
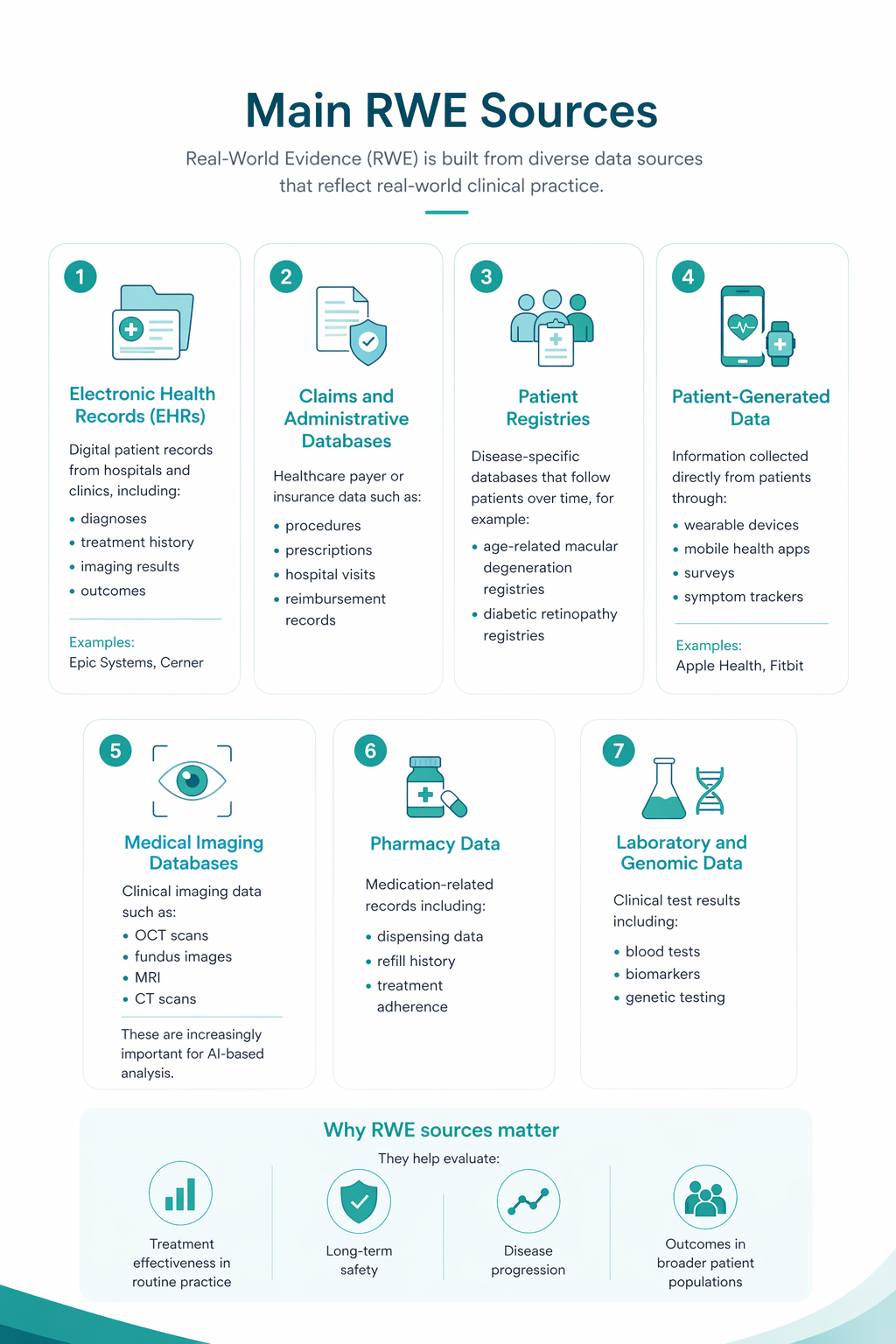
Pharma has no shortage of data in ophthalmology—EHRs, imaging repositories, claims, registries—but the industry still faces a persistent RWE gap when it comes to turning that data into commercially actionable insight.
This gap becomes especially critical in the context of modern ophthalmic therapies, where timing, disease stage, and adherence directly impact outcomes—and, by extension, market performance. Without precise, scalable ways to identify eligible patients (e.g., early-stage AMD, DME with specific biomarkers), commercial teams are left relying on proxies or delayed claims data. At the same time, tracking real-world outcomes remains reactive and retrospective, limiting the ability to support value-based narratives, optimize field strategy, or respond dynamically to physician behavior.
For commercial and market access leaders, closing the RWE gap is no longer a “data strategy” initiative—it’s a growth imperative. The organizations that will lead are those that move beyond passive data aggregation toward active, AI-driven interpretation of multimodal ophthalmic data, enabling near real-time patient identification and outcome tracking. This shift not only strengthens evidence generation but directly translates into sharper targeting, more credible value communication, and ultimately, stronger adoption curves in an increasingly competitive therapeutic landscape.
RWE Key Components
Real-world evidence (RWE) in ophthalmology is fundamentally multimodal—it emerges from the combination of
- OCT imaging,
- clinical metrics,
- treatment patterns,
- and patient context over time.
When these components are connected, they move beyond descriptive data and become decision-grade insight for commercial, medical, and market access teams.
Imaging (OCT): the gold mine of RWE
OCT is the anchor, but only when integrated with outcomes, treatment data, and longitudinal context does it unlock its full commercial value—turning raw data into actionable RWE that can directly shape strategy and growth.
Leverage data from past or ongoing clinical trials with the ability to standardize it within a unified ecosystem. OCT segmentation model can enable you to extract robust, clinically meaningful insights from your trial data to:
- Gain a deeper understanding of patient responses to treatments by characterizing disease progression and outcomes over time.
- Segment patient populations into subgroups to evaluate biomarker-driven profiles and build predictive analytics—helping streamline future trials by refining endpoints and optimizing inclusion and exclusion criteria.
All that, as well as to build one of the most extensive and comprehensive real-world evidence (RWE) databases in ophthalmology may be quite possible within one OCT-vendor neutral data analysis platform ecosystem.
Clinical outcomes: functional reality
Clinical measures—most notably visual acuity (VA), intraocular pressure, and physician-reported assessments—represent the functional impact of disease and treatment. These endpoints are still central to regulatory and commercial narratives, but in isolation they are often lagging indicators. By the time vision declines, disease progression may already be advanced. When paired with imaging, however, clinical outcomes provide the critical link between anatomical change and patient benefit, strengthening real-world value stories and payer communication.
RWE delivers critical insights into how therapies perform in routine clinical practice—such as healthcare outcomes, treatment adherence, and protocol efficiency—often revealing patterns that differ from those observed in controlled clinical trials.

Treatment data: what actually happens in practice
Treatment data captures real-world behavior—which therapies are used, dosing frequency, switching patterns, and adherence. This is where the gap between clinical trial protocols and actual practice becomes visible. For commercial teams, this layer reveals drop-off points, under-treatment, and competitive dynamics at a granular level. When combined with OCT and outcomes, this approach makes it possible to understand not just what is happening but why—for example, whether discontinuation is driven by lack of response, disease stabilization, or operational constraints.
OCT segmentation enables high-throughput processing and deep interrogation of large-scale datasets, enhancing the interpretation of real-world data. It can validate existing assumptions, uncover new patterns, and support hypothesis generation and testing. This analysis provides a clearer view of treatment efficacy and safety, disease progression in real-world settings, and a more precise understanding of target patient populations.
Demographics and patient context
Demographics (age, gender, geography) and broader patient context (comorbidities, access to care) provide the segmentation layer for RWE. These factors influence disease prevalence, treatment eligibility, and adherence patterns. While less granular than imaging, they are essential for market sizing, targeting, and equity considerations, helping commercial leaders understand where the highest-value opportunities—and barriers—exist across populations.
Data analysis includes population-level distributions of retinal layer thickness and fluid volumes; longitudinal tracking of layer and fluid changes over time; and assessment of retinal layer attenuation/loss (depletion maps) and atrophic regions (Figure 2), among other endpoints.
Longitudinal progression: the real differentiator
The true power of RWE lies in its longitudinal nature—tracking how patients evolve over time across imaging, outcomes, and treatment. This enables identification of disease trajectories, early signals of response or non-response, and optimal intervention windows. For pharma decision-makers, longitudinal RWE transforms static snapshots into predictive insight, supporting earlier intervention strategies, more precise patient journeys, and stronger, evidence-backed differentiation in crowded markets.
The Core Problem
There are core RWE problems worth mentioning. pay attention to the following bottlenecks: fragmented data, unstructured imaging, lack of standardization, and broken longitudinal tracking.
- Fragmented data: Patient information is dispersed across multiple systems, limiting the ability to generate a unified, comprehensive view.
- Unstructured imaging: Large volumes of imaging data, such as OCT scans, remain unstructured and difficult to analyze at scale.
- Lack of standardization: Variability in data formats and clinical protocols hinders consistent analysis and comparison.
- Broken longitudinal tracking: Incomplete or disconnected patient timelines prevent accurate assessment of disease progression and treatment outcomes over time.
The core issue isn’t volume; it’s fragmentation and lack of standardization. OCT scans sit in one system, visual acuity in another, treatment histories elsewhere, often unstructured or inconsistently coded. As a result, even well-resourced teams struggle to answer seemingly simple questions like: Who are the untreated but eligible patients? or Which cohorts are actually benefiting from therapy in real-world settings?
However, AI is transforming the RWE in ophthalmology research to:
- Design smarter, biomarker-driven trials
- Estimate disease burden and patient volumes
- Track outcomes, safety, and progression
- Strengthen regulatory and market access strategies
Resources to aid ophthalmologists in evaluating the quality of RWE are available, such as the Good Research for Comparative Effectiveness (GRACE) principles, which can support the evaluation of observational comparative effectiveness studies.
GRACE checklist to support ophthalmologists in the evaluation of RWE:

| Data | Methods |
| ✓ Were treatment and/or important details of treatment exposure adequately recorded for the study purpose in the data source(s)? | ✓ Was the study (or analysis) population restricted to new initiators of treatment or those starting a new course of treatment?
|
| ✓ Were the primary outcomes adequately recorded for the study purpose? | ✓ If one or more comparison groups were used, were they concurrent comparators? If not, did the authors justify the use of historical comparison groups?
|
| ✓ Was the primary clinical outcome(s) measured objectively rather than subject to clinical judgment? | ✓ Were important confounding and effect-modifying variables taken into account in the design and/or analysis?
|
| ✓ Were primary outcomes validated, adjudicated, or otherwise known to be valid in a similar population? | ✓ Is the classification of exposed and unexposed person-time free of “immortal time bias”?
|
| ✓ Was the primary outcome(s) measured or identified in an equivalent manner between the treatment/intervention group and the comparison group? | ✓ Were any meaningful analyses conducted to test key assumptions on which primary results are based?
|
| ✓ Were important covariates that may be known confounders or effect modifiers available and recorded? |
|
*Table adapted from Dreyer NA et al. J Manag Care Pharm 2014; 20 (3): 301–308 (Table 1). While used with permission of the publisher, the publisher disclaims all endorsement of any organization, product or technique as a matter of policy.

Why Imaging Is Critical?
OCT is a stepping stone to understanding Geographic Atrophy. Since the approval of the first therapy targeting geographic atrophy in early 2023, interest in the disease has increased dramatically. At the same time, a growing number of clinical trials are underway, evaluating the safety and efficacy of multiple investigational compounds.
Imaging—particularly OCT (optical coherence tomography)—is the backbone of meaningful real-world evidence in retinal disease, because it captures what clinical codes and claims data simply cannot: anatomical change over time. Without structured OCT data, RWE becomes fragmented and largely inferential, relying on indirect proxies like treatment patterns or visual acuity alone. This creates a major blind spot in understanding disease progression, especially in chronic degenerative conditions where structural deterioration often precedes functional loss.
In geographic atrophy (GA), this gap is especially critical for therapies such as Syfovre (pegcetacoplan) and Izervay (avacincaptad pegol). These treatments are designed to slow structural progression, not just improve symptoms, meaning their real-world impact can only be properly assessed through consistent, longitudinal imaging markers—lesion growth, retinal layer integrity, and atrophy expansion. When OCT data is unstructured or missing, it becomes impossible to reliably track these anatomical endpoints across time and across care settings.
As a result, RWE datasets without standardized OCT integration fail to support robust patient journey reconstruction, dilute treatment effect signals, and limit the ability to identify responders vs non-responders. For pharma and clinical stakeholders, this means missed opportunities to demonstrate value, optimize patient selection, and build predictive models that depend on continuous structural imaging rather than episodic clinical snapshots.
The Way Forward
AI-driven structuring of imaging data is emerging as the missing link between raw clinical information and truly actionable real-world evidence (RWE). In ophthalmology, vast volumes of OCT scans remain underutilized because they are stored as unstructured images, making large-scale analysis slow, inconsistent, and often impractical.
By applying advanced algorithms to automatically segment retinal layers, detect biomarkers, and standardize measurements, platforms like Altris AI transform imaging data into structured, quantifiable, and interoperable datasets. This enables pharma and clinical teams to move beyond anecdotal insights toward statistically robust, evidence-driven decision-making.
With AI-powered structuring, imaging data becomes scalable and longitudinal by design. Instead of isolated snapshots, clinicians and researchers gain continuous, comparable measurements across time, patients, and sites. This unlocks real-time monitoring of disease progression and treatment response, supports precise patient stratification, and accelerates cohort identification for therapies such as GA treatments.
Ultimately, structured imaging powered by AI bridges the gap between clinical practice and research—turning OCT into a high-value, real-time RWE engine that is both clinically meaningful and commercially actionable.
References:
https://onlinelibrary.wiley.com/doi/full/10.1111/aos.14698
https://www.visionacademy.org/media/3251/download
