Working Group, is a characteristic that can be objectively measured and evaluated as an indicator of normal biological processes, disease processes, or responses to therapeutic intervention. Key characteristics of a useful biomarker include specificity, sensitivity, simplicity, reliability, reproducibility, multiplexing capability, and cost-effectiveness.
Determining a biomarker’s performance involves assessing its:
- analytical validity – how accurately it measures what it claims to measure;
- clinical validity – how well it reflects a clinical feature or outcome;
- clinical utility – how it improves patient outcomes or guides treatment decisions.
In the context of drug regulation, qualified biomarkers can serve as endpoints in clinical trials, potentially offering a more objective and less placebo-susceptible alternative to traditional patient-reported outcomes.
Imaging biomarkers are a particularly attractive option for clinical use due to their non-invasive, real-time, and cost-effective nature.
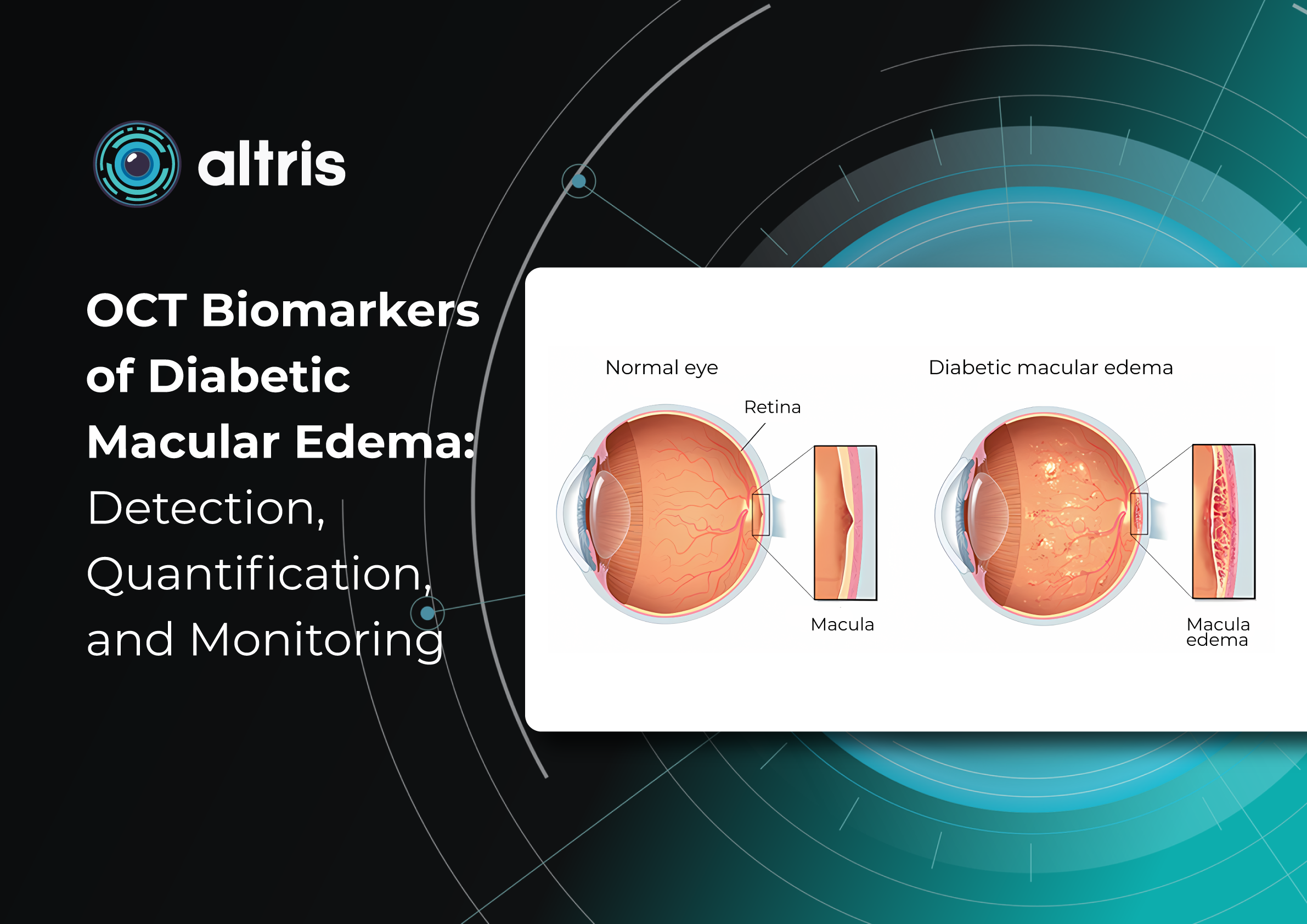
In ophthalmology, AI-powered analysis of OCT scans can provide precise, quantitative measurements of retinal thickness, fluid volume, and other biomarkers relevant to diseases like diabetic retinopathy and age-related macular degeneration. These measurements can aid in diagnosis, disease staging, treatment monitoring, and prediction of treatment response.

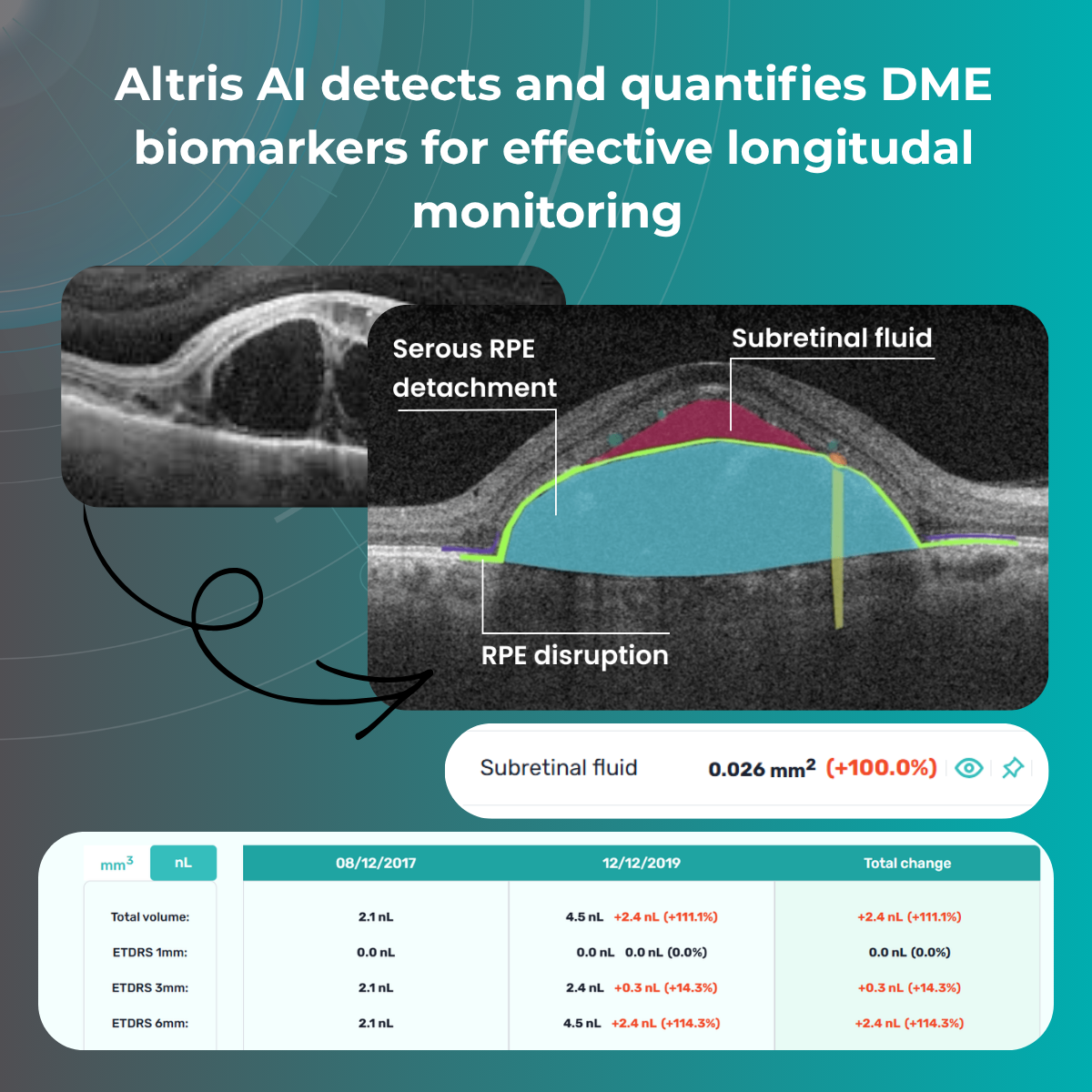
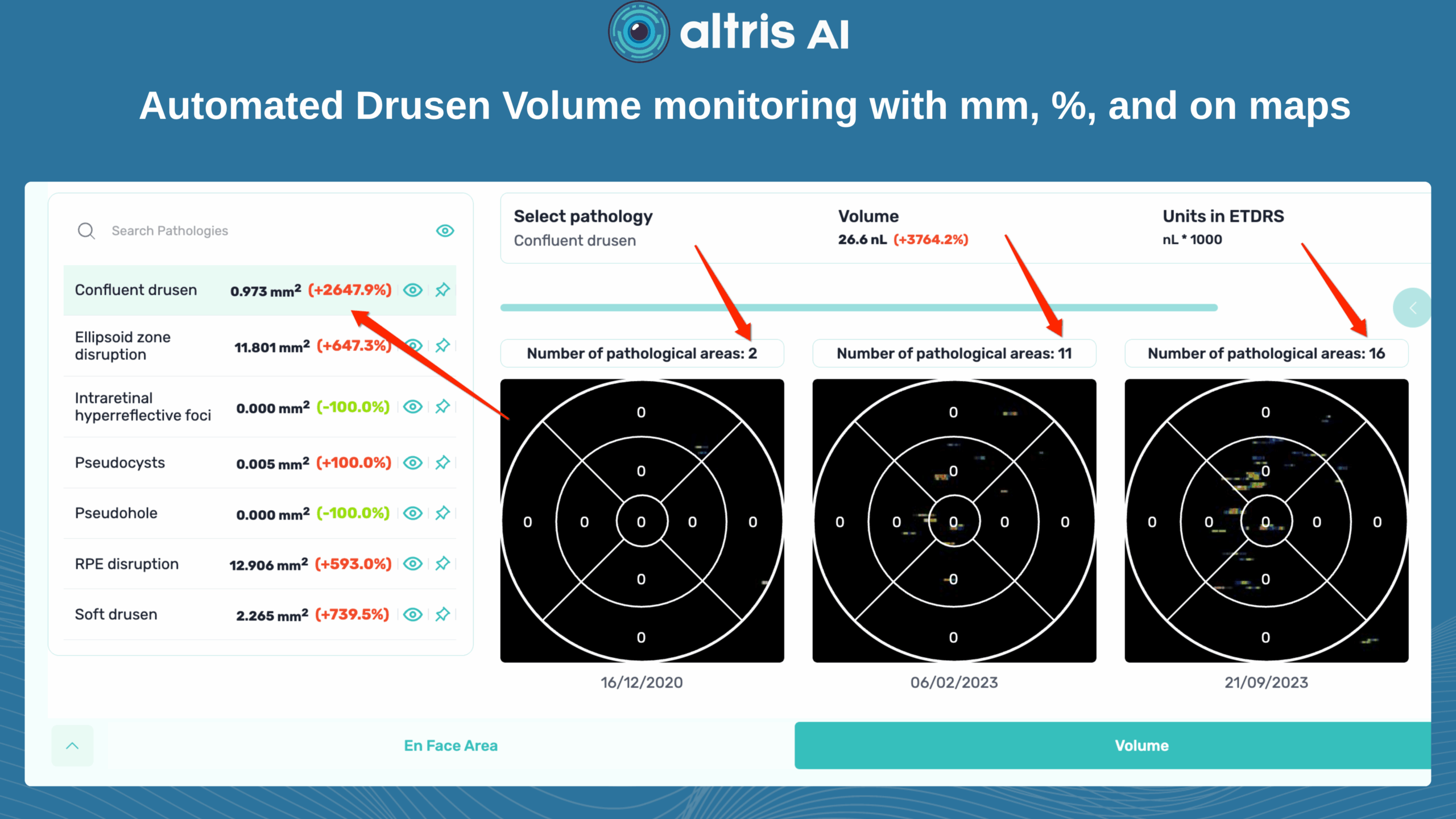
Systems like Altris for pathology detection and segmentation enabled automated disease characterization and longitudinal monitoring of therapeutic response in AMD. Multiple studies have demonstrated the value of volumetric fluid characterization, compartment-specific OCT feature evaluation, and subretinal fibrosis and hyperreflective material quantification.
A study has shown the potential of AI to predict conversion from early or intermediate non-neovascular AMD to the neovascular form, using quantitative imaging features like drusen shape and volume.
The extraction of quantitative fluid features and assessment of retinal multi-layer segmentation from OCT scans have offered valuable insights into disease prognosis and longitudinal dynamics of Diabetic Retinopathy.
A recent study demonstrated that quantitative improvement in ellipsoid zone integrity following anti-VEGF therapy for DME significantly correlated with visual function recovery. Furthermore, novel imaging biomarkers, such as the retinal fluid index (RFI), are emerging as tools for precisely monitoring treatment response. Studies have shown that early RFI volatility can predict long-term instability in visual outcomes after treatment.
Building on these advancements, researchers are now exploring the relationship between imaging biomarkers and underlying disease pathways. A recent study linked levels of various cytokines, including VEGF, MCP-1, and IL-6, with specific OCT-derived biomarkers like fluid parameters and outer retinal integrity.
By automating the analysis of OCT scans, AI not only streamlines the process but also uncovers subtle details and patterns that might be missed by human observation.
Enhanced by AI precision enables more accurate identification and quantification of biomarkers, leading to better patient stratification, treatment monitoring, and prediction of therapeutic responses.
-
Data Annotation for Clinical Trials
An ophthalmologist’s report noting the presence of edema on an OCT scan is not the same as stating that its height and length are 411 and 3213 µm, accordingly.
Imaging biomarkers can range from simple measurements of size or shape to complex computational models, providing valuable information to complement traditional diagnostic methods. They can also determine the presence and severity of a disorder, assess its progression, and evaluate treatment response.
While biomarkers can be derived from various imaging modalities, OCT stands out in ophthalmology due to its high resolution and ability to visualize subtle retinal changes.

Parametric images, which visually represent the spatial distribution of biomarker values, further enhance the analysis of OCT scans. This combination of quantitative data and visual representation empowers clinicians and researchers to make more informed decisions about diagnosis, treatment, and disease management.
Traditionally, medical image interpretation has relied heavily on visual assessment by experts, who recognize patterns and deviations from normal anatomy based on their accumulated knowledge.
While semi-quantitative scoring systems offer some level of objectivity, the field is rapidly evolving towards more quantitative and automated approaches. This shift is driven by advancements in standardization, sophisticated image analysis techniques, and the rise of machine and deep learning.
In some clinical scenarios, automated image quantification can surpass manual assessment in objectivity and accuracy, interpreting subsequent changes with greater precision and clinical relevance by establishing thresholds for disease states. Unlike physical biomaterials, medical images are easily and rapidly shared for analysis, facilitating automated, reproducible, and blinded biomarker extraction.
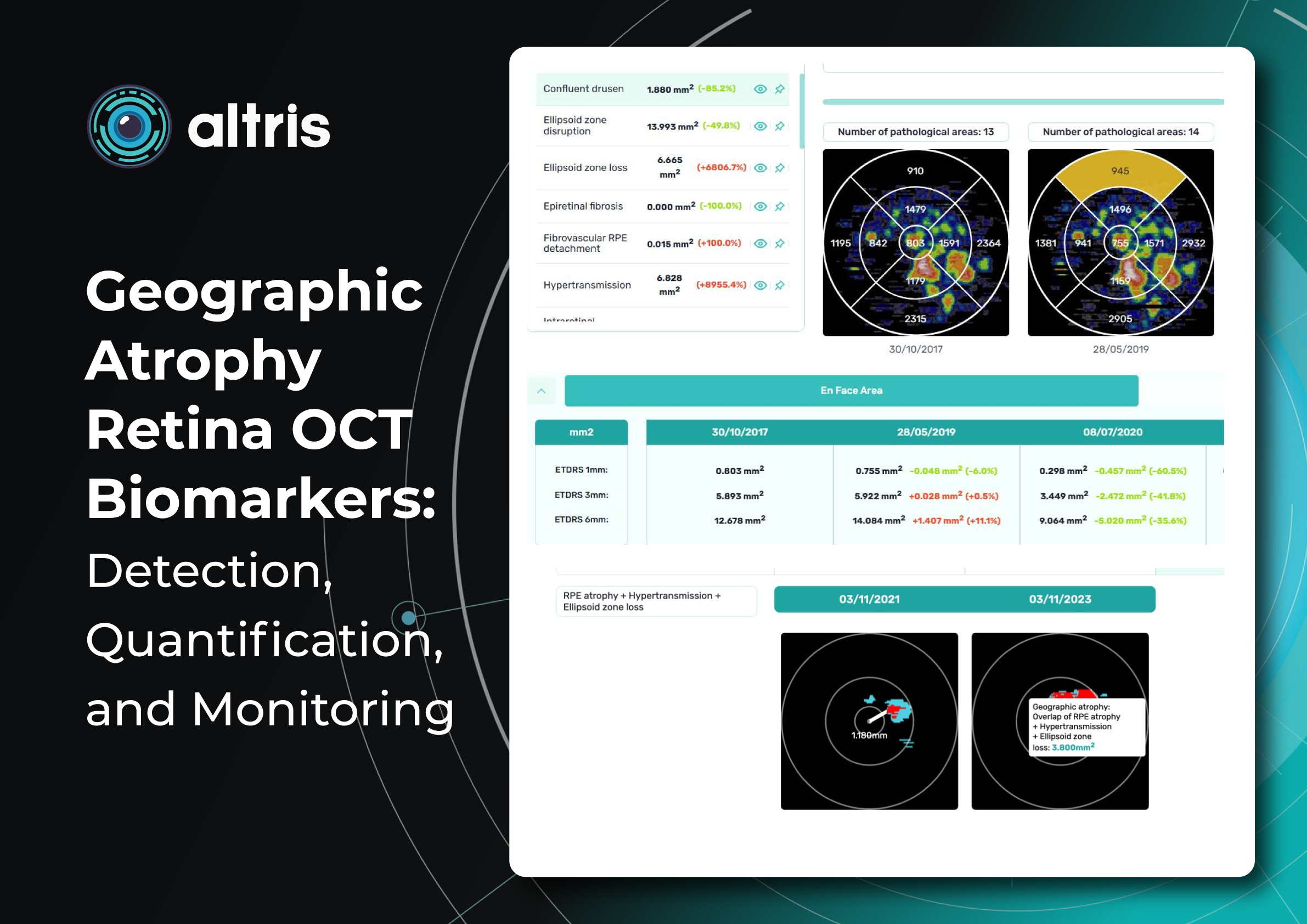
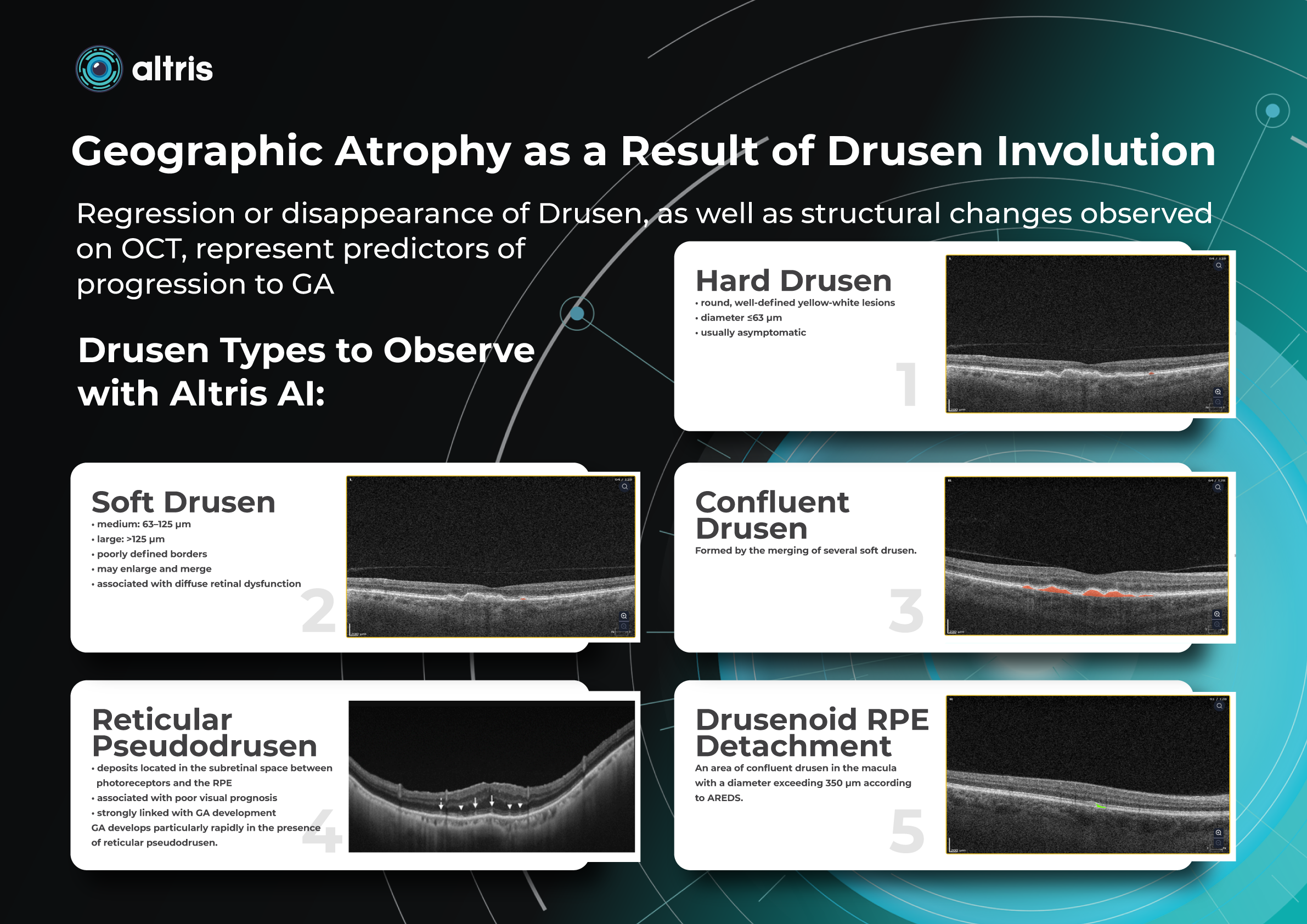
This transition to quantitative analysis is particularly evident in the study of AMD. For instance, non-neovascular (dry) AMD has been extensively evaluated using various imaging biomarkers, such as intraretinal hyper-reflective foci, complex drusenoid lesions, subretinal drusenoid deposits, and drusen burden.
While SD-OCT has traditionally described these features qualitatively, recent studies have demonstrated the predictive power of quantitative measures like ellipsoid zone integrity, sub-RPE compartment thickness, and automated drusen volume quantification.
These quantitative biomarkers have shown stronger associations with disease progression than qualitative features, particularly in predicting the development of geographic atrophy.
This predictive power of AI extends to diabetic retinopathy as well. In DR, quantitative measures like central subfield retinal thickness and retinal nerve fiber layer thickness have been linked to disease severity. Disruption of retinal inner layers has been associated with worse visual acuity, and its presence is highly specific for macular nonperfusion. Both DRIL and outer retinal disruption are linked to visual acuity in DR and diabetic macular edema.
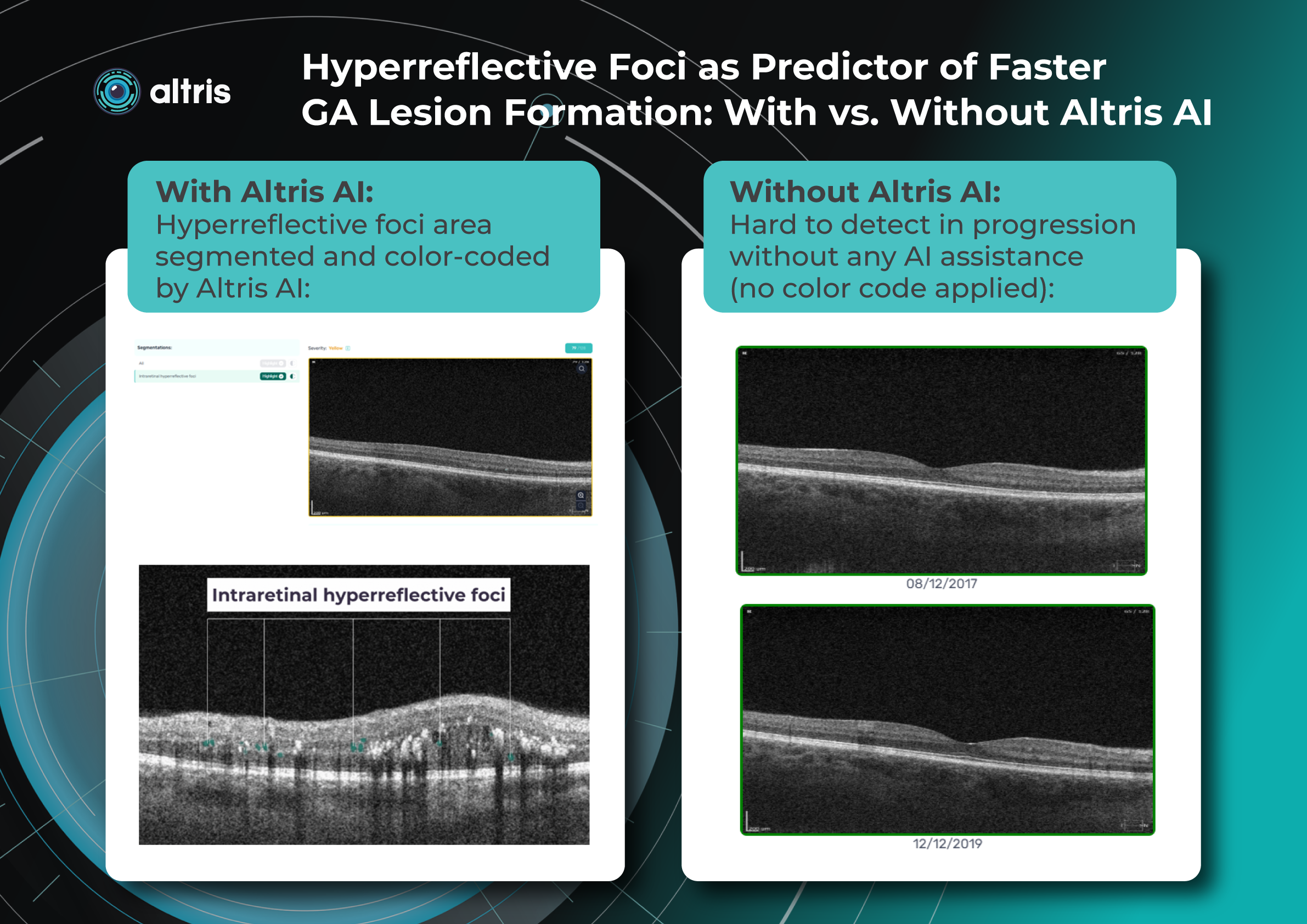
Furthermore, morphological signs like hyperreflective foci, representing lipid extravasation and inflammatory cell aggregates, have emerged as potential biomarkers for monitoring inflammatory activity in diabetic eye disease. AI-powered segmentation and quantification of HRF can track changes in response to anti-VEGF and steroid injections.
-
Enrollment of the right patients
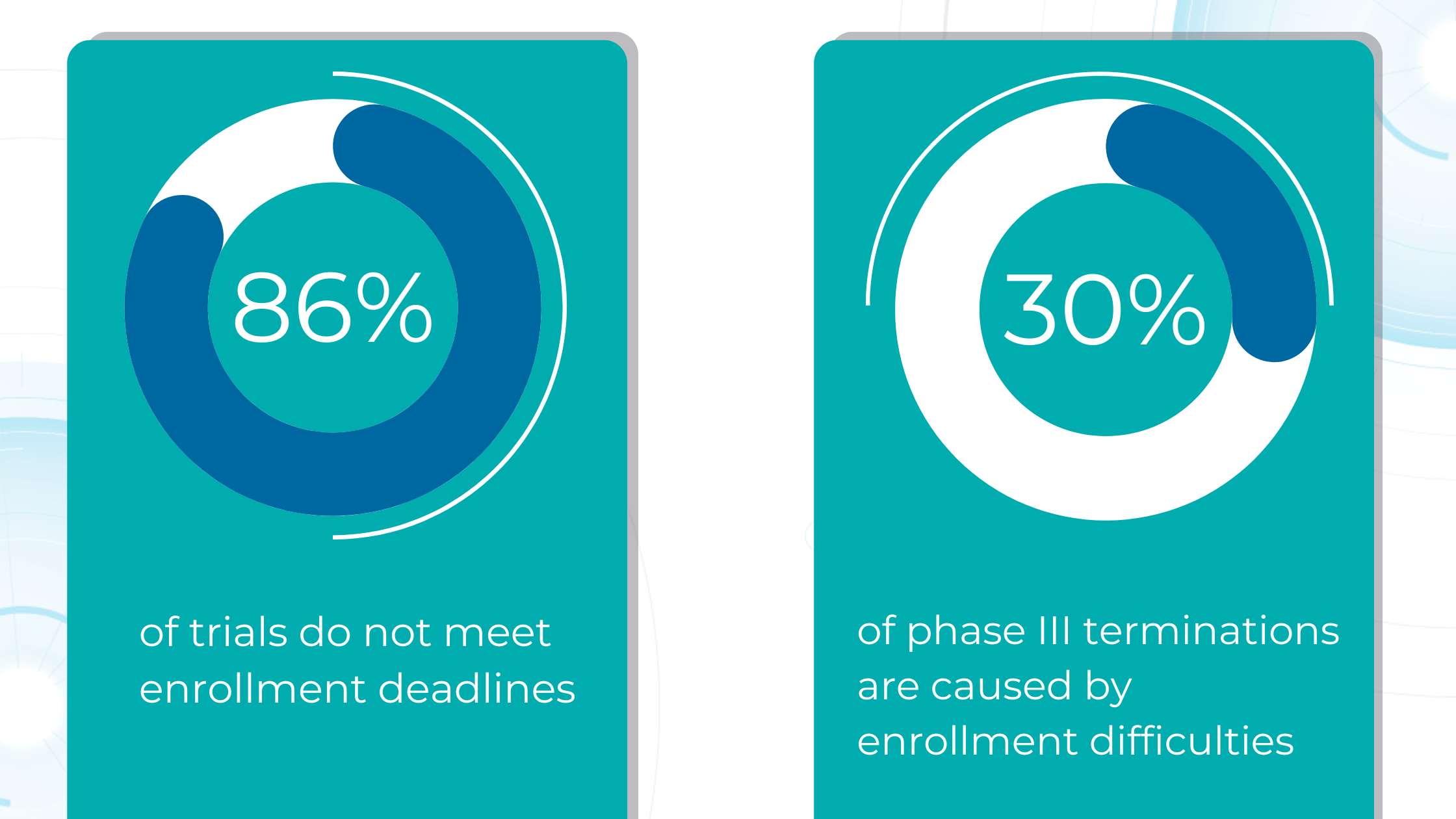
Due to their complexity and scale, clinical trials, particularly Phase III trials, consume a significant portion of the budget required to bring a new drug to the market. However, the success rate for compounds entering clinical trials is dismal, with only about one in ten progressing to FDA approval. This high failure rate stems largely from ineffective patient recruitment, as each clinical trial has unique participant requirements, including eligibility criteria, disease stage, and specific sub-phenotypes.
Manual review of electronic medical records is time-consuming and prone to error, as staff must sift through vast amounts of data to identify eligible candidates.

AI can automate this process, rapidly analyzing medical imaging and extracting relevant information to determine patient eligibility. This reduces the burden on staff and allows for faster identification and enrollment of suitable participants, streamlining patient selection and ultimately leading to more efficient and successful clinical trials.
A targeted approach can dramatically improve recruitment efficiency by pinpointing ideal candidates and even revealing disease hotspots for geographically focused efforts.
In later phases of clinical trials (Phase II and III), AI-powered image analysis can also play a pivotal role. In ophthalmology, AI can analyze OCT scans to precisely quantify disease biomarkers, ensuring that the trial participants are those most likely to benefit from the investigated drug. This improves the success rate of trials and minimizes potential harm to patients who might not be suitable candidates.
AI-powered image analysis offers a crucial advantage: reducing variability in interpretation.
AI algorithms can standardize the imaging overview process by consistently identifying and quantifying key biomarkers, ensuring that different readers arrive at similar conclusions.
-
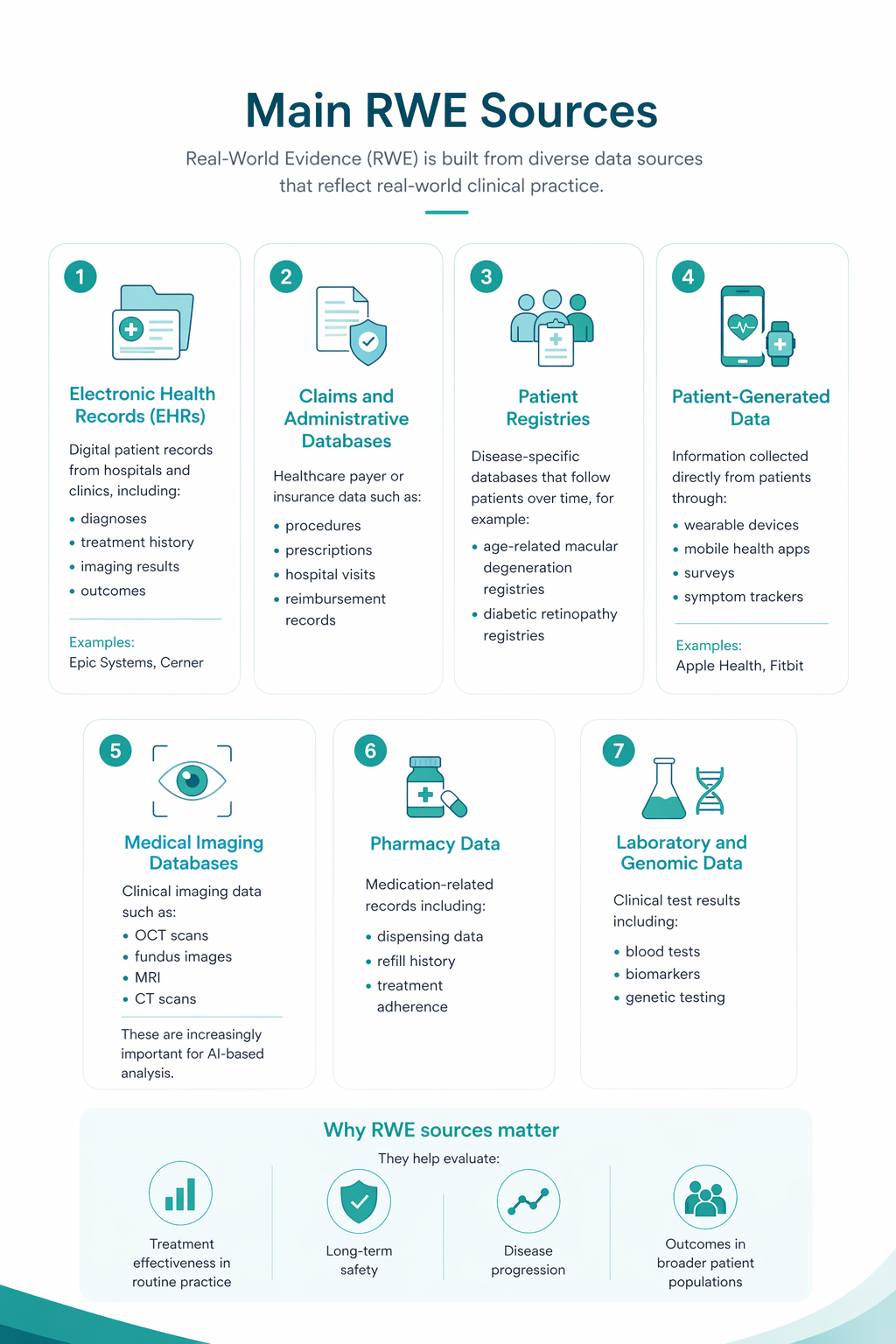
Real World Evidence
Randomized controlled trials have long been the gold standard for evaluating the efficacy and safety of new therapies. However, controlled environments with strict inclusion and exclusion criteria may not fully reflect the diversity and complexity of real-world patient populations.
Real-world data (RWD) that is collected during routine clinical practice can provide critical insights into disease biomarkers and significantly impact the drug development process. This RWD can be transformed into real-world evidence (RWE) when appropriately analyzed.
RWE is bridging the gap between clinical trials and real-world patient care, providing a more representative view of disease progression, treatment patterns, and long-term outcomes in everyday clinical settings.
In ophthalmology, RWE already has played a crucial role in understanding the impact of anti-VEGF therapies for neovascular age-related macular degeneration. While RCTs demonstrated the initial efficacy of these treatments, RWE studies have shown variations in real-world outcomes and highlighted the need for continued and higher than previously provided treatment frequency and new treatment regimens such as treat-and-extend.
Big data, encompassing a vast array of structured and unstructured information, is now an integral part of modern medicine, including ophthalmology. By integrating RWE with traditional clinical trial data, researchers can better understand how a drug performs in the real world and conduct more pragmatic clinical trials designed to evaluate treatments in real-world settings with broader patient populations, ultimately accelerating the development of safer and more effective therapies.
The future of ophthalmic drug trials

The global AI-in-drug discovery market is poised for significant growth, driven by advancements in machine learning, natural language processing, and deep learning.
Artificial intelligence has the potential to significantly impact drug discovery by enabling more creative and efficient experimentation. It can also reduce the cost and time associated with failures throughout the drug development process. By identifying promising leads earlier and eliminating less viable options, AI can streamline each stage, potentially halving the total cost of a single project.

Advanced simulation and modeling techniques powered by AI are also poised to revolutionize our understanding of disease mechanisms and accelerate the discovery of new drugs.
The promising potential of AI in clinical trials extends to the proactive identification and mitigation of adverse events, enhancing patient safety and reducing trial risks. Data-driven AI tools are poised to revolutionize the entire clinical trial process, from design to execution. By streamlining patient recruitment, continuously monitoring participants, and facilitating comprehensive data analysis, AI can increase trial success rates, improve adherence, and yield more reliable endpoints.
The future of ophthalmic drug trials is here, and it’s powered by AI. By embracing this technology, researchers and clinicians can unlock new possibilities for preventing blindness and preserving vision for future generations.
-
-

Educating Patients about Eye Health
 Maria Znamenska
26.04.20239 min read
Maria Znamenska
26.04.20239 min readEducating Patients about Eye Health with AI
Today patients are curious about AI, but they may also have some reservations. Researches suggest a cautious attitude towards autonomous AI in healthcare, but what happens when AI becomes a collaborative tool, assisting eye care professionals in educating and treating patients? This shift in focus can significantly affect patients’ comfort levels and acceptance of AI.
Patients have some concerns about AI in healthcare. Let’s delve into the patient perspective and discover how addressing these apprehensions and implementing AI-assisted OCT in eye care can lead to a better understanding of the technology and, ultimately, healthier outcomes.
Educating Patients about Eye Health
Interestingly, while surveys extensively document how eye care professionals feel about and interact with AI, the perspectives of the main beneficiary—the patient—remain less understood. The limited research available indicates mixed feelings towards this technology. Few studies examine patient attitudes toward AI in healthcare and eye care, suggesting a degree of caution.

However, these studies have focused on scenarios where AI fully replaces human healthcare providers. Patients demonstrated significant resistance to medical AI in these cases driven mostly by “uniqueness neglect” – concern that AI providers are less able than humans to account for a person’s unique characteristics and circumstances.
For example, in the “Resistance to Medical Artificial Intelligence” study, participants demonstrated less interest in using a stress assessment and were willing to pay less for it when administered by an automated system rather than a human, even with equivalent accuracy. Additionally, participants showed a weaker preference for a provider offering clearly superior performance if it was an AI system.
A survey of 926 patients reveals a mix of attitudes towards AI in healthcare but also gives us clues to understand the reasons behind it. While a majority believe AI could improve care, there’s also a significant undercurrent of caution:
- Desire for Transparency: Over 95% of respondents felt it was either very or somewhat important to know if AI played a significant role in their diagnosis or treatment.
- Unexplainable AI = Uncomfortable: Over 70% expressed discomfort with receiving an accurate diagnosis from an AI system that couldn’t explain its reasoning. This discomfort was more pronounced among those unsure about AI’s overall impact on healthcare.
- Application Matters: Patients were more comfortable with AI for analyzing chest X-rays than for making cancer diagnoses.
- Minority Concerns: Respondents from racial and ethnic minority groups expressed higher levels of concern about potential AI downsides, such as misdiagnosis, privacy breaches, reduced clinician interaction, and increased costs.
These findings highlight the importance of being transparent with patients about how AI is used in their care. Explaining the role of AI and reassuring patients that it’s a tool for assisting your clinical judgment (not replacing it) will be essential. Additionally, being mindful of potential heightened concerns among minority patients is crucial for providing equitable care.
A study solely focused on overcoming patients’ resistance to AI in healthcare found that demonstrating social proof (like highlighting satisfied customer reviews) increased trust in AI-involved help.
The team has identified several additional strategies for reducing patient apprehension of AI recommendations. One effective approach is to emphasize AI’s collaborative nature, where a human doctor endorses recommendations. This highlights AI as a tool to assist, not replace, physicians. Demonstrating AI capabilities through real-world examples where AI exhibits nuanced reasoning can also encourage greater reliance on the technology.
How to inform patients with AI in eye care
AI offers a powerful way to transform your practice and set yourself apart. It brings world-class diagnostic expertise directly to your community, potentially saving patients’ sight by catching eye diseases in their earliest stages. Here’s how to position AI for patients:
- Emphasize Early Detection
It brings world-class diagnostic expertise directly to your community, potentially saving patients’ sight by catching eye diseases in their earliest stages, including early signs of glaucoma, AMD, and many other pathologies that would often be invisible during a regular visit. Some retinal changes are so microscopic that they elude the human eye, making the program’s ability to detect tiny retinal changes invaluable. This makes AI a powerful tool during routine exams, potentially uncovering issues you may not even have been aware of as a patient.
- More time for personalized care with optometry patient education
Patients expect personalized experiences, and AI empowers you to deliver exactly that. By analyzing each patient’s unique OCT image data, AI helps identify potential pathologies with greater accuracy.

Additionally, since AI acts as a meticulous assistant, double-checking your assessments and minimizing the risk of missed diagnoses, it frees up your time. This allows for more meaningful one-on-one conversations with patients, where you can explain their results and discuss the next steps, setting your practice apart regarding patient satisfaction.
- Your old good eye care professional, but with superpower
With AI-assisted OCT, you have the combined knowledge and experience of leading eye care specialists at your fingertips for every patient. This technology leverages massive datasets of medical images and clinical data meticulously analyzed by retinal experts during AI development. It is a valuable second opinion tool, helping you confirm diagnoses and identify subtle patterns the human eye might miss.

This offers your patients peace of mind – knowing their diagnosis has been informed by insights from a team of experts incorporated into the AI’s analysis.
It’s crucial to emphasize that AI will never replace the human touch. It’s a powerful tool that frees up your time for what matters most: building trust through personalized care and addressing patient concerns with empathy.
How to explain what AI is to patients

Patient understanding is vital for building trust with you and any technology you use. It is especially important when talking about a sophisticated instrument like AI. In case of AI, which remains a mystery to many, patient education in optometry is a must.
For instance, we’ve found that patients sometimes struggle to understand how Altris AI, our AI-powered OCT analysis tool, works. We’ve crafted an explanation that helps them grasp the concept more quickly, covering how retinal specialists have taught the system to do its job, the AI’s role as a doctor’s help, and direct benefits for patients.
OCT scans provide incredibly detailed images of the retina, the important layer at the back of your eye. Eye doctors carefully analyze these scans to spot any potential problems. To make this process even more thorough, AI systems are now being used to assist with OCT analysis.

How does the system know how to do that? Real doctors have taught it. It works by first learning from thousands of OCT scans graphically labeled by experienced eye doctors.
The doctors analyzed images from real patients to detect and accurately measure over 70 pathologies and signs of pathology, including age-related macular degeneration and glaucoma, teaching the AI what to look for.
The system leverages a massive dataset of thousands of OCT scans collected from 11 ophthalmic clinics over the years. Carefully segmented and labeled by retinal professionals, these scans were used to train the AI. By analyzing each pixel of an image and its position relative to others, the AI has learned to distinguish between different biomarkers and pathologies.
The platform visualizes what is going on with the retina using color coding. This means that every problem on the OCT scan will be colored differently and signed so you will be able to understand what is going on with your retina.

As with any innovative tool, Altris AI partially automates some routine tasks, so clinicians have more time for what is important: talking to patients, learning more about their eye health, and providing treatment advice.
Why does this matter to you? Altris AI can help spot even the tiniest changes in your eyes, leading to earlier treatment and better protection of your eye health. Knowing a smart computer system is also double-checking your scans gives both you and your doctor extra confidence in the results.
With the help of Altris AI, you will be able to see how the treatment affects you. For example, if you have fluid in the retina (that is not supposed to be there), you will be able to see if its volume is decreasing or increasing with the help of color coding.

Altris AI was designed by eye doctors for eye doctors. It’s a tool to help us take even better care of patients.
AI color coding in eye care: how learning about diagnosis influences treatment adherence
Patient-centered care, a key principle outlined by the Institute of Medicine, emphasizes optometry patient education and involvement in decision-making. This is vital in ophthalmology, where insufficient patient engagement can lead to irreversible blindness.
Research specifically targeting the ophthalmology patient population, which often includes older and potentially visually impaired individuals, reveals a clear preference for individualized education sessions and materials endorsed by their eye care provider.
According to Wolters Kluwer Health, patients crave educational materials from their providers, yet only two-thirds actually get them. This leaves patients searching for information, potentially exposing them to unreliable sources.
Providing clear, accessible patient education is crucial to ensure understanding and treatment adherence.
The human brain’s ability to process visual information far surpasses its speed with text, making visual aids a powerful tool for health education. In the field of eye care, this becomes even more critical. Patients often experience vision difficulties, potentially hindering their ability to absorb written materials. Providing clear visual representations of diagnoses can significantly improve patient understanding and compliance.
A study shows a strong preference for personalized educational materials, especially among older visually impaired patients. Seeing photos of their condition, like glaucoma progression, builds trust and reinforces the importance of treatment recommendations.
Surveying eye care professionals specializing in dry eye disease revealed a strong emphasis on visual aids during patient education. Photodocumentation is a favored tool for demonstrating the condition to asymptomatic patients, tracking progress, and highlighting the positive outcomes of treatment.
A visual approach is particularly motivating for patients. It provides tangible evidence of the benefits of their treatment investment, allowing for a deeper understanding of the “why” behind treatment recommendations and paving the way for ongoing collaboration with the patient.
Understanding complex eye conditions can be challenging for patients. Altris AI aims to bridge this gap by using color coding for pathologies and their signs, severity grading, and pathology progression over time within its OCT analysis.
With Altris AI, scans are color-coded for instant interpretation: all the detected pathologies are painted in different colors, highlighting the littlest bits that the unprepared eye of a patient would miss otherwise.

This easy-to-understand visual system empowers patients. They can clearly see what’s happening within their eyes and track the progress of any conditions during treatment.
Eye care professionals are enthusiastic about its impact.

The power of visuals goes beyond understanding a diagnosis. When patients see the interconnected structures that make up their vision, they gain a deeper appreciation for its complexity and the importance of preventative care. This understanding fosters a true partnership between doctor and patient, where the patient is an active, informed participant in their own eye health.
Summing up: Educating Patients about Eye Health
Patient education in optometry is vital today and AI is the perfect tool for that. Patients are increasingly curious and open to AI’s potential in general healthcare and eye care in particular, but naturally, some questions and hesitation remain. They stem from a desire to ensure AI considers their individual needs. By addressing these concerns proactively and clarifying when and how AI is used in their care, emphasize the collaborative doctor-AI model—highlight that YOU review and endorse all AI recommendations.
You can successfully integrate this powerful technology into your practice by addressing patient concerns with empathy and highlighting AI’s benefits. This leads to better patient education in optometry and empowered patient experience, improving understanding, adherence to treatment, and, ultimately, better health outcomes.
Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.
-

Early Glaucoma Detection Challenges and Solutions
 Maria Martynova
09.04.202310 min read
Maria Martynova
09.04.202310 min readGlaucoma’s silent progression highlights a challenge we all face as clinicians. Millions of individuals remain at risk for irreversible vision loss due to undiagnosed disease – 50% or more of all cases. This emphasizes our responsibility to enhance early detection strategies for this sight-threatening condition.
Existing clinical, structural, and functional tests depend on both baseline exams and the need to observe changes over time, delaying the assessment of treatment effectiveness and the identification of rapid progression.
In this article, we will consolidate our knowledge as eye care professionals about Glaucoma, explore current clinical detection practices, and discuss potential areas to optimize early Glaucoma detection.
What we know about Glaucoma
Glaucoma is a complex neurodegeneration fundamentally linked to changes occurring in two locations: the anterior eye (elevated pressure) and the posterior eye (optic neuropathy). Factors influencing glaucoma development include:
- age,
- ethnicity,
- family history,
- corneal thickness,
- blood pressure,
- cerebrospinal fluid pressure,
- intraocular pressure (IOP),
- and vascular dysregulation.
Early stages of Glaucoma are often asymptomatic, highlighting the importance of comprehensive eye exams, even without apparent vision issues. Current diagnostic criteria are insufficient and lack markers of early disease.
Glaucoma is broadly divided into primary and secondary types, with primary open-angle Glaucoma (POAG) representing approximately three-quarters (74%) of all glaucoma cases.
Primary glaucomas develop independently of other eye conditions, while secondary glaucomas arise as a complication of various eye diseases, injuries, or medications.
POAG is characterized by an open iridocorneal angle, IOP usually > 21 mmHg, and optic neuropathy. Risk factors include age (over 50), African ancestry, and elevated IOP. While IOP is a significant factor, it’s unpredictable – some patients with high IOP don’t develop Glaucoma, and some glaucoma progresses even at normal IOP.
Normal-tension Glaucoma (NTG) shares POAG’s optic nerve degeneration but with consistently normal IOP levels (<21mmHg). Vascular dysregulation and low blood pressure are risk factors. While rarer than POAG, IOP lowering can still be beneficial.
Primary Angle-Closure Glaucoma (PACG) is caused by narrowing the iridocorneal angle, blocking aqueous humor flow. More common in East Asian populations, it can be acute (severe symptoms, IOP often > 30mmHg) or chronic.
Secondary glaucomas are caused by underlying conditions that elevate IOP. Examples include pseudoexfoliative, neovascular, pigmentary, and steroid-induced Glaucoma.
Age is a central risk factor for glaucoma progression, linked to cellular senescence, oxidative stress, and reduced resilience in retinal ganglion cells and the trabecular meshwork. Intraocular pressure (IOP) remains the most significant modifiable risk factor. Understanding individual susceptibility to IOP-related damage is crucial. Existing IOP-lowering treatments have limitations in both efficacy and side effects.

Glaucoma has a strong genetic component, with complex interactions between genes, signaling pathways, and environmental stressors. For now, we know that mutations in each of three genes, myocilin (MYOC), optineurin (OPTN), and TANK binding kinase 1 (TBK1), may cause primary open-angle Glaucoma (POAG), which is inherited as a Mendelian trait and is responsible for ~5% of cases (Mendelian genes in primary open-angle Glaucoma).
More extensive effect mutations are rare, and more minor variants are common. Genome-wide association studies (GWAS) reveal additional genes potentially involved in pressure sensitivity, mechanotransduction, and metabolic signaling.
Recent research also suggests a window of potential reversibility even at late stages of apoptosis (a programmed cell death pathway, which is likely the final step in RGC loss). Cells may recover if the harmful stimulus is removed. This offers hope that dysfunctional but not yet dead RGCs could be rescued.
The Challenges of Early Glaucoma Detection
One of the most insidious aspects of Glaucoma is its largely asymptomatic nature, especially in the early stages. This highlights the limitations of relying on symptoms alone and underscores the importance of proactive detection strategies.
Relying on intraocular pressure (IOP) as a stand-alone glaucoma biomarker leads to missed diagnoses, especially in patients with normal-tension Glaucoma. Structural changes, such as optic disc cupping, also lack the desired sensitivity and specificity for early detection.
Optic nerve head evaluations remain subjective, with studies indicating that even experienced ophthalmologists can underestimate or overestimate glaucoma likelihood.
According to the research, even experienced clinicians can have difficulty evaluating the optic disc for Glaucoma. Both trainees and comprehensive ophthalmologists have been found to underestimate glaucoma likelihood in approximately 20% of disc photos. They may also misjudge risk due to factors like variations in cup-to-disc ratio, subtle RNFL atrophy, or disc hemorrhages.
Current Glaucoma Diagnosis in Clinical Practice
Eye care professionals typically encounter new glaucoma diagnoses in one of two ways:
- Firstly, during routine preventive examinations. A patient may come in for various reasons, including work requirements, and be found to have elevated intraocular pressure. This finding prompts further evaluation, potentially leading to a glaucoma diagnosis.
- Secondly, it is a finding in older patients (often over 50-60). A patient may present with significant vision loss in one eye, and examination reveals Glaucoma. Unfortunately, vision loss at this stage is often irreversible.
Alternatively, a patient may seek care for an unrelated eye problem. During the comprehensive examination, the eye care professional may discover changes suggestive of Glaucoma.

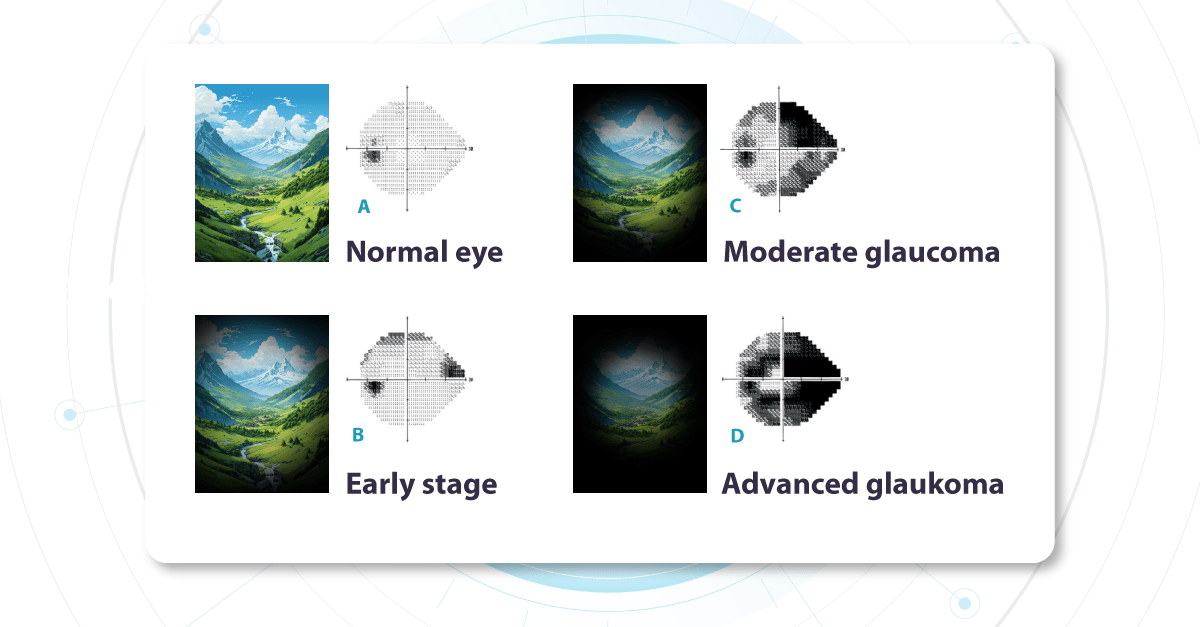
As it is statistically prevalent, we most often work with primary Glaucoma, where no other underlying eye diseases are present. Functional changes, specifically as seen on visual field testing, help diagnose and stage glaucoma. During the test, a patient indicates which light signals are visible within their field of vision, building a map of each eye’s visual function.
Vision Field Test for Glaucoma Detection

The optic nerve (a nerve fiber layer of the retina consisting of the axons of the ganglion neurons coursing on the vitreal surface of the retina to the optic disk) transmits visual information from the retina to the brain. Each part of the retina transmits data via a corresponding set of fibers within the optic nerve. Damage to specific nerve fibers results in loss of the associated portion of the visual field.
Challenges with this test include its complexity, especially for older patients, and its subjective nature.
Changes in the visual field determine glaucoma severity. These changes indicate how much of the visual field is already damaged and which parts of the optic nerve are compromised. We call these ‘functional changes‘ as they directly impact visual function.
Fundus photo for Glaucoma detection: What does early glaucoma look like?
Alongside functional changes, Glaucoma causes visible structural changes in the optic nerve that can be observed during a fundus examination. The optic nerve begins at a point on the retina where all the nerve fibers gather, forming the optic disc (or optic nerve head). The nerve fibers are thickest near the optic disc, creating a depression or ‘hole’ within it. As Glaucoma progresses, this depression deepens due to increased pressure inside the eye. This pressure causes mechanical damage to the nerve fibers, leading to thinning and loss of function.
Another crucial area on the retina is the macula, which contains a high density of receptors responsible for image perception. While the entire retina senses images, the macula provides the sharpest, clearest vision. We use this area for tasks like reading, writing, and looking at fine details. Therefore, the damage to the macular area significantly impacts a patient’s visual quality and clarity. Nerve fibers carrying visual information from this crucial region are essential when evaluating the visual field. We prioritize assessing the macula’s health because it directly determines the quality of a patient’s central vision.
Unfortunately, even if the macula is healthy, damage to the nerve fibers transmitting its signals will still compromise vision.
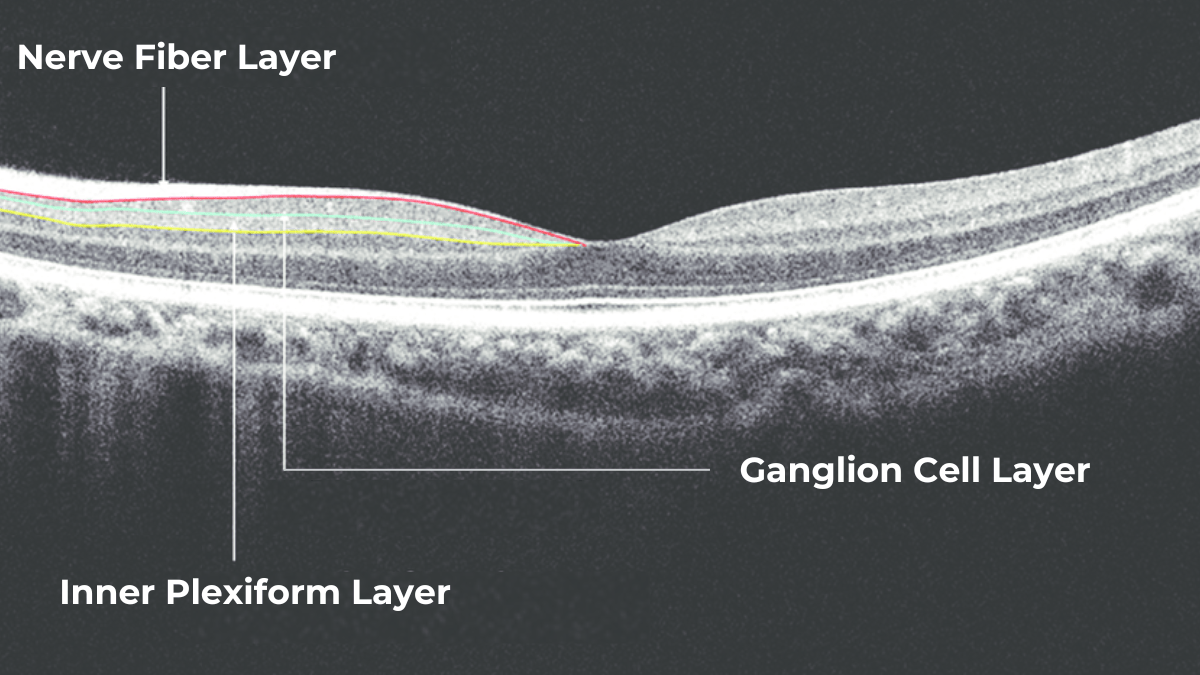
Glaucoma OCT detection
The most effective way to get information about nerve states is OCT, which allows us to penetrate deep into the layers to see the nerve fiber layer separately, making it possible to assess the extent of damage and thinning to this layer in much more detail.

The Glaucoma OCT test provides valuable information about ganglion cells. These cells form the nerve fiber layer and consist of a nucleus and two processes. The short process collects information from other retinal layers, forming the inner plexiform layer. The ganglion cell layer comprises the cell nuclei, while the long processes extend out to create the nerve fiber layer.
Damage to the ganglion cells or their processes leads to thinning across these layers, which we can measure as the thickness of the ganglion cell complex. OCT often detects these microscopic changes before we can see them directly. This enables the detection of structural changes alongside the functional changes observed with standard visual field tests.
Ideally, OCT would be more widely accessible, as the human eye cannot detect early changes. However, how often a patient undergoes OCT depends on various factors. These include the doctor’s proficiency with the technology, the patient’s financial situation (as OCT can be expensive), and the overall clinical picture.
Ways to Enhance Early Glaucoma Detection
We surveyed eye care specialists, and there was a strong consensus that the most efficient ways to boost early glaucoma detection are regular eye check-ups (47%) and utilizing AI technology (40%). Educating patients was considered less significant (13%).

AI as a second opinion tool
AI offers valuable insights into glaucoma detection, analyzing changes that may not be visible to the naked eye or even on standard OCT imaging.
The Altris AI Early Glaucoma Risk Assessment Module specifically focuses on analyzing the OCT ganglion cell layer, measuring its thickness, and identifying any thinning or asymmetry. These measurements help determine a patient’s glaucoma risk. If the ganglion cell complex has an average thickness and is symmetrical throughout the macula, the module will assign a low probability of Glaucoma.

Asymmetries or variations in thickness increase the calculated risk, indicated by a yellow result color. Glaucoma GCC is often characterized by thinning or asymmetry, suggesting glaucomatous atrophy, indicating a high risk, and triggering a red result color.
Changes are labeled as ‘risk’ rather than a diagnosis, as other clinical factors contribute to a confirmed glaucoma diagnosis. Indicators of atrophy could also signal different optic nerve problems, such as those caused by inflammation, trauma, or even conditions within the brain.

It’s crucial to remember that AI ganglion cell layer OCT detection tools like this are assistive – they cannot independently make a diagnosis. Similarly, while helpful in assessing risk, they cannot completely rule out the possibility of developing a disease. This limitation stems from their reliance on a limited set of indicators. Like other technical devices, the module helps flag potential pathology but does not replace the clinician’s judgment.
AI can be incredibly valuable as a supplemental tool, especially during preventive exams or alongside other tests, to catch possible early signs of concern. However, medicine remains a field with inherent variability. While we strive for precise measurements, individual patients, not just statistical averages, must be considered.
Therefore, it is unrealistic to expect devices to provide definitive diagnoses without the context of a complete clinical picture.
Public Health Education

The asymptomatic nature of Glaucoma in its early stages, paired with limited public awareness, creates a fundamental barrier to early detection.
For example, 76% of Swiss survey respondents could not correctly describe Glaucoma or associate it with eye health.
A Canadian study similarly shows that less than a quarter of participants understand eye care professionals’ roles correctly and that most people are unaware eye diseases can be asymptomatic.
Crucially, these studies also found a strong desire across populations for more information about eye care, including Glaucoma (e.g., 97% of Swiss respondents agreed the public lacks knowledge, and 71% want more information). This indicates a receptive audience for targeted education initiatives.
Health education programs, like the USA EQUALITY study, demonstrate the potential to address this challenge. This study combined accessible eye care settings with a culturally sensitive eye health education program, targeting communities with high percentages of individuals at risk for Glaucoma.

Participants showed significant improvements in both glaucoma knowledge (a 62% increase in knowledge questions) and positive attitudes toward the importance of regular eye care (52% improvement).
These results show us that improving glaucoma detection involves more than medical tools. Successful education strategies should prioritize community outreach, partnering with community centers, primary care clinics, and local organizations to reach those lacking access or awareness of regular eye care.
Information about Glaucoma must be presented clearly and accessible, focusing on the basics—what Glaucoma is, its risk factors, and the importance of early detection. Addressing common misconceptions, such as the belief that Glaucoma can’t be present if vision is good, is crucial, as is targeting high-risk groups, including older adults, those with a family history of Glaucoma, and certain ethnicities.
Screening Programs and Regular visits
Community-based studies consistently demonstrate the benefits of targeted screening programs for early glaucoma detection in high-risk populations.
These programs are essential, as traditional glaucoma screening methods often miss individuals with undetected disease.

The USA Centers for Disease Control and Prevention (CDC) funded SIGHT studies focused on underserved communities, including those in urban areas with high poverty rates (MI-SIGHT, Michigan), residents of public housing and senior centers (NYC-SIGHT, New York), and the rural regions with limited access to specialist eye care (AL-SIGHT, Alabama). These programs successfully reached populations who often don’t have regular eye care.
Notably, the results across all three studies demonstrate the effectiveness of targeted programs – approximately 25% of participants screened positive for Glaucoma or suspected Glaucoma.
The SIGHT studies recognize that screening is just the first step, highlighting the importance of follow-up care, testing ways to improve follow-through, using strategies like personalized education, patient navigators, financial incentives, and providing free eyeglasses when needed.
Summing up
Glaucoma’s insidious nature demands better early detection strategies. While existing methods are essential, we must also invest in new technologies like AI, enhance public health education about Glaucoma, and focus on targeted screening within at-risk populations. Combining these approaches can protect sight and reduce the burden of glaucoma-related blindness.
Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.
-

Effective Eye Care Innovation: Altris for the Eye Place
 Altris Inc.
1 min.
Altris Inc.
1 min.The Client: the Eye Place is an optometry center in Ohio, the United States. It is a renowned center that provides comprehensive eye examinations, infant and pediatric eye care, emergency care, LASIK evaluations, and cataract assessment. They offer precise personalized care plans to better treat and prevent ocular disease and chronic illness. Scott Sedlacek, the optometry center owner, is an experienced OD, an American Optometric Association member, and a true innovator who implemented AI for OCT in the optometry practice among the first in the USA.
The Problem: The Eye Place owner has always been searching for innovations to transform the center making it truly digital. The aim of the innovation was also to augment the analysis ability of the optometry specialists using it, while allowing for better visualization of the retinal layers affected for doctors and patients.
The Solution: The Altris AI system was introduced in the Eye Place and it transformed the practice making it more efficient. Scott Sedlacek, the owner of the practice admits that:
“We are one of the first Optometry offices with this AI technology. It is amazing at detecting and defining pathology in the 3D digital images I take with my Topcon Maestro2 OCT. We use Image Net6 software to export Dicom files to Altris AI. It’s fast and easy. If you want the right diagnosis, right away, this is the way to go.
I’ve been using this technology on every patient every day since the beginning of January 2024. There is no other technology in my 25 years being an optometrist that was easier to implement and more impactful immediately.”

ROI of the AI for OCT scan analysis
Many eye care specialists worry about the ROI of Altris AI: will the system pay off? After all, it is an investment. That is the experience of Scott, the owner of the Eye Place:
“Altris AI identified and described pathology that I could not. Early detection changes the treatment from doing nothing to something. Also, Altris AI described something that I thought was worse than it was. Saved me from over-referring. Patients love to see the color-coded images which help as an educational tool and get buy-in on the treatment plan which helps compliance. There is a wow factor for me and my patients that sets your practice apart from the others.”
Effective Eye Care Innovation: What Else?
Apart from AI for OCT analysis, the Eye Place utilizes advanced technology for diagnostics.
- For instance, 3D OCT equipment is a highly advanced screening system that checks for serious conditions such as glaucoma, diabetes, macular degeneration, vitreous detachments, and more. Using this technology we can simultaneously take a digital photograph and a 3-D cross-section of the retina.
- Additionally, AdaptDX Pro can detect macular degeneration earlier than by any other means.
- Cognivue Thrive is a personalized, consistent, and reliable way to receive an overall screening of brain health.It is interactive, non-invasive, self-administered, secure, and confidential. It is a five-minute screening for patients of all ages, and you get immediate results in a simple 1-page report.
These are just some examples of innovative tools that optometry centers can use to automate and improve the level of diagnostics. If you want to imagine how Optometry Centers might look like in 2040, here is the article for you. The future is here, and those centers that digitalize have more chances of winning the competition and the hearts of the clients, much like the Eye Place which is highly appreciated by patients.
As you see, effective eye care innovations are an integral part of the work of the Eye Place which is why Artificial Intelligence for OCT analysis was seamlessly integrated into the workflow of the optometry center.
Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.





































 As David Parkins, the ex-president of the College of Optometrists,
As David Parkins, the ex-president of the College of Optometrists,