Table of Contents
The pathogenesis of OCT biomarkers in diabetic macular edema (DME) is complex and multifactorial. The key mechanism is disruption of the inner blood–retinal barrier resulting from chronic hyperglycemia.
- Introduction. Brief overview of diabetic macular edema (DME)
- Main OCT biomarkers of DME
2.1 Signs of DME on OCT
2.2 Quantitative parameters for monitoring DME on OCT
DME management and patient education: key aspects of the modern approach
References
Introduction. Brief Overview of Diabetic Macular Edema (DME)
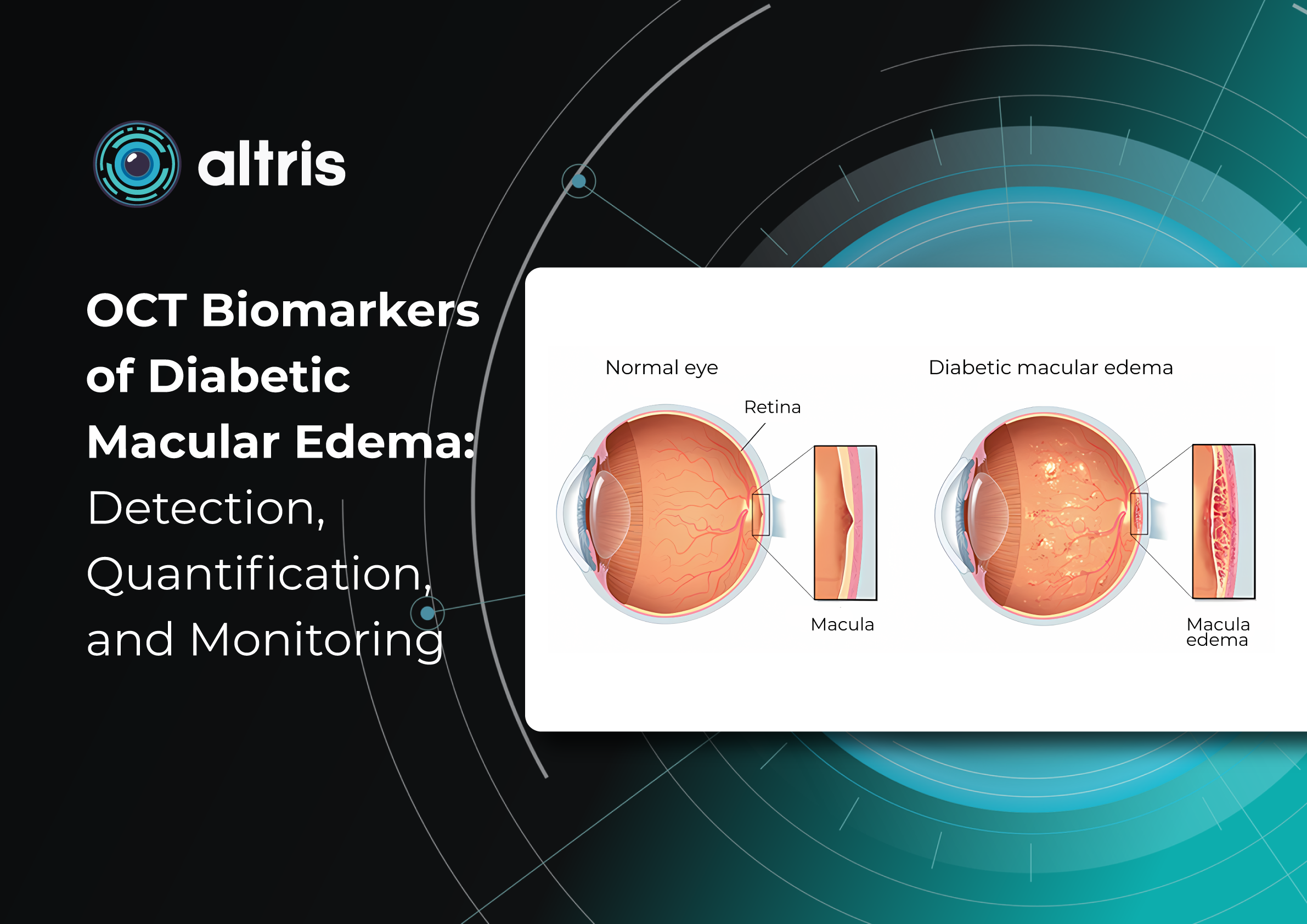
The pathogenesis of OCT biomarkers in diabetic macular edema (DME) is complex and multifactorial. The key mechanism is disruption of the inner blood–retinal barrier resulting from chronic hyperglycemia. This leads to increased vascular permeability, plasma extravasation, and fluid accumulation within the retinal layers. Inflammatory processes, cytokine and growth factor activation—particularly vascular endothelial growth factor (VEGF)—also play an important role by further increasing vascular permeability and sustaining chronic edema. For this reason, DME is now regarded not only as a vascular disorder but also as a neurodegenerative and inflammatory pathology.
Morphologically, DME is characterized by retinal thickening, the formation of intraretinal cystoid spaces, accumulation of subretinal fluid, and progressive photoreceptor damage. Importantly, these structural changes often develop long before the onset of clinical symptoms. A patient may not experience significant visual deterioration, while irreversible changes are already occurring at the microstructural level. This underscores the critical importance of early diagnosis and regular monitoring.
Current clinical guidelines, including those of the American Academy of Ophthalmology, emphasize that the timely detection of DME and the early initiation of treatment significantly improve functional outcomes. However, effective patient management is impossible without precise instrumental monitoring, particularly with optical coherence tomography (OCT), which enables assessment of both the presence and progression of the pathological process.
OCT has become the imaging modality that fundamentally transformed the approach to the diagnosis and treatment of DME. It provides noninvasive visualization of the retina with micron-level resolution, allowing detailed analysis of its layered structure.
Moreover, modern tomographic systems enable a transition from qualitative to quantitative assessment. Measurement of central retinal thickness, lesion area, and other parameters provides objective monitoring of disease progression. This is especially important in the era of personalized medicine, when treatment decisions are based not only on the clinical picture but also on precise numerical indicators.
Thus, modern management of DME cannot be envisioned without the systematic use of OCT and the analysis of its biomarkers. Ophthalmology has progressed from simply detecting edema to achieving a deeper understanding of microstructural changes and their clinical significance.
The aim of this article is to summarize current evidence on OCT biomarkers of diabetic macular edema, their roles in detection, quantitative assessment, and disease monitoring, and the practical aspects of their use in clinical practice to optimize patient management.
2. Main OCT Biomarkers in DME
The modern approach to DME is based on a fundamentally new understanding of OCT’s role. OCT biomarkers in DME enable not only assessment of fluid presence but also determination of its type, localization, severity, and impact on key functional structures, particularly the photoreceptors. This is critically important because DME may have different pathogenic mechanisms across patients, ranging from vascular and inflammatory processes to a predominantly tractional component.
Particular attention should be paid to the role of OCT biomarkers in predicting the course of DME and treatment response. Features such as disorganization of the retinal inner layers (DRIL), the condition of the ellipsoid zone, and the presence of hyperreflective foci are now considered important indicators for predicting disease progression and functional outcomes. These biomarkers enable clinicians to anticipate which patients are more likely to respond favorably to anti-VEGF therapy and in which cases a less favourable visual outcome should be expected.
Each structural element observed on an OCT scan carries its own clinical significance. These findings allow the physician to answer several key clinical questions:
- how active the disease process is,
- whether the condition is acute or chronic,
- which structures have already undergone irreversible changes,
- which pathogenic mechanism predominates,
- and, most importantly, what the likely therapeutic response will be.
Thus, OCT interpretation extends far beyond simple visual assessment. It becomes an analytical process in which the clinician integrates morphological features with quantitative measurements and the patient’s clinical data.
According to current international recommendations, no single parameter—such as central retinal thickness—can be sufficient for clinical decision-making. Instead, analysis of a combination of morphological patterns, quantitative indicators, and their temporal changes is recommended.
The transition from static assessment to dynamic monitoring is particularly important via OCT biomarkers in dme. The rate of change, stability, or progression of individual biomarkers is often more informative than their absolute values. For example, a slight but persistent increase in intraretinal fluid may have greater clinical significance than a single high retinal thickness measurement.
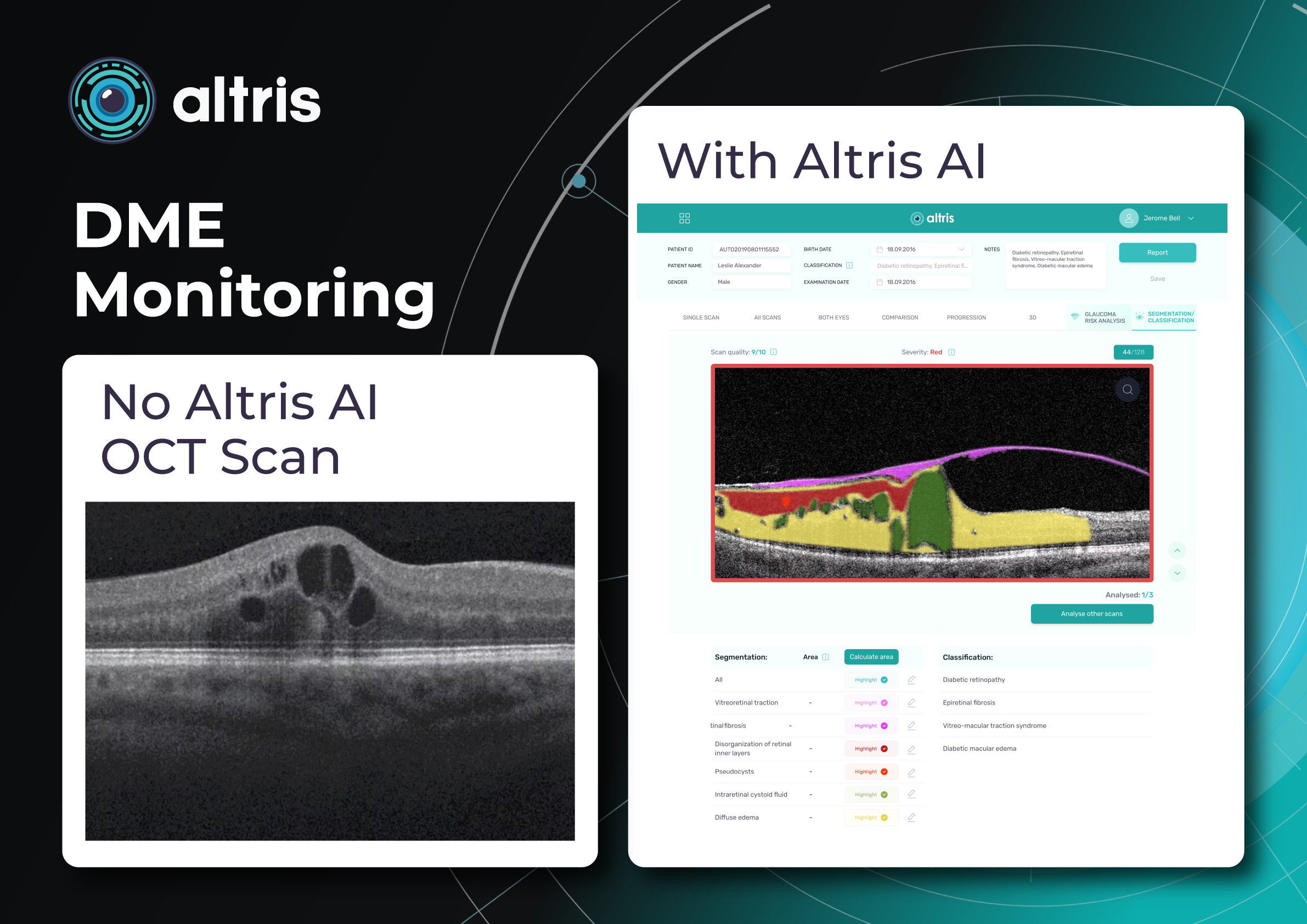
In addition, modern technologies have considerably expanded the capabilities of OCT analysis. The use of automated retinal layer segmentation, quantitative fluid volume assessment, and artificial intelligence algorithms helps reduce subjective interpretation and improve the reproducibility of results. This is especially important both in clinical practice and in scientific research, where accuracy and standardization are essential.
In summary, the contemporary concept of OCT biomarkers in DME is based on three interconnected levels of analysis:
- Morphological level — identification of structural changes and edema type.
- Quantitative level — measurement of retinal thickness, fluid volume, and lesion area.
- Prognostic level — assessment of the risk of progression and treatment response.
It is precisely this multilevel evaluation that enables a transition from standardized treatment protocols to a personalized approach, in which the therapeutic strategy is determined by each patient’s OCT biomarker profile.
What are the key OCT biomarkers for monitoring diabetic macular edema (DME)?
The most clinically relevant OCT biomarkers in DME include:
- Intraretinal Fluid (IRF) — cystic fluid accumulation within retinal layers, strongly associated with active edema and vision impairment.
- Subretinal Fluid (SRF) — fluid beneath the neurosensory retina, often linked to inflammatory activity and treatment response.
- Hyperreflective Foci (HRF) — small reflective spots that may indicate inflammation, lipid extravasation, or retinal tissue damage.
- Disorganization of Retinal Inner Layers (DRIL) — disruption of inner retinal architecture associated with poorer visual outcomes.
- Ellipsoid Zone (EZ) Disruption — damage to photoreceptor integrity, often correlated with reduced visual acuity.
- Central Retinal Thickness (CRT) — a widely used quantitative metric for assessing edema severity and treatment response.
- Neurosensory Retina Atrophy — thinning and structural loss that may reflect chronic retinal damage and disease progression.
Monitoring these biomarkers over time helps clinicians evaluate disease activity, predict visual prognosis, assess therapeutic response, and optimize individualized treatment strategies. AI-powered OCT analysis further improves reproducibility and enables scalable, quantitative longitudinal monitoring.
2.1 OCT Features of Diabetic Macular Edema (DME)
There are several distinct OCT patterns associated with DME, including diffuse retinal thickening, cystoid macular edema (intraretinal cystic spaces), and serous retinal detachment (subretinal fluid).
1. Diffuse retinal thickening
This is characterized by a uniform increase in macular thickness, resulting from fluid accumulation in the extracellular space of the neurosensory retina due to disruption of the inner blood–retinal barrier and an imbalance between fluid leakage and the resorptive capacity of the retinal pigment epithelium (RPE).
When cystic spaces are present, their diameter must not exceed 50 μm; otherwise, the edema is classified as cystoid.
In chronic edema (lasting more than 6–9 months), irreversible photoreceptor damage may occur, along with the development of retinal atrophy.
Key features:
- may be the only finding in early stages
- requires careful quantitative assessment (central retinal thickness, CRT measurement)
- Macular thickness dynamics are an important biomarker of treatment efficacy
2. Cystoid macular edema (intraretinal cystic spaces)
This is a key structural marker of DME. On OCT, it appears as hyporeflective, round or oval cavities, predominantly located in the inner nuclear layer (INL) and the outer plexiform layer (OPL).
Clinical significance:
- reflects vascular hyperpermeability
- indicates active edema
- large and confluent cysts may suggest a chronic process and are associated with a worse functional prognosis
Long-standing cysts can lead to mechanical stretching of retinal tissue and secondary photoreceptor damage.
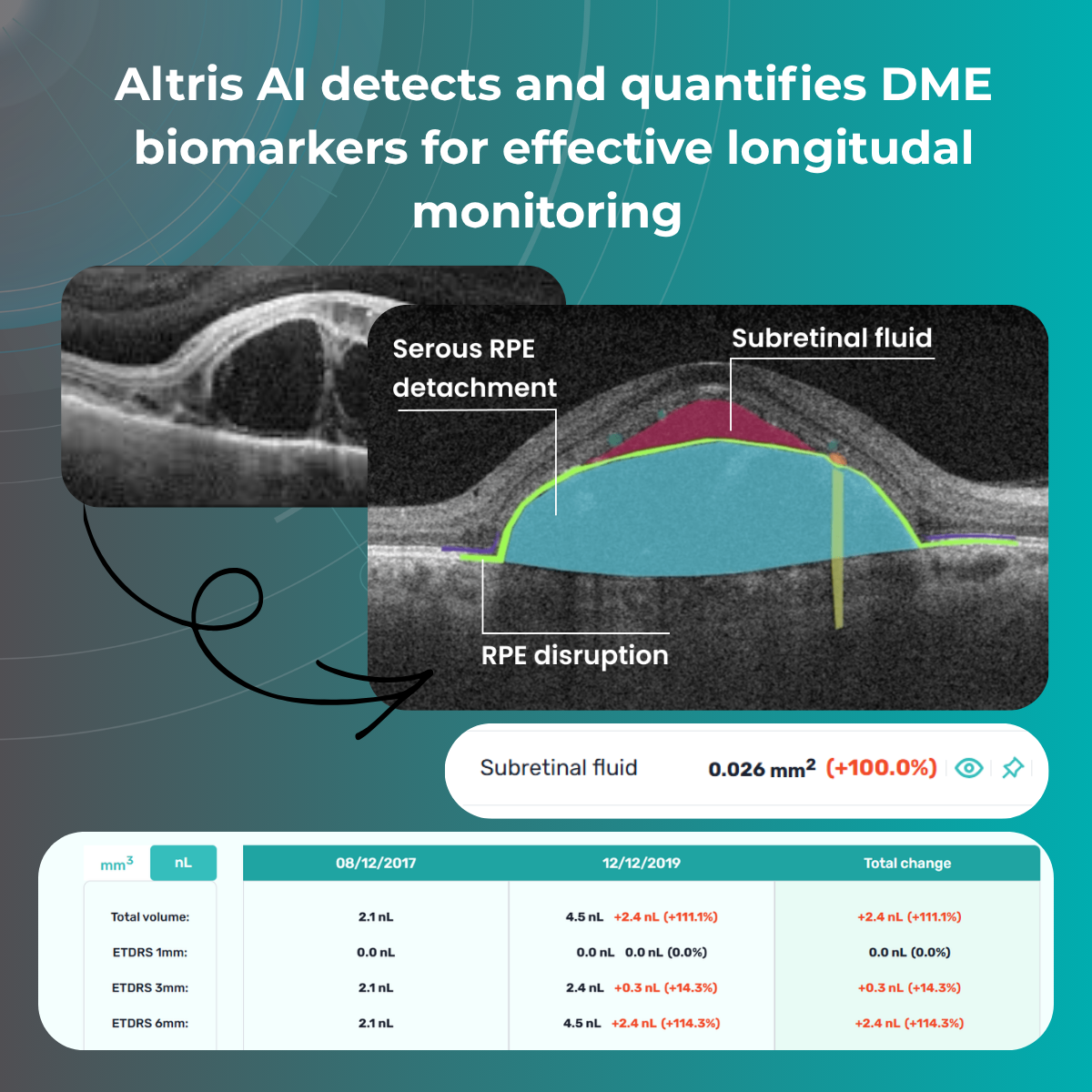
3. Serous retinal detachment (subretinal fluid)
Subretinal fluid refers to the accumulation of fluid between the neurosensory retina and the retinal pigment epithelium (RPE).
On OCT, it appears as a hyporeflective space above the RPE and is associated with neurosensory retinal detachment.
Clinical interpretation:
- may be a marker of active disease
- in some cases is associated with a better response to anti-VEGF therapy
- causes less photoreceptor damage than chronic intraretinal cysts
Although subretinal fluid is generally associated with a relatively better visual prognosis, its presence requires careful monitoring and should be considered when planning anti-VEGF treatment.
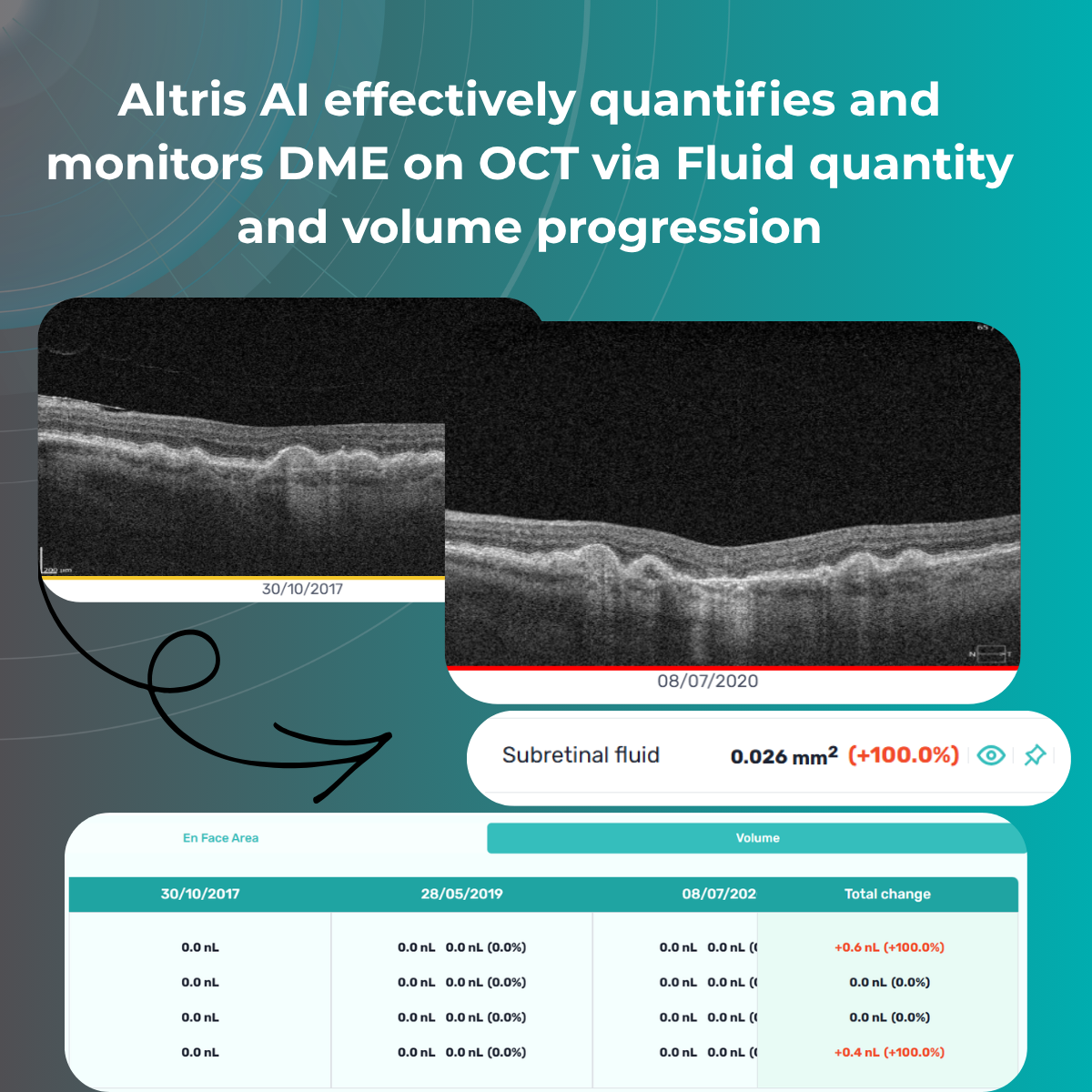
2.2 Quantitative parameters for monitoring DME on OCT
After morphological assessment, the next step is quantitative analysis. Currently, several key quantitative parameters can be obtained:
- Central retinal thickness (CRT) – the most widely used parameter
- Macular volume
- Fluid quantity and volume
These measurements enable precise monitoring of treatment response and help guide decisions regarding injection intervals.
3. Management of DME and patient education: key aspects of the modern approach
Modern management of diabetic macular edema (DME) is based on a comprehensive, personalized strategy in which OCT plays a central role. Today, therapeutic decisions are influenced by the morphological type of edema, disease activity, integrity of neurosensory retinal structures, individual patient characteristics, comorbidities, and prognostic biomarkers.
A key principle of the contemporary approach is the integration of structural OCT biomarkers in DME into clinical decision-making. These biomarkers not only help determine whether treatment is necessary, but also assist in selecting the optimal therapeutic modality, assessing response, and timely adjustment of management strategy.
Additional factors influencing therapy selection include:
- Presence of disorganization of the retinal inner layers (DRIL) and disruption of the ellipsoid zone (EZ)
- Response to previous treatments
- Systemic comorbidities (renal impairment, hypertension, adherence/compliance issues)
Treatment
Anti-VEGF therapy
Anti-VEGF agents (aflibercept, ranibizumab, bevacizumab) remain the first-line treatment for DME, as they directly target the key pathogenic mechanism—vascular hyperpermeability.
Newer agents with extended durability are emerging, including implantable drug delivery systems.
However, not all patients respond equally to anti-VEGF therapy. Therefore, OCT biomarker analysis is crucial: for example, a predominance of intraretinal cystic changes is usually associated with a good response to anti-VEGF, whereas a high number of hyperreflective foci or signs of chronic edema may indicate a significant inflammatory component and support consideration of steroid therapy.
Intravitreal corticosteroid implants
Steroids are used in cases of chronic and refractory DME, insufficient response to anti-VEGF therapy, and in patients with a pro-inflammatory phenotype.
Laser therapy
Although laser treatment has become less central in current practice, it remains useful in selected clinical scenarios. Subthreshold micropulse laser is more commonly used in patients with focal edema without involvement of the foveal center.
Thus, treatment decisions today are no longer universal; they are based on an individualized OCT-based patient profile.
Role of OCT in treatment
OCT accompanies the patient throughout all stages of treatment and performs several key functions:
- determination of indications for initiating therapy (presence of fluid, macular thickening, involvement of the foveal region)
- assessment of treatment response (reduction of fluid, normalization of thickness, structural restoration)
- detection of resistance or partial response
- optimization of injection intervals (treat-and-extend or pro re nata strategies)
A particularly important aspect is that OCT can detect subclinical changes. For example, minimal fluid accumulation may appear before any subjective deterioration in vision. This enables timely treatment adjustments and the prevention of functional loss.
In addition, OCT helps avoid both under- and overtreatment. In patients with a stable anatomical profile and no fluid, injection intervals can be gradually extended, reducing the burden on both the patient and the healthcare system.
Monitoring
The frequency of follow-up depends on disease stage and activity:
- active treatment phase – monthly visits with OCT control
- stabilization phase – every 2–4 months
- long-term follow-up – individualized, depending on recurrence risk and associated risk factors
It is important to emphasize that monitoring must remain regular even in the absence of symptoms. DME can progress asymptomatically, and only OCT allows objective assessment of retinal status.
Dynamic follow-up is critical: comparison of sequential scans provides the most valuable information about disease progression.
What is important to explain to the patient
Effective management of DME is not possible without active patient participation; therefore, communication is a key component of treatment.
The patient must clearly understand that:
- DME is a chronic condition requiring long-term monitoring
- treatment aims to stabilize and slow disease progression, not always to fully restore vision
- interruption of therapy without medical advice may lead to deterioration
- regular visits and OCT monitoring are critical, even if vision appears stable
It is especially important to explain the role of OCT to patients. Showing scans and explaining changes significantly improves treatment adherence.
Lifestyle and systemic control
Since diabetic macular edema (DME) is a complication of a systemic disease, control of the patient’s overall health is of critical importance.
Key recommendations include:
- optimal glycemic control
- blood pressure management
- correction of lipid profile
- healthy diet rich in antioxidants
- regular physical activity
- smoking cessation

Psychological aspects and treatment adherence
DME often follows a long and fluctuating course, which may lead to treatment fatigue or reduced motivation in patients. Many patients underestimate the severity of the condition, especially in early stages when visual acuity is still preserved.
In this context, OCT becomes not only a diagnostic tool but also a communication instrument. Visualization of pathological changes helps patients better understand the disease and the necessity of treatment.
Establishing a partnership between physician and patient is essential for successful long-term management.
Conclusion
OCT biomarkers in DME now allow not only precise diagnosis but also an approach that goes far beyond traditional retinal assessment. Thanks to its high resolution and ability to visualize microstructural changes, OCT enables the detection of subtle abnormalities before clinically significant symptoms appear. This opens the way to a new level of patient management—shifting from descriptive assessment to quantitative evaluation of pathological changes, their dynamics, and treatment response. Furthermore, OCT biomarker analysis allows prediction of disease course, identification of progression risk, and individualization of therapeutic strategies for each patient.
Modern DME management is not just diagnosis and treatment, but a comprehensive clinical decision-making system based on objective, standardized data. There is a clear shift from subjective interpretation of fundus changes to structured analytics, where every parameter matters: retinal thickness, presence of intra- or subretinal fluid, status of outer retinal layers, and macular architectural disruption. OCT has become the key tool transforming clinical practice, making it more precise, reproducible, and evidence-based. It allows clinicians not only to confirm the presence of pathology but also to better understand its nature, activity, and potential reversibility.
Ultimately, effective DME management today is the result of synergy between modern imaging technologies, clinical reasoning, and active patient engagement. Proper interpretation of OCT images must be integrated into the overall clinical picture, taking into account systemic factors, diabetes duration, and individual patient characteristics. In this process, OCT acts as a central link—a bridge between diagnosis and treatment—uniting all oct biomarkers in diabetic macular edema components into a coherent clinical system. This approach leads to better functional outcomes, preservation of vision, and significant long-term improvement in patients’ quality of life.
FAQ
1. How can AI help detect OCT biomarkers in DME?
AI can automatically identify key OCT biomarkers such as intraretinal fluid, subretinal fluid, hyperreflective foci, and retinal layer disruptions, helping clinicians detect disease activity faster and more consistently.
2. Why is quantitative biomarker analysis important in DME?
Quantification enables objective measurement of biomarker volume, area, and progression over time, supporting treatment decisions, therapy response assessment, and longitudinal patient monitoring.
3. Which OCT biomarkers are most relevant for monitoring DME progression?
Commonly monitored biomarkers include intraretinal fluid (IRF), subretinal fluid (SRF), hyperreflective foci (HRF), disorganization of retinal inner layers (DRIL), and ellipsoid zone disruption.
4. How can automated OCT analysis improve clinical workflow?
Automated analysis reduces manual interpretation time, improves reproducibility, standardizes reporting, and helps clinicians prioritize patients who may require closer follow-up or treatment adjustments.
References
- https://pubmed.ncbi.nlm.nih.gov/38460657/
- https://brief.euretina.org/research/association-of-retinal-oct-biomarkers-with-reading-performance-in-patients-with-diabetic-macular-edema-dme
- https://www.mdpi.com/2075-4418/14/1/76
- https://www.sciencedirect.com/science/article/pii/S1572100024000814
- https://link.springer.com/article/10.1186/s40942-023-00473-w
- https://www.medscape.com/viewarticle/1001580
- https://www.cureus.com/articles/227801-innovations-in-diabetic-macular-edema-management-a-comprehensive-review-of-automated-quantification-and-anti-vascular-endothelial-growth-factor-intervention#!/
