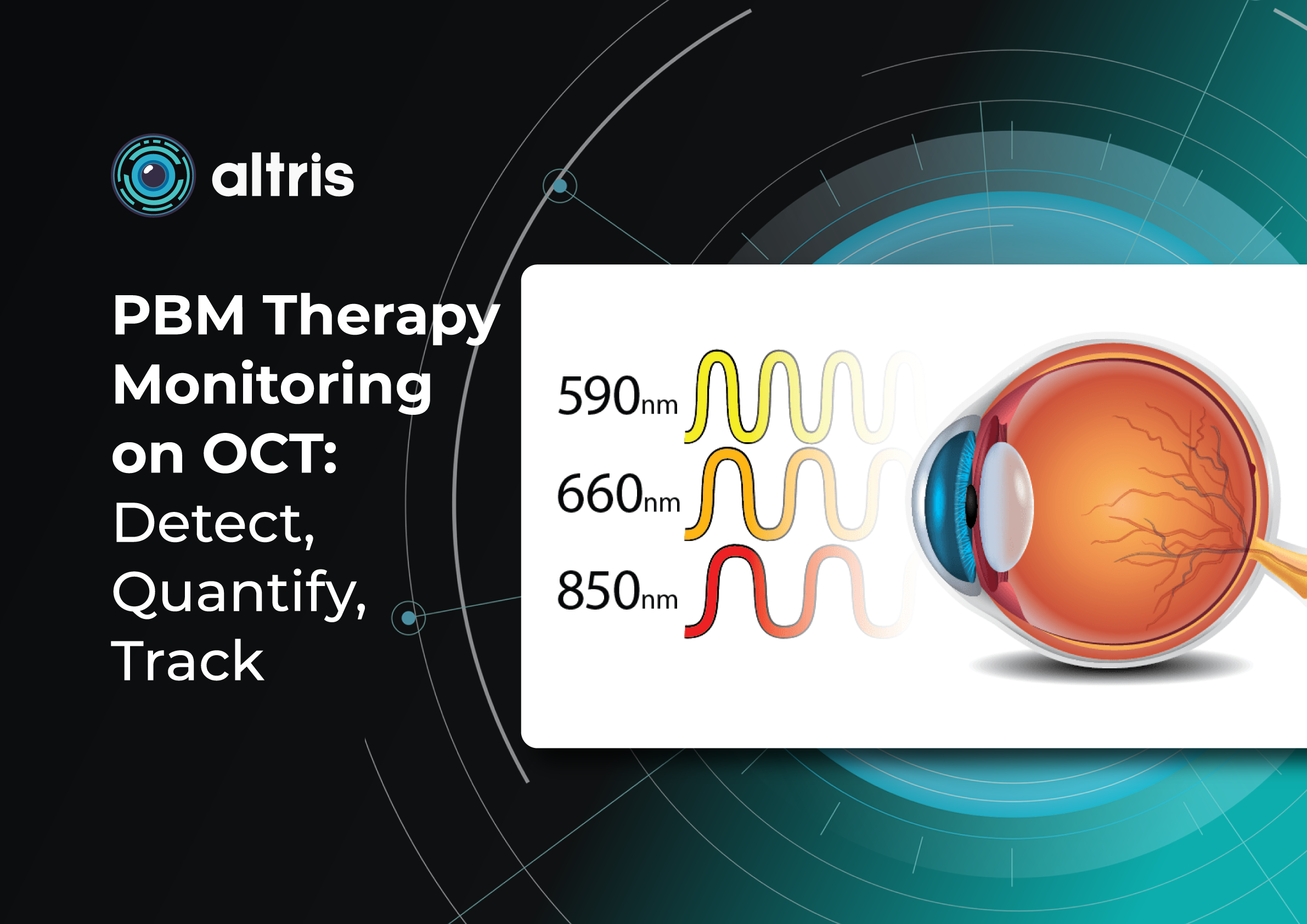
Introduction: Role of PBM in Retinal Disease Management
Photobiomodulation (PBM), also referred to as low-level light or laser therapy, has emerged as a promising non-invasive therapeutic strategy in ophthalmology, particularly for the management of retinal diseases. PBM utilizes low-energy light in the red-to-near-infrared spectrum (typically 600–1000 nm) to modulate cellular function through photochemical rather than thermal mechanisms.
At the cellular level, PBM primarily absorbs photons from mitochondrial chromophores, most notably cytochrome c oxidase, thereby enhancing mitochondrial respiration and increasing adenosine triphosphate (ATP) production. This process is accompanied by modulation of reactive oxygen species (ROS) and activation of intracellular signaling pathways, collectively promoting cellular survival, repair, and regeneration. Importantly, PBM has also been shown to reduce oxidative stress and inflammation—two central mechanisms implicated in the pathogenesis of many retinal disorders—by decreasing pro-inflammatory cytokines and enhancing antioxidant defenses.
The retina, characterized by high metabolic demand and susceptibility to oxidative damage, represents a particularly suitable target for PBM therapy. Experimental and clinical studies have demonstrated that PBM can improve photoreceptor survival, support retinal pigment epithelium (RPE) function, and enhance overall retinal homeostasis. In animal models, PBM has been associated with preservation of retinal structure and improved electrophysiological responses, while early clinical studies suggest potential benefits for visual acuity, contrast sensitivity, and reductions in pathological features such as drusen and retinal edema.
Clinically, PBM is being explored as an adjunctive or alternative treatment for several retinal diseases, including age-related macular degeneration (AMD), diabetic retinopathy (DR), and diabetic macular edema (DME). Its advantages include a favorable safety profile, non-invasive delivery, and relatively low cost compared to conventional therapies. Furthermore, advances in light-emitting diode (LED) technology have facilitated broader clinical application by enabling safe, uniform, and cost-effective retinal illumination.
Key OCT Biomarkers to Track During PBM Therapy
Optical coherence tomography (OCT) has become an indispensable tool for monitoring retinal structure and treatment response in patients undergoing photobiomodulation (PBM) therapy. Given the mechanism of PBM—targeting mitochondrial function, reducing oxidative stress, and modulating inflammation—several OCT-derived biomarkers are particularly relevant for assessing therapeutic efficacy of dry AMD progression and in other retinal disorders. Track and quantify the following biomarkers with Altris AI:
1.PBM monitoring on OCT: Drusen Progression
For patients on photobiomodulation (PBM), OCT monitoring of drusen is about one core question: Are we stabilizing or reversing RPE–Bruch’s membrane dysfunction, or is the eye still progressing toward atrophy? So, on B-scans, drusen are seen as:
- RPE elevations (dome-shaped or irregular)
- Material between RPE and Bruch’s membrane
- Variable internal reflectivity
Here are the key drusen biomarkers to track under PBM:
- Drusen volume (MOST important)
- Measured via OCT segmentation (cube scans)
- Represents total disease burden
As the PBM goal here is the stabilization or reduction in drusen volume
Red flag:
- Continuous increase → disease progression
- Drusen height & area
- Local structural impact on photoreceptors
PBM signal:
- Flattening = potential response
- Increasing height = worsening RPE dysfunction
- Internal reflectivity
- Homogeneous vs heterogeneous content
An important nuance is that increasing heterogeneity may indicate:
- calcification
- regression OR collapse before atrophy
So, it needs to be correlated with other signs.
You may observe Drusen regression patterns. However, not all regression is good.
“Good” regression:
- Gradual flattening
- No photoreceptor loss
- Stable RPE
“Bad” regression (collapse):
- Sudden disappearance
- Followed by:
- RPE loss
- outer retinal thinning
Leads to geographic atrophy (GA)
To summarise, for PBM-treated patients, prioritize:
- Drusen volume trend (longitudinal)
- Photoreceptor integrity (EZ/ONL)
- Signs of atrophy risk (HRF, collapse patterns)
Dry AMD progression matters in PBM monitoring. PBM is currently aimed at:
- early → intermediate dry AMD
So when you monitor drusen on OCT, you’re not just tracking morphology — you’re tracking disease trajectory: Is the eye staying in intermediate AMD, or moving toward advanced stages (GA / nAMD)?
2. Central retinal thickness (CRT)
In addition to PBM monitoring on OCT: drusen progression, one of the primary biomarkers is also central retinal thickness (CRT), which reflects changes in retinal edema and overall retinal integrity. Reductions in CRT during PBM therapy may indicate decreased inflammatory activity and improved fluid homeostasis, particularly in conditions such as diabetic macular edema (DME) and neovascular retinal diseases. However, in non-exudative conditions such as dry age-related macular degeneration (AMD), CRT changes may be subtle, necessitating the evaluation of additional structural parameters.
3. The outer retinal layers
The outer retinal layers, especially the integrity of the ellipsoid zone (EZ) and external limiting membrane (ELM), represent critical biomarkers of photoreceptor health. PBM has been associated with improved mitochondrial activity within photoreceptors, and preservation or restoration of EZ continuity on OCT may serve as a surrogate marker of functional recovery. Disruptions in these layers are strongly correlated with visual impairment, making them highly relevant endpoints in PBM studies.
4. Retinal pigment epithelium (RPE)
Another key biomarker is retinal pigment epithelium (RPE) morphology, including the presence and evolution of drusen, subretinal drusenoid deposits (SDD), and RPE irregularities. PBM has been hypothesized to enhance RPE function and reduce oxidative burden, potentially leading to stabilization or regression of drusen volume over time. Quantitative drusen analysis using OCT can therefore provide insight into disease modification, particularly in intermediate AMD.
5. Hyperreflective foci (HRF)
Hyperreflective foci (HRF) are also important indicators of retinal inflammation and microglial activation. A reduction in HRF number or density during PBM therapy may reflect decreased inflammatory signaling, aligning with the known anti-inflammatory effects of light-based treatment. Similarly, subretinal and intraretinal fluid—when present—should be carefully monitored, as their resolution may indicate improved retinal barrier function and treatment response.
6. Other
Emerging OCT biomarkers include choroidal thickness and choriocapillaris integrity, which are increasingly recognized as contributors to the pathophysiology of retinal disease. PBM may influence choroidal perfusion and metabolic exchange, and changes in choroidal thickness could serve as an indirect marker of vascular and metabolic improvements.
PBM Treatment Monitoring Protocol (Step-by-Step)
1. Patient Selection and Baseline Assessment
Appropriate patient selection is critical to optimizing outcomes of photobiomodulation (PBM) therapy in dry AMD progression. Current evidence supports its use primarily in non-exudative retinal diseases, particularly intermediate Age-related Macular Degeneration, as well as emerging applications in Diabetic Retinopathy and Diabetic Macular Edema.
Inclusion considerations:
- Intermediate AMD (presence of drusen and/or subretinal drusenoid deposits)
- Stable retinal conditions without active neovascularization
- Best-corrected visual acuity (BCVA) is sufficient for functional monitoring
Exclusion criteria:
- Active neovascular AMD or significant intraretinal/subretinal fluid
- Recent anti-VEGF injections (unless PBM is used adjunctively in controlled settings)
- Significant media opacity affecting light delivery or imaging quality
Baseline evaluation should include:
- Visual function testing: BCVA, contrast sensitivity
- Structural imaging: spectral-domain OCT (mandatory)
- Optional advanced imaging: OCT angiography (OCTA) for vascular assessment
- Key OCT biomarkers (baseline reference):
-
- Central retinal thickness (CRT)
- Ellipsoid zone (EZ) integrity
- Retinal pigment epithelium (RPE) status and drusen volume
- Presence of hyperreflective foci (HRF)
Establishing a robust baseline is essential, as PBM-induced changes are often gradual and require longitudinal comparison.
2. Treatment Session Procedure
PBM is delivered using low-level light in the red-to-near-infrared spectrum (typically ~600–1000 nm), most commonly via LED-based systems designed for retinal applications.
Standard session workflow:
- Patient preparation
- No pharmacologic dilation is typically required (device-dependent)
- Proper alignment and fixation ensured
- Device application
- Light delivered trans-pupillary using controlled, non-thermal energy
- Multi-wavelength protocols (e.g., combinations of ~590 nm, 660 nm, 850 nm) are commonly used in clinical studies
- Treatment duration
- Typically, a few minutes per eye per session (device-specific)
- Sequential or simultaneous bilateral treatment, depending on the system
- Safety monitoring
- PBM is non-invasive and well-tolerated
- No significant adverse retinal effects have been reported in the current literature
- Monitor for discomfort or visual disturbances (rare)
Immediate post-session:
- No recovery time required
- Patients resume normal activities immediately
The mechanism of action—enhancing mitochondrial activity and reducing oxidative stress—does not produce immediate anatomical changes, reinforcing the need for structured follow-up.
3. Treatment Series and Frequency
PBM is not a single-session therapy for observing dry AMD progression or any other condition, but is administered as a treatment series, followed by monitoring and potential retreatment cycles.
Typical treatment regimen (based on clinical studies):
- Induction phase:
- 2–3 sessions per week
- Duration: 3–5 weeks
- Total: ~9–12 sessions per cycle
- Follow-up period:
- Reassessment at 1–3 months post-treatment
- OCT imaging to evaluate structural response
- Retreatment strategy:
- Repeat cycles every 4–6 months, depending on disease progression and response
- Individualized based on OCT biomarkers and functional outcomes
Monitoring during and after therapy:
- Short-term (during treatment):
- Limited structural change expected
- Intermediate-term (1–3 months):
- Possible reduction in drusen volume
- Stabilization of EZ and RPE integrity
- Decrease in HRF (inflammatory markers)
- Long-term:
- Disease stabilization rather than reversal is the primary goal
Outcome measures:
- Functional: BCVA, contrast sensitivity
- Structural: OCT biomarkers (drusen, EZ, CRT, HRF)
- Optional: OCTA vascular parameters
A structured PBM protocol integrates careful patient selection, standardized treatment delivery, and longitudinal OCT-based monitoring. The therapy is best suited for chronic, non-exudative retinal conditions, where its cumulative biological effects—rather than immediate anatomical changes—drive clinical benefit. Consistent imaging and biomarker tracking are essential for guiding retreatment decisions and evaluating long-term efficacy.
Devices & AI for PBM Monitoring
LumiThera (Valeda)
Valeda is a photobiomodulation (PBM) device — currently the most clinically validated retinal light therapy. Company: LumiThera (now acquired by Alcon) Mechanism (PBM):
- Uses multiwavelength light:
- ~590 nm
- ~660 nm
- ~850 nm
- Targets mitochondria → increases ATP
- Reduces:
- oxidative stress
- inflammation
- Improves retinal cellular function
Clinical positioning
- Indicated for:
- early–intermediate dry AMD
- First device to:
- demonstrate visual improvement, not just slowing the disease
Regulatory & evidence strength:
- FDA-authorized (De Novo, 2024)
- CE marked, global use
- Backed by:
- LIGHTSITE trials (I–III)
- Demonstrated:
- ~1 line BCVA improvement over 24 months
Treatment protocol
- Standard cycle:
- 9 sessions over 3–5 weeks
- Repeat:
- every 4–6 months
- Fully clinic-based
Clinical effects
- Functional:
- ↑ visual acuity
- ↑ contrast sensitivity
- Structural (OCT):
- drusen stabilization/reduction
- preservation of retinal layers
- Strong safety profile
Thus, Valeda is currently:
The only FDA-authorized retinal PBM device
The only one with phase III randomized clinical trial data (LIGHTSITE)
And it has shown actual visual acuity improvement, not just safety.
Espansione
This is an ophthalmology PBM platform (incl. retina use). Espansione develops photobiomodulation (PBM / LLLT) devices specifically for eye care. Espansione is a multi-technology ophthalmic platform, not just a PBM lamp.
It combines:
- LM™ LLLT (photobiomodulation / PBM) → core tech for retina
- OPE™ IPL → only for anterior segment
Their flagship eye-light® is used for:
- anterior segment (DED, MGD)
- posterior segment (including dry AMD progression)
- It has CE approval for retinal conditions like dry AMD
- The company explicitly states expansion into posterior segment care (retina)
This is another ophthalmic PBM device platform, with retina applications.
Light characteristics
- Wavelength range: ~500–1000 nm
- Typical retina protocol:
- Yellow (~590 nm)
- Red (~625 nm)
- Delivered via LED mask over closed eyes
Mechanism (important for retina positioning)
PBM targets mitochondria:
- ↑ ATP production
- ↓ oxidative stress (ROS)
- ↑ cellular repair
- ↓ inflammation
For retina specifically:
-
Improves RPE + photoreceptor metabolism
-
Supports choriocapillaris perfusion
-
Slows degeneration processes (AMD biology)
Retina applications
- Dry AMD progression (main indication)
- CE (MDR) cleared for dry AMD (dAMD)
- Focus: early–intermediate stages (AREDS 1–3)
Clinical effects shown:
- ↑ Visual acuity (BCVA gains)
- ↓ Drusen volume
- ↑ Choriocapillaris perfusion
Thus, it is disease-modifying intent, not just symptomatic.
Typical session:
- Duration: ~10–12 minutes
- Mask-based (non-contact)
- Eyes:
- mostly closed
- brief opening phases possible
Typical course:
- ~8 sessions / 3–4 weeks (in studies)
Very different from laser or injections:
- No dilation
- No contact lens
- No invasive steps
Here’s a comparison chart of the key characteristics and features of both devices:
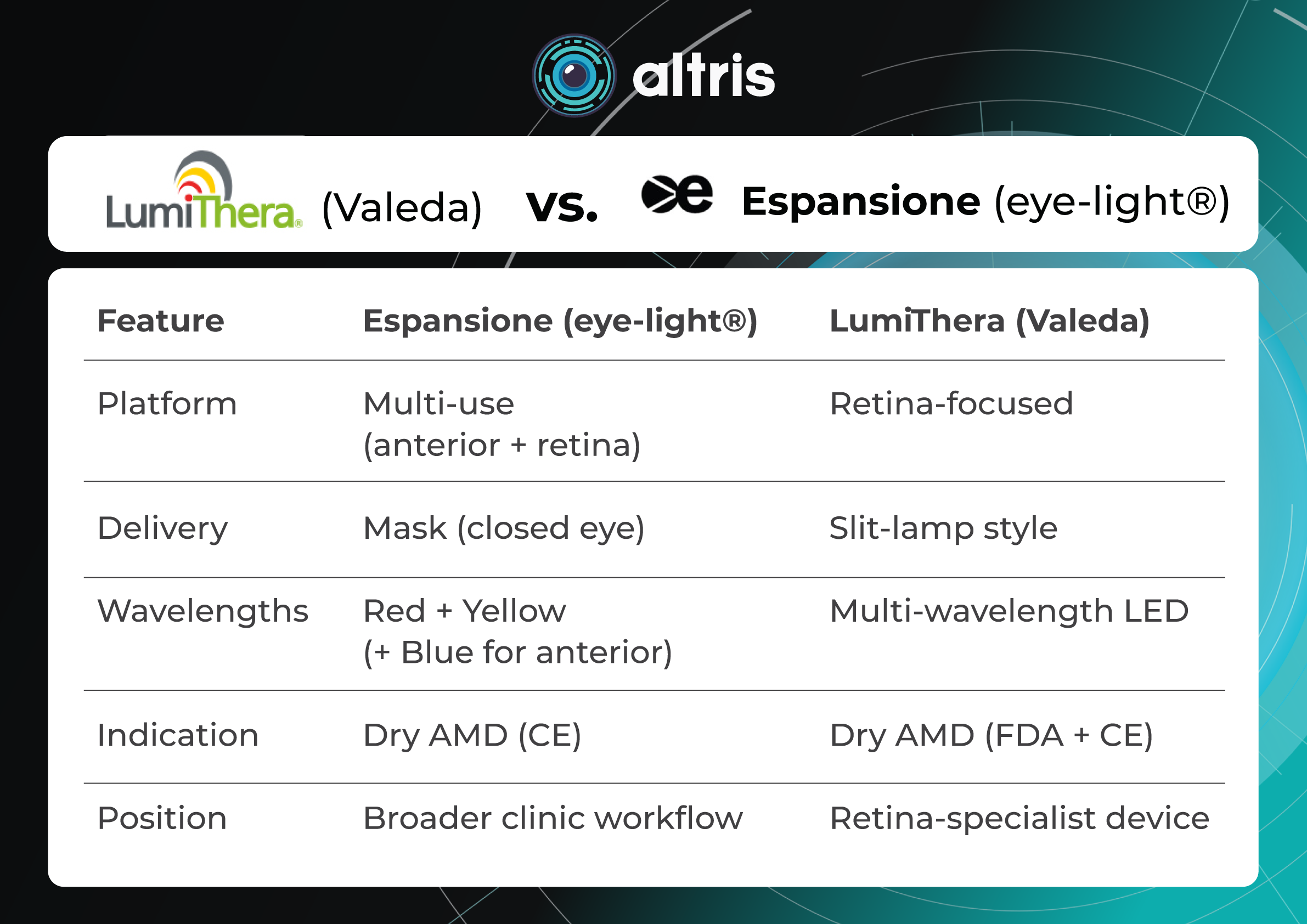
LumiThera (Valeda) vs. Espansione (eye-light®)
| Feature
|
Espansione (eye-light®) | LumiThera (Valeda) |
| Platform | Multi-use (anterior + retina) | Retina-focused |
| Delivery | Mask (closed eye) | Slit-lamp style |
| Wavelengths | Red + Yellow (+ Blue for anterior) | Multi-wavelength LED |
| Indication | Dry AMD (CE) | Dry AMD (FDA + CE) |
| Position | Broader clinic workflow | Retina-specialist device |
Other Generic LED / Low-Level Light Therapy (LLLT) platforms
These are NOT branded retinal systems like Valeda or Espansione, but rather:
- LED arrays or laser-based PBM setups
- used in:
- early trials
- pilot studies
- lab settings
Typical characteristics:
- Wavelengths: 500–1000 nm
- Delivery:
- masks
- panels
- slit-lamp–mounted systems
Examples (non-standardized):
- custom LED rigs used in AMD studies
- early PBM prototypes before Valeda
- academic research devices
Problem:
- No standard protocol
- No regulatory approval
- Hard to compare outcomes
Experimental PBM Directions also include expanded PBM indications of
- diabetic retinopathy (DR)
- diabetic macular edema (DME)
- geographic atrophy
Next-gen PBM systems
Nevertheless, research in eye care is further exploring:
- different wavelength combinations
- dose optimization
- portable/home PBM (not validated yet)
Despite the fact that nothing close to Valeda-level evidence has emerged yet.
Modern PBM ecosystem classification
Tier 1 (validated):
- Valeda (LumiThera / now Alcon)
Tier 2 (investigational retinal PBM):
- academic LED systems
- prototype devices
Tier 3 (not true retinal PBM):
- dry eye LLLT
- cosmetic red light
- wellness devices
Clinical Application and Results
The integration of digital technologies and artificial intelligence (AI) into retinal imaging has significantly enhanced the ability to monitor treatment response in photobiomodulation (PBM) therapy. Given that PBM induces gradual, often subtle structural and functional changes, advanced analytical tools are essential for detecting and quantifying these effects with precision and reproducibility. Here are some real cases of application in Ophthalmology (Dry AMD, DME, etc).
Dry AMD
Photobiomodulation (PBM) has been clinically evaluated primarily in patients with early-to-intermediate Age-related Macular Degeneration, where no widely accepted disease-modifying therapy exists. The most robust evidence comes from the LIGHTSITE clinical trial program, in which PBM is delivered as multiwavelength light therapy (590, 660, and 850 nm) in repeated treatment cycles (typically 9 sessions over 3–5 weeks, repeated every 4 months).
Across the LIGHTSITE I–III studies, PBM has consistently demonstrated functional improvements, particularly in best-corrected visual acuity (BCVA) and contrast sensitivity, and has shown favorable safety outcomes, with no evidence of phototoxicity. In LIGHTSITE II, PBM-treated eyes showed a mean ~4-letter gain in BCVA at 9 months, with approximately one-third of patients achieving ≥5-letter improvement, while sham-treated eyes showed minimal change. Earlier studies also reported improvements in contrast sensitivity, microperimetry, and reductions in drusen burden, suggesting both functional and anatomical benefits .
More recent data from the pivotal LIGHTSITE III trial further support these findings, demonstrating statistically significant gains in visual acuity compared with sham treatment, with mean improvements exceeding 5 letters and a substantial proportion of patients achieving clinically meaningful gains. At 24 months, PBM-treated eyes showed sustained visual improvement (+6.2 letters) and a reduced progression to geographic atrophy (6.8% vs 24.0% in controls), suggesting potential disease-modifying effects.
However, despite these encouraging results, meta-analyses indicate that overall effect sizes remain modest and that variability across studies, small sample sizes, and protocol heterogeneity limit definitive conclusions regarding long-term clinical benefit. Thus, while PBM represents a promising and biologically plausible therapy for dry AMD, its role in routine clinical practice continues to evolve, with ongoing studies needed to confirm durability, optimal patient selection, and real-world effectiveness.
DME
Photobiomodulation (PBM) has been explored as a non-invasive adjunctive or alternative therapy for Diabetic Macular Edema, targeting key pathogenic mechanisms, including mitochondrial dysfunction, oxidative stress, and chronic inflammation.
Unlike anti-VEGF therapy, which primarily addresses vascular permeability, PBM aims to restore retinal metabolic balance through light-induced activation of mitochondrial pathways. Early clinical studies using red-to-near-infrared wavelengths (typically ~630–850 nm) have demonstrated reductions in central retinal thickness (CRT) and improvements in retinal morphology on OCT, alongside stabilization or modest gains in best-corrected visual acuity (BCVA). These effects are particularly notable in mild-to-moderate DME and in patients with non-center-involving edema, where PBM may reduce inflammatory signaling and improve fluid homeostasis.
Clinical data, although still limited compared to age-related macular degeneration, suggest that PBM may have value as an adjunct to standard of care, potentially reducing treatment burden in patients requiring repeated intravitreal injections. Some studies report decreased intraretinal fluid and improvement in OCT biomarkers following PBM treatment cycles, with a favorable safety profile and no evidence of retinal damage.
However, results remain heterogeneous, with variability in treatment protocols, patient populations, and outcome measures. Importantly, PBM has not yet demonstrated efficacy comparable to anti-VEGF therapy in center-involving DME, and its role is best considered complementary rather than substitutive at this stage. Larger randomized controlled trials are needed to define optimal dosing strategies, identify responder phenotypes, and clarify long-term functional benefits in DME management.
Why is OCT Critical for PBM Monitoring?
Optical Coherence Tomography (OCT) is essential for monitoring photobiomodulation (PBM) therapy because PBM aims to induce subtle, progressive structural and functional changes in the retina—especially in conditions like dry AMD progression.
Unlike anti-VEGF treatments, where effects can be more immediate, PBM outcomes are gradual and microstructural, such as changes in drusen volume, RPE integrity, outer retinal layers, and choriocapillaris perfusion. These changes are often invisible on fundus photography or visual acuity alone, making OCT the only practical tool for objective, layer-by-layer tracking over time.
Serial OCT scans allow clinicians to detect early signals of response (e.g., drusen regression or stabilization) and differentiate them from natural disease progression, which is critical for validating PBM efficacy in real-world practice.
AI-Assisted OCT Analysis
Artificial intelligence–driven analysis of optical coherence tomography (OCT) enables automated, quantitative assessment of retinal biomarkers critical to PBM monitoring. Machine learning and deep learning algorithms can segment retinal layers and identify pathological features such as drusen, hyperreflective foci (HRF), and fluid compartments with high accuracy.
In the context of PBM, AI tools provide:
- Automated retinal layer segmentation, including ellipsoid zone (EZ) and retinal pigment epithelium (RPE)
- Quantification of drusen volume and distribution, particularly relevant in Age-related Macular Degeneration
- Detection and tracking of subtle structural changes over time that may not be apparent on qualitative review
These capabilities are especially important because PBM effects are often incremental rather than dramatic, requiring sensitive longitudinal comparison.
Longitudinal Tracking and Progression Analysis
Digital platforms enable time-series analysis of OCT data, allowing clinicians to monitor the disease trajectory before, during, and after PBM therapy. Automated registration of sequential scans ensures that the same retinal locations are compared over time.
Key advantages include:
- Change detection algorithms for early identification of treatment response
- Trend analysis of biomarkers such as central retinal thickness, EZ integrity, and HRF density
- Objective progression metrics, reducing inter-observer variability
Such tools are critical for distinguishing true therapeutic effects from natural fluctuations in disease, particularly in slowly progressing conditions like dry AMD progression.
Digital tools and AI are transforming PBM monitoring by enabling precise, quantitative, and longitudinal assessment of retinal biomarkers. From automated OCT analysis to AI-driven decision support and remote monitoring, these technologies address the key challenge of PBM therapy—detecting subtle, progressive changes over time, like in dry AMD progression, etc. Their continued development will be essential for standardizing PBM protocols and optimizing patient outcomes in retinal disease management.
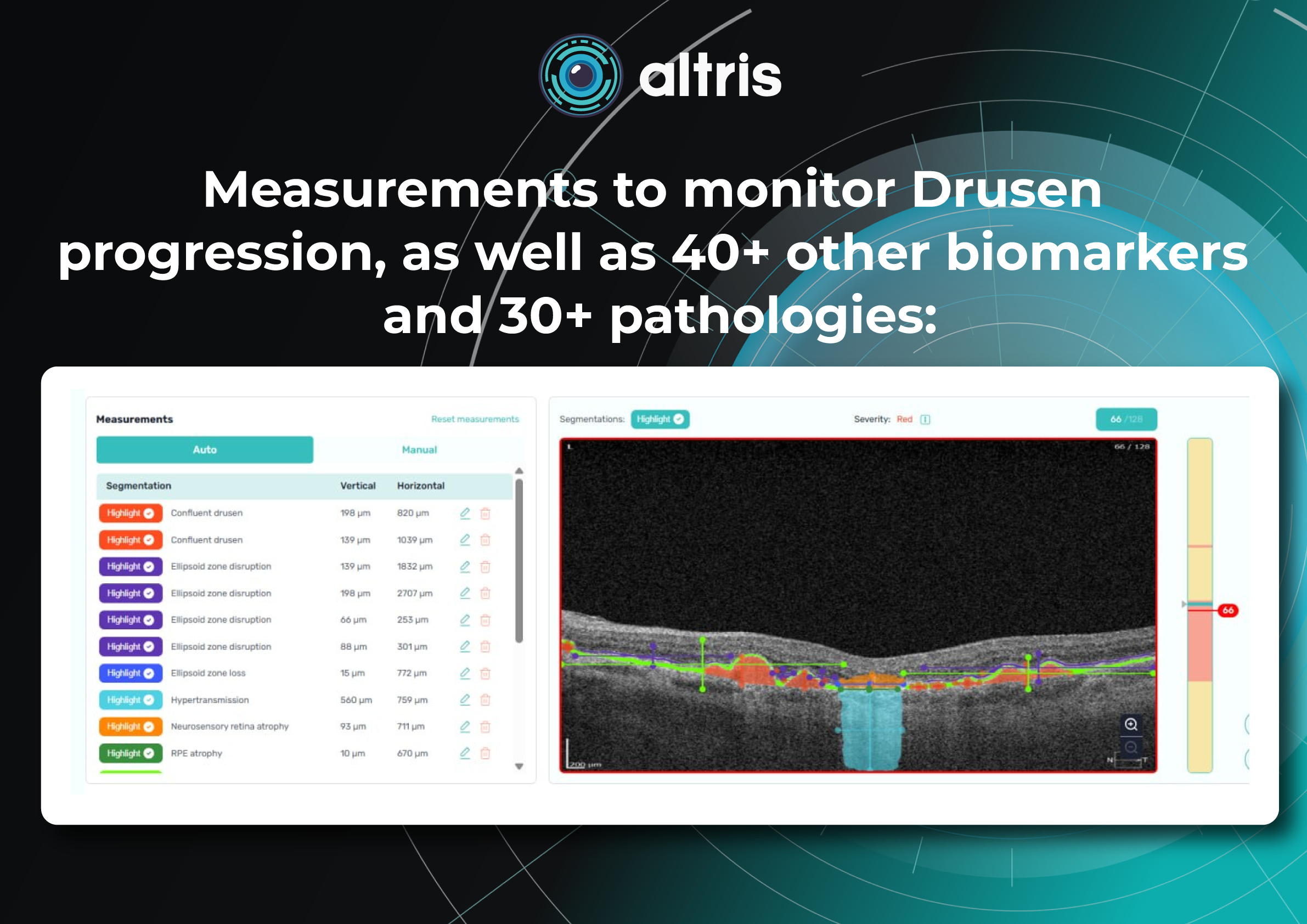
Altris AI for PBM monitoring on OCT: Drusen Progression +40 biomarkers
Altris AI has contributed to PBM monitoring on OCT: Drusen progression, as well as 40+ other biomarkers and 30+ pathologies, which may be monitored with the system. Altris AI enhances this process by turning OCT into a quantitative, standardized monitoring system rather than a subjective review. It automatically segments retinal layers and biomarkers (e.g., drusen, hyperreflective foci, fluid), calculates precise volumetric metrics, and enables longitudinal comparison across visits with high reproducibility. Here are the examples of biomarker monitoring cases via Altris AI:
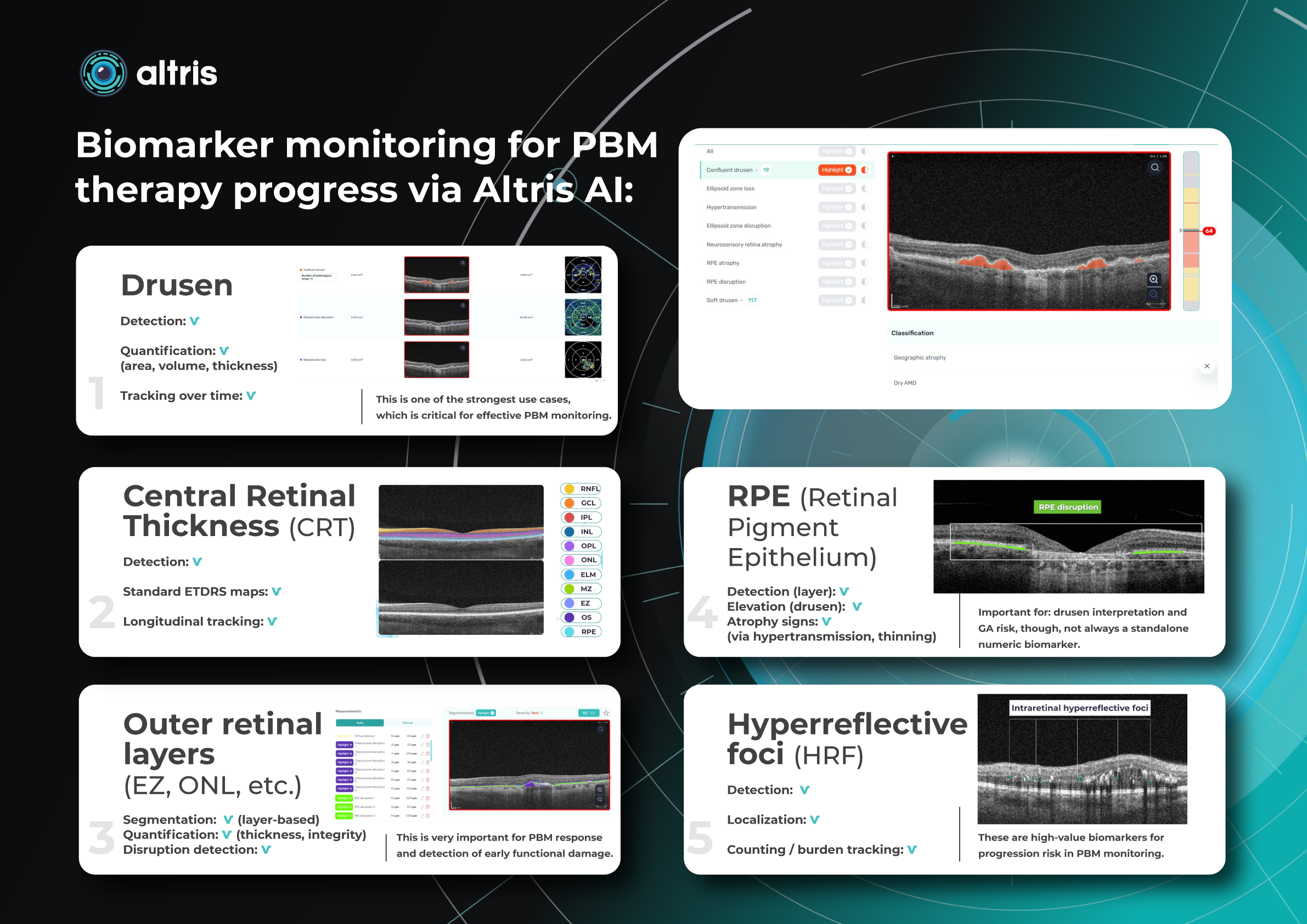
- Drusen
Detection: ✔️
Quantification: ✔️ (area, volume, thickness)
Tracking over time: ✔️
This is one of the strongest use cases, which is critical for effective PBM monitoring.
- Central Retinal Thickness (CRT)
Detection: ✔️
Standard ETDRS maps: ✔️
Longitudinal tracking: ✔️
- Outer retinal layers (EZ, ONL, etc.)
Segmentation: ✔️ (layer-based)
Quantification: ✔️ (thickness, integrity)
Disruption detection: ✔️
This is very important for PBM response and detection of early functional damage.
- RPE (Retinal Pigment Epithelium)
Detection (layer): ✔️
Elevation (drusen): ✔️
Atrophy signs: ✔️ (via hypertransmission, thinning)
Important for: drusen interpretation and GA risk, though, not always a standalone numeric biomarker.
- Hyperreflective foci (HRF)
Detection: ✔️
Localization: ✔️
Counting / burden tracking: ✔️
These are high-value biomarkers for progression risk in PBM monitoring.
Assistance like this allows clinicians to track PBM response objectively, identify responders vs non-responders earlier, and generate consistent reports for clinical decision-making or research. In short, while OCT provides the necessary imaging depth, Altris AI unlocks its full value for PBM by making subtle retinal changes measurable, comparable, and clinically actionable.
Conclusion
PBM represents a novel and biologically plausible therapeutic modality that targets key pathological mechanisms in retinal disease. By enhancing mitochondrial function, reducing oxidative stress, and modulating inflammation, PBM holds significant potential to complement existing treatment strategies and improve outcomes in retinal disease management. However, further research is required to fully define its role in routine clinical practice.
Despite the promising findings, the clinical integration of PBM remains in an evolving stage. Variability in treatment parameters—including wavelength, dose, and treatment protocols—has limited standardization and comparability across studies. Moreover, much of the current evidence is derived from small-scale clinical trials and preclinical models, underscoring the need for large, randomized controlled trials to establish optimal treatment regimens for dry AMD progression and to assess long-term efficacy in other eye pathologies.
In this context, OCT—especially when enhanced with AI-driven analysis—plays a critical role in advancing PBM adoption. Quantitative OCT biomarkers such as drusen volume, outer retinal integrity, and subtle structural changes provide objective endpoints for assessing therapeutic response. AI-based platforms further enable precise, reproducible, and longitudinal analysis of these changes, helping to standardize evaluation, identify responders earlier, and strengthen the clinical evidence base for PBM.
FAQ Section
1. How do I objectively measure response to PBM therapy?
Clinicians look for quantifiable OCT biomarkers, not just visual acuity:
- Drusen volume (regression or stabilization)
- Outer retinal layer integrity (EZ, RPE)
- Hypertransmission / atrophy areas
The challenge: changes are subtle → require precise, longitudinal OCT comparison.
2. Which OCT biomarkers are most relevant for PBM monitoring?
The most discussed and clinically relevant:
- Drusen volume/area
- RPE atrophy
- Hypertransmission
- Ellipsoid Zone (EZ) disruption/loss
- Hyperreflective foci (secondary)
For GA specifically:
Overlap of RPE atrophy + hypertransmission + EZ loss = key composite metric.
3. How often should I monitor patients on PBM?
Typical real-world patterns:
- Baseline OCT before starting PBM
- Follow up every 3–6 months
- More frequent (monthly) in studies.
4. How do I distinguish PBM effect from natural AMD progression?
Distinguishing the effect of PBM from the natural progression of AMD remains one of the key clinical challenges. AMD typically progresses slowly and can show natural fluctuations, while PBM-related changes tend to be gradual and relatively modest. To differentiate between the two, clinicians rely on consistent OCT metrics tracked over time, comparing trends rather than single visits. Bilateral analysis—evaluating treated versus untreated eyes—can provide additional context, while assessing the rate of change, such as slowing of drusen growth or stabilization of atrophic areas, helps determine whether observed changes are likely treatment-related rather than part of the disease’s natural course.
5.Do I need AI/software for PBM monitoring, or is manual OCT review enough?
Whether AI/software is needed for PBM monitoring versus manual OCT review is an increasingly important question in clinical practice. While manual assessment can provide a general, qualitative understanding, it is often variable, time-consuming, and limited in its ability to detect subtle changes. PBM, however, requires identification of micron-level structural differences and high reproducibility across visits to accurately assess treatment response. AI-based OCT analysis addresses these challenges by enabling automated segmentation of key biomarkers, delivering precise volumetric measurements, and supporting reliable longitudinal tracking in standardized units such as mm², mm³, and percentage change. This level of consistency also helps clinicians more confidently distinguish responders from non-responders, making monitoring more objective and clinically actionable.
References:
https://pmc.ncbi.nlm.nih.gov/articles/PMC11488463/
https://link.springer.com/article/10.1007/s40135-025-00340-x
https://d-nb.info/136218389X/34
https://www.frontiersin.org/journals/ophthalmology/articles/10.3389/fopht.2024.1388602/full
https://retinatoday.com/articles/2020-may-june/photobiomodulation-as-a-treatment-in-dry-amd
https://espansionegroup.it/it/
