Recently Posted
-

PBM Monitoring on OCT: Drusen Progression
 Maria Znamenska
5 min.
Maria Znamenska
5 min.Introduction: Role of PBM in Retinal Disease Management
Photobiomodulation (PBM), also referred to as low-level light or laser therapy, has emerged as a promising non-invasive therapeutic strategy in ophthalmology, particularly for the management of retinal diseases. PBM utilizes low-energy light in the red-to-near-infrared spectrum (typically 600–1000 nm) to modulate cellular function through photochemical rather than thermal mechanisms.
At the cellular level, PBM primarily absorbs photons from mitochondrial chromophores, most notably cytochrome c oxidase, thereby enhancing mitochondrial respiration and increasing adenosine triphosphate (ATP) production. This process is accompanied by modulation of reactive oxygen species (ROS) and activation of intracellular signaling pathways, collectively promoting cellular survival, repair, and regeneration. Importantly, PBM has also been shown to reduce oxidative stress and inflammation—two central mechanisms implicated in the pathogenesis of many retinal disorders—by decreasing pro-inflammatory cytokines and enhancing antioxidant defenses.
The retina, characterized by high metabolic demand and susceptibility to oxidative damage, represents a particularly suitable target for PBM therapy. Experimental and clinical studies have demonstrated that PBM can improve photoreceptor survival, support retinal pigment epithelium (RPE) function, and enhance overall retinal homeostasis. In animal models, PBM has been associated with preservation of retinal structure and improved electrophysiological responses, while early clinical studies suggest potential benefits for visual acuity, contrast sensitivity, and reductions in pathological features such as drusen and retinal edema.
Clinically, PBM is being explored as an adjunctive or alternative treatment for several retinal diseases, including age-related macular degeneration (AMD), diabetic retinopathy (DR), and diabetic macular edema (DME). Its advantages include a favorable safety profile, non-invasive delivery, and relatively low cost compared to conventional therapies. Furthermore, advances in light-emitting diode (LED) technology have facilitated broader clinical application by enabling safe, uniform, and cost-effective retinal illumination.
Key OCT Biomarkers to Track During PBM Therapy
Optical coherence tomography (OCT) has become an indispensable tool for monitoring retinal structure and treatment response in patients undergoing photobiomodulation (PBM) therapy. Given the mechanism of PBM—targeting mitochondrial function, reducing oxidative stress, and modulating inflammation—several OCT-derived biomarkers are particularly relevant for assessing therapeutic efficacy of dry AMD progression and in other retinal disorders. Track and quantify the following biomarkers with Altris AI:
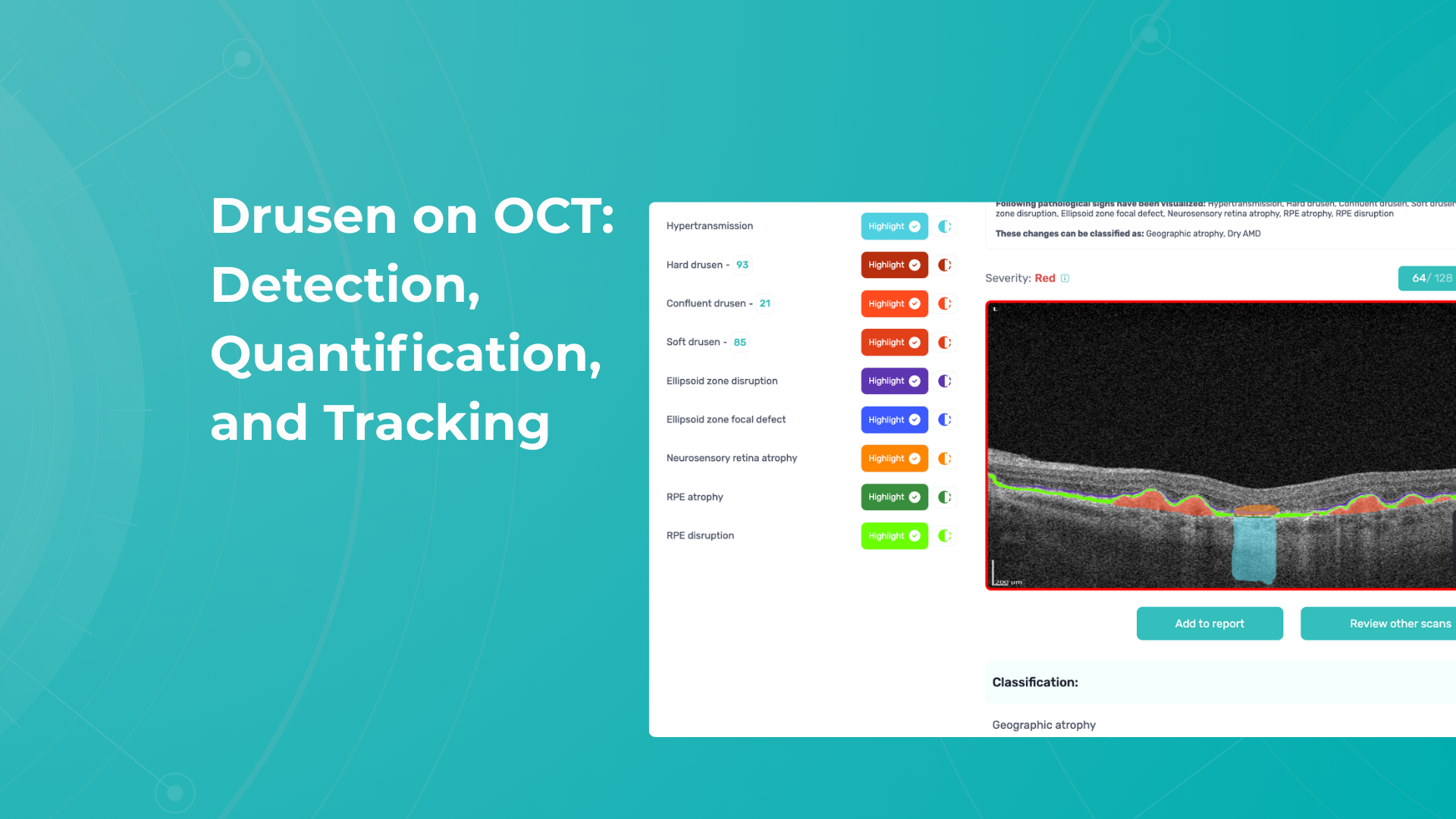
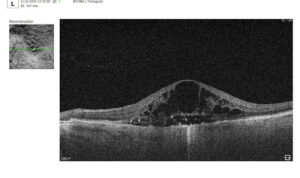
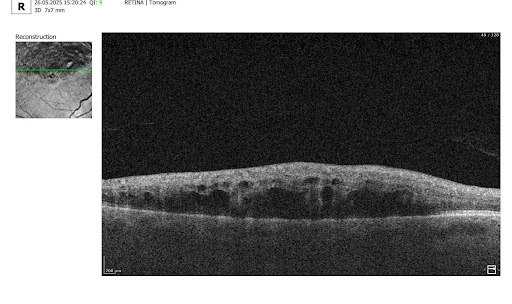
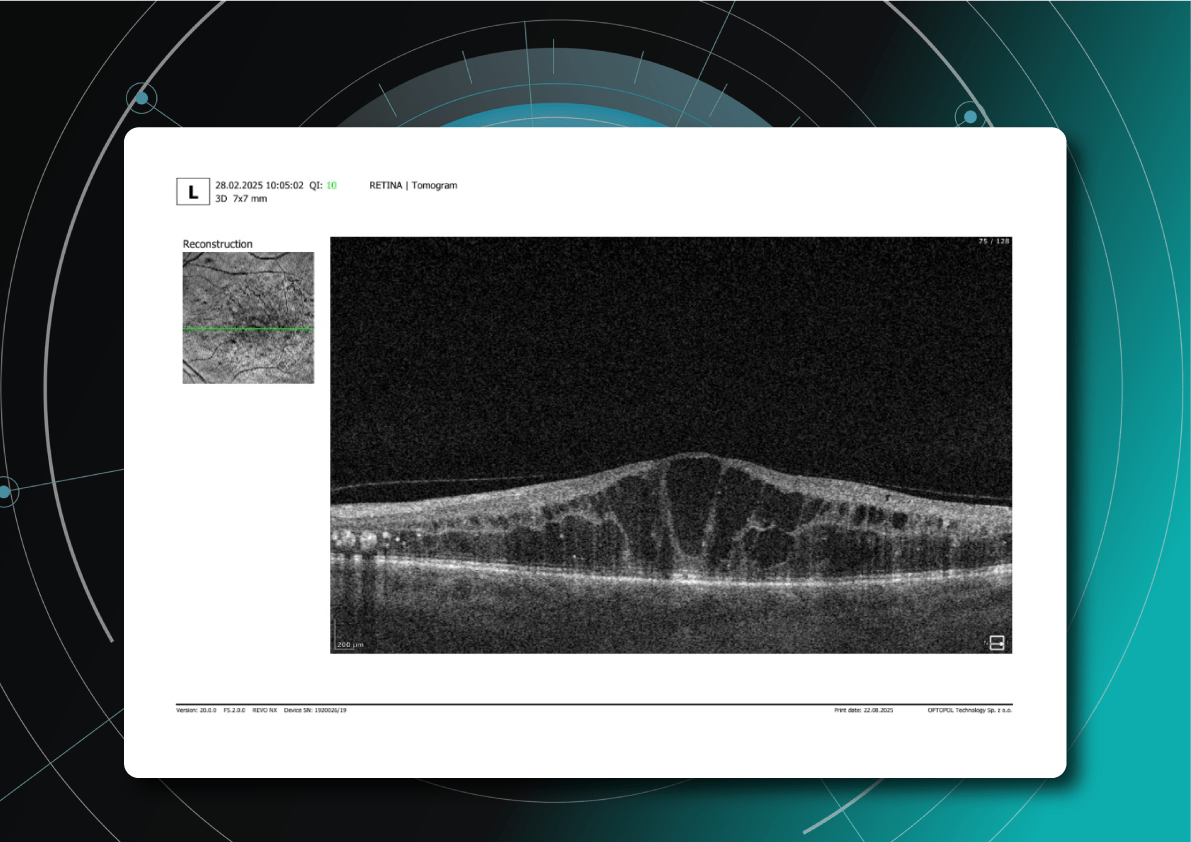
1.PBM monitoring on OCT: Drusen Progression
For patients on photobiomodulation (PBM), OCT monitoring of drusen is about one core question: Are we stabilizing or reversing RPE–Bruch’s membrane dysfunction, or is the eye still progressing toward atrophy? So, on B-scans, drusen are seen as:
- RPE elevations (dome-shaped or irregular)
- Material between RPE and Bruch’s membrane
- Variable internal reflectivity
Here are the key drusen biomarkers to track under PBM:
- Drusen volume (MOST important)
- Measured via OCT segmentation (cube scans)
- Represents total disease burden
As the PBM goal here is the stabilization or reduction in drusen volume
Red flag:
- Continuous increase → disease progression
- Drusen height & area
- Local structural impact on photoreceptors
PBM signal:
- Flattening = potential response
- Increasing height = worsening RPE dysfunction
- Internal reflectivity
- Homogeneous vs heterogeneous content
An important nuance is that increasing heterogeneity may indicate:
- calcification
- regression OR collapse before atrophy
So, it needs to be correlated with other signs.
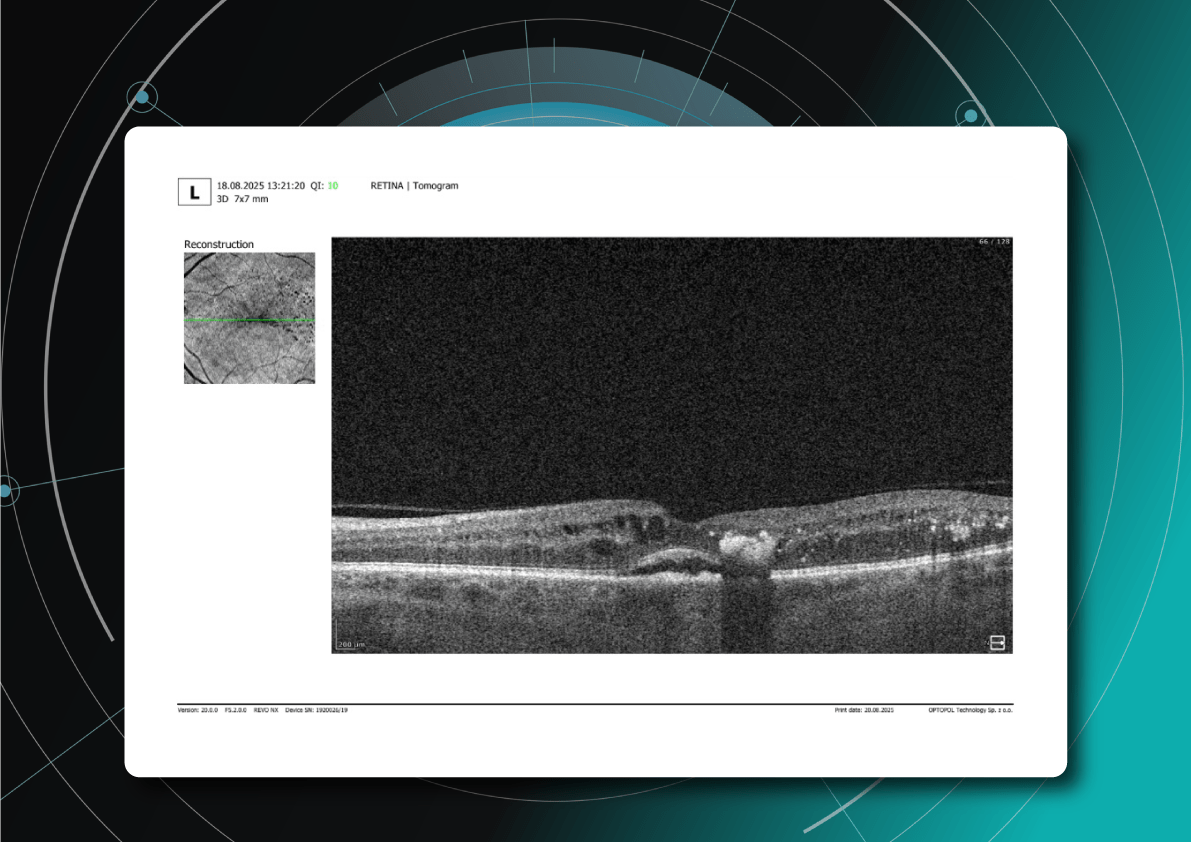
You may observe Drusen regression patterns. However, not all regression is good.

“Good” regression:
- Gradual flattening
- No photoreceptor loss
- Stable RPE
“Bad” regression (collapse):
- Sudden disappearance
- Followed by:
- RPE loss
- outer retinal thinning
Leads to geographic atrophy (GA)
To summarise, for PBM-treated patients, prioritize:
- Drusen volume trend (longitudinal)
- Photoreceptor integrity (EZ/ONL)
- Signs of atrophy risk (HRF, collapse patterns)
Dry AMD progression matters in PBM monitoring. PBM is currently aimed at:
- early → intermediate dry AMD
So when you monitor drusen on OCT, you’re not just tracking morphology — you’re tracking disease trajectory: Is the eye staying in intermediate AMD, or moving toward advanced stages (GA / nAMD)?
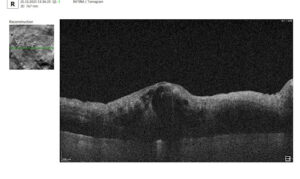
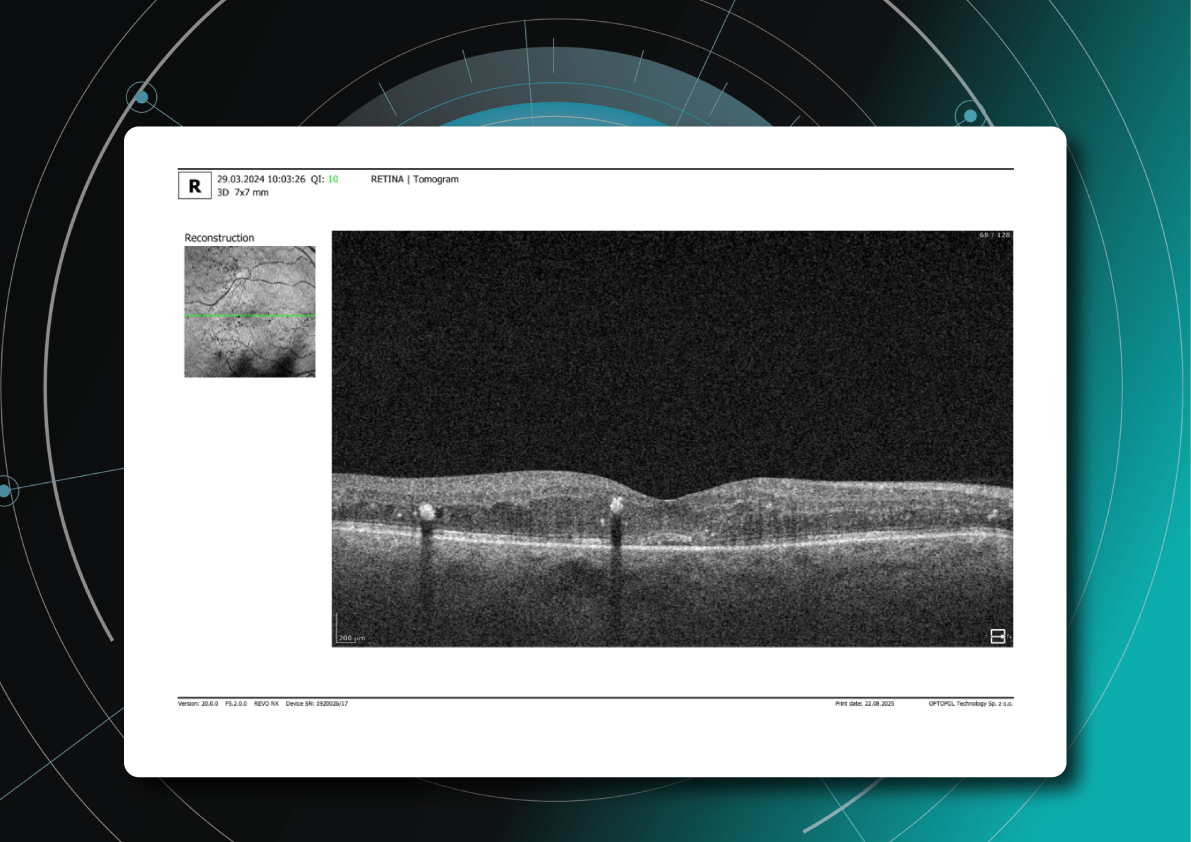
2. Central retinal thickness (CRT)
In addition to PBM monitoring on OCT: drusen progression, one of the primary biomarkers is also central retinal thickness (CRT), which reflects changes in retinal edema and overall retinal integrity. Reductions in CRT during PBM therapy may indicate decreased inflammatory activity and improved fluid homeostasis, particularly in conditions such as diabetic macular edema (DME) and neovascular retinal diseases. However, in non-exudative conditions such as dry age-related macular degeneration (AMD), CRT changes may be subtle, necessitating the evaluation of additional structural parameters.
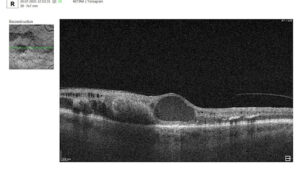
3. The outer retinal layers
The outer retinal layers, especially the integrity of the ellipsoid zone (EZ) and external limiting membrane (ELM), represent critical biomarkers of photoreceptor health. PBM has been associated with improved mitochondrial activity within photoreceptors, and preservation or restoration of EZ continuity on OCT may serve as a surrogate marker of functional recovery. Disruptions in these layers are strongly correlated with visual impairment, making them highly relevant endpoints in PBM studies.
4. Retinal pigment epithelium (RPE)
Another key biomarker is retinal pigment epithelium (RPE) morphology, including the presence and evolution of drusen, subretinal drusenoid deposits (SDD), and RPE irregularities. PBM has been hypothesized to enhance RPE function and reduce oxidative burden, potentially leading to stabilization or regression of drusen volume over time. Quantitative drusen analysis using OCT can therefore provide insight into disease modification, particularly in intermediate AMD.
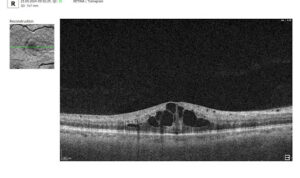
5. Hyperreflective foci (HRF)
Hyperreflective foci (HRF) are also important indicators of retinal inflammation and microglial activation. A reduction in HRF number or density during PBM therapy may reflect decreased inflammatory signaling, aligning with the known anti-inflammatory effects of light-based treatment. Similarly, subretinal and intraretinal fluid—when present—should be carefully monitored, as their resolution may indicate improved retinal barrier function and treatment response.
6. Other
Emerging OCT biomarkers include choroidal thickness and choriocapillaris integrity, which are increasingly recognized as contributors to the pathophysiology of retinal disease. PBM may influence choroidal perfusion and metabolic exchange, and changes in choroidal thickness could serve as an indirect marker of vascular and metabolic improvements.
PBM Treatment Monitoring Protocol (Step-by-Step)
1. Patient Selection and Baseline Assessment
Appropriate patient selection is critical to optimizing outcomes of photobiomodulation (PBM) therapy in dry AMD progression. Current evidence supports its use primarily in non-exudative retinal diseases, particularly intermediate Age-related Macular Degeneration, as well as emerging applications in Diabetic Retinopathy and Diabetic Macular Edema.
Inclusion considerations:
- Intermediate AMD (presence of drusen and/or subretinal drusenoid deposits)
- Stable retinal conditions without active neovascularization
- Best-corrected visual acuity (BCVA) is sufficient for functional monitoring
Exclusion criteria:
- Active neovascular AMD or significant intraretinal/subretinal fluid
- Recent anti-VEGF injections (unless PBM is used adjunctively in controlled settings)
- Significant media opacity affecting light delivery or imaging quality
Baseline evaluation should include:
- Visual function testing: BCVA, contrast sensitivity
- Structural imaging: spectral-domain OCT (mandatory)
- Optional advanced imaging: OCT angiography (OCTA) for vascular assessment
- Key OCT biomarkers (baseline reference):
-
- Central retinal thickness (CRT)
- Ellipsoid zone (EZ) integrity
- Retinal pigment epithelium (RPE) status and drusen volume
- Presence of hyperreflective foci (HRF)
Establishing a robust baseline is essential, as PBM-induced changes are often gradual and require longitudinal comparison.
2. Treatment Session Procedure
PBM is delivered using low-level light in the red-to-near-infrared spectrum (typically ~600–1000 nm), most commonly via LED-based systems designed for retinal applications.
Standard session workflow:
- Patient preparation
- No pharmacologic dilation is typically required (device-dependent)
- Proper alignment and fixation ensured
- Device application
- Light delivered trans-pupillary using controlled, non-thermal energy
- Multi-wavelength protocols (e.g., combinations of ~590 nm, 660 nm, 850 nm) are commonly used in clinical studies
- Treatment duration
- Typically, a few minutes per eye per session (device-specific)
- Sequential or simultaneous bilateral treatment, depending on the system
- Safety monitoring
- PBM is non-invasive and well-tolerated
- No significant adverse retinal effects have been reported in the current literature
- Monitor for discomfort or visual disturbances (rare)
Immediate post-session:
- No recovery time required
- Patients resume normal activities immediately
The mechanism of action—enhancing mitochondrial activity and reducing oxidative stress—does not produce immediate anatomical changes, reinforcing the need for structured follow-up.
3. Treatment Series and Frequency
PBM is not a single-session therapy for observing dry AMD progression or any other condition, but is administered as a treatment series, followed by monitoring and potential retreatment cycles.
Typical treatment regimen (based on clinical studies):
- Induction phase:
- 2–3 sessions per week
- Duration: 3–5 weeks
- Total: ~9–12 sessions per cycle
- Follow-up period:
- Reassessment at 1–3 months post-treatment
- OCT imaging to evaluate structural response
- Retreatment strategy:
- Repeat cycles every 4–6 months, depending on disease progression and response
- Individualized based on OCT biomarkers and functional outcomes
Monitoring during and after therapy:
- Short-term (during treatment):
- Limited structural change expected
- Intermediate-term (1–3 months):
- Possible reduction in drusen volume
- Stabilization of EZ and RPE integrity
- Decrease in HRF (inflammatory markers)
- Long-term:
- Disease stabilization rather than reversal is the primary goal
Outcome measures:
- Functional: BCVA, contrast sensitivity
- Structural: OCT biomarkers (drusen, EZ, CRT, HRF)
- Optional: OCTA vascular parameters
A structured PBM protocol integrates careful patient selection, standardized treatment delivery, and longitudinal OCT-based monitoring. The therapy is best suited for chronic, non-exudative retinal conditions, where its cumulative biological effects—rather than immediate anatomical changes—drive clinical benefit. Consistent imaging and biomarker tracking are essential for guiding retreatment decisions and evaluating long-term efficacy.
Devices & AI for PBM Monitoring
LumiThera (Valeda)
Valeda is a photobiomodulation (PBM) device — currently the most clinically validated retinal light therapy. Company: LumiThera (now acquired by Alcon) Mechanism (PBM):
- Uses multiwavelength light:
- ~590 nm
- ~660 nm
- ~850 nm
- Targets mitochondria → increases ATP
- Reduces:
- oxidative stress
- inflammation
- Improves retinal cellular function
Clinical positioning
- Indicated for:
- early–intermediate dry AMD
- First device to:
- demonstrate visual improvement, not just slowing the disease
Regulatory & evidence strength:
- FDA-authorized (De Novo, 2024)
- CE marked, global use
- Backed by:
- LIGHTSITE trials (I–III)
- Demonstrated:
- ~1 line BCVA improvement over 24 months
Treatment protocol
- Standard cycle:
- 9 sessions over 3–5 weeks
- Repeat:
- every 4–6 months
- Fully clinic-based
Clinical effects
- Functional:
- ↑ visual acuity
- ↑ contrast sensitivity
- Structural (OCT):
- drusen stabilization/reduction
- preservation of retinal layers
- Strong safety profile
Thus, Valeda is currently:
The only FDA-authorized retinal PBM device
The only one with phase III randomized clinical trial data (LIGHTSITE)
And it has shown actual visual acuity improvement, not just safety.Espansione
This is an ophthalmology PBM platform (incl. retina use). Espansione develops photobiomodulation (PBM / LLLT) devices specifically for eye care. Espansione is a multi-technology ophthalmic platform, not just a PBM lamp.
It combines:
- LM™ LLLT (photobiomodulation / PBM) → core tech for retina
- OPE™ IPL → only for anterior segment
Their flagship eye-light® is used for:
- anterior segment (DED, MGD)
- posterior segment (including dry AMD progression)
- It has CE approval for retinal conditions like dry AMD
- The company explicitly states expansion into posterior segment care (retina)
This is another ophthalmic PBM device platform, with retina applications.
Light characteristics
- Wavelength range: ~500–1000 nm
- Typical retina protocol:
- Yellow (~590 nm)
- Red (~625 nm)
- Delivered via LED mask over closed eyes
Mechanism (important for retina positioning)
PBM targets mitochondria:
- ↑ ATP production
- ↓ oxidative stress (ROS)
- ↑ cellular repair
- ↓ inflammation
For retina specifically:
-
Improves RPE + photoreceptor metabolism
-
Supports choriocapillaris perfusion
-
Slows degeneration processes (AMD biology)
Retina applications
- Dry AMD progression (main indication)
- CE (MDR) cleared for dry AMD (dAMD)
- Focus: early–intermediate stages (AREDS 1–3)
Clinical effects shown:
- ↑ Visual acuity (BCVA gains)
- ↓ Drusen volume
- ↑ Choriocapillaris perfusion
Thus, it is disease-modifying intent, not just symptomatic.
Typical session:
- Duration: ~10–12 minutes
- Mask-based (non-contact)
- Eyes:
- mostly closed
- brief opening phases possible
Typical course:
- ~8 sessions / 3–4 weeks (in studies)
Very different from laser or injections:
- No dilation
- No contact lens
- No invasive steps
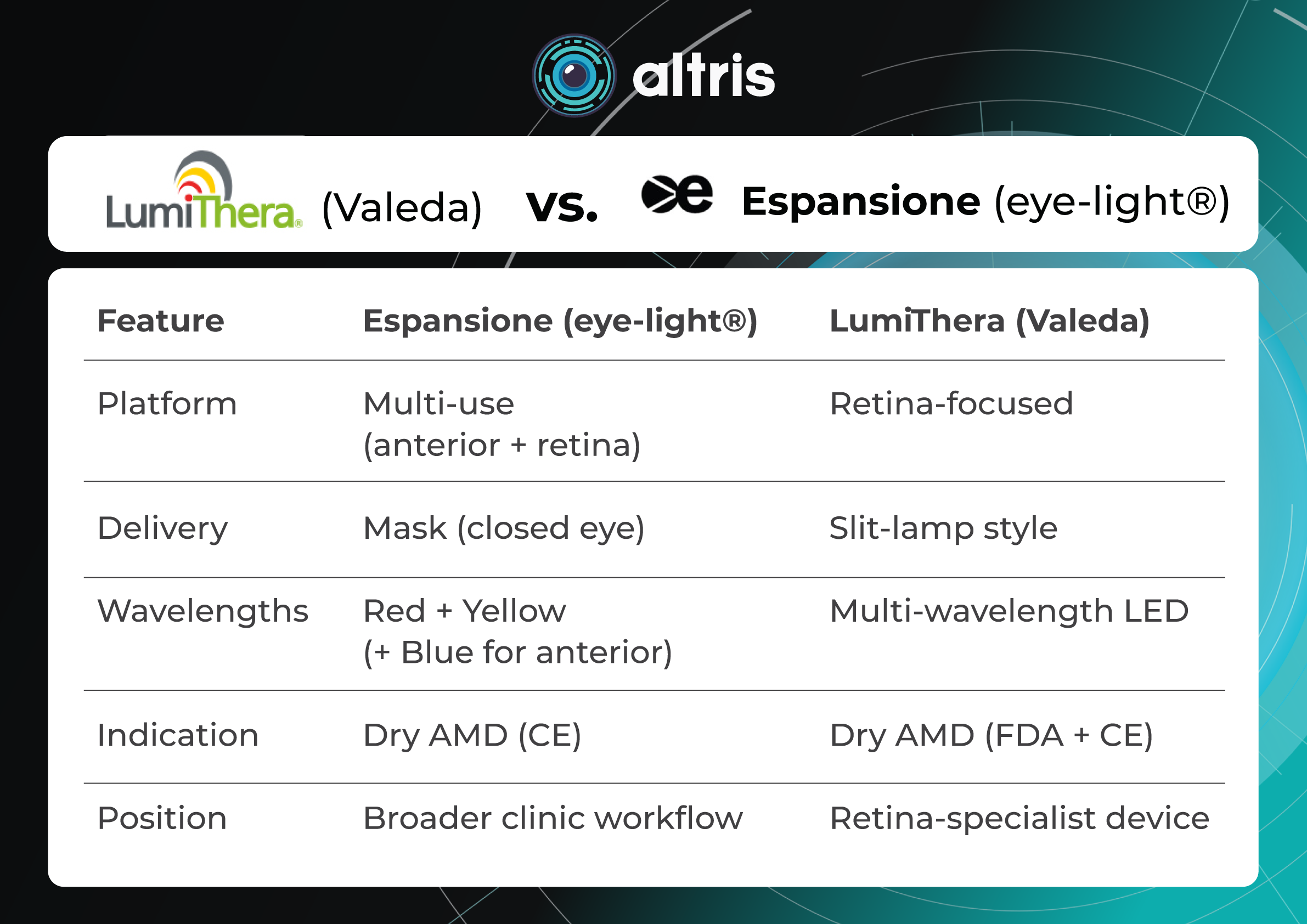
Here’s a comparison chart of the key characteristics and features of both devices:

LumiThera (Valeda) vs. Espansione (eye-light®)
Feature Espansione (eye-light®) LumiThera (Valeda) Platform Multi-use (anterior + retina) Retina-focused Delivery Mask (closed eye) Slit-lamp style Wavelengths Red + Yellow (+ Blue for anterior) Multi-wavelength LED Indication Dry AMD (CE) Dry AMD (FDA + CE) Position Broader clinic workflow Retina-specialist device Other Generic LED / Low-Level Light Therapy (LLLT) platforms
These are NOT branded retinal systems like Valeda or Espansione, but rather:
- LED arrays or laser-based PBM setups
- used in:
- early trials
- pilot studies
- lab settings
Typical characteristics:
- Wavelengths: 500–1000 nm
- Delivery:
- masks
- panels
- slit-lamp–mounted systems
Examples (non-standardized):
- custom LED rigs used in AMD studies
- early PBM prototypes before Valeda
- academic research devices
Problem:
- No standard protocol
- No regulatory approval
- Hard to compare outcomes
Experimental PBM Directions also include expanded PBM indications of
- diabetic retinopathy (DR)
- diabetic macular edema (DME)
- geographic atrophy
Next-gen PBM systems
Nevertheless, research in eye care is further exploring:
- different wavelength combinations
- dose optimization
- portable/home PBM (not validated yet)
Despite the fact that nothing close to Valeda-level evidence has emerged yet.
Modern PBM ecosystem classification
Tier 1 (validated):
- Valeda (LumiThera / now Alcon)
Tier 2 (investigational retinal PBM):
- academic LED systems
- prototype devices
Tier 3 (not true retinal PBM):
- dry eye LLLT
- cosmetic red light
- wellness devices
Clinical Application and Results
The integration of digital technologies and artificial intelligence (AI) into retinal imaging has significantly enhanced the ability to monitor treatment response in photobiomodulation (PBM) therapy. Given that PBM induces gradual, often subtle structural and functional changes, advanced analytical tools are essential for detecting and quantifying these effects with precision and reproducibility. Here are some real cases of application in Ophthalmology (Dry AMD, DME, etc).
Dry AMD
Photobiomodulation (PBM) has been clinically evaluated primarily in patients with early-to-intermediate Age-related Macular Degeneration, where no widely accepted disease-modifying therapy exists. The most robust evidence comes from the LIGHTSITE clinical trial program, in which PBM is delivered as multiwavelength light therapy (590, 660, and 850 nm) in repeated treatment cycles (typically 9 sessions over 3–5 weeks, repeated every 4 months).
Across the LIGHTSITE I–III studies, PBM has consistently demonstrated functional improvements, particularly in best-corrected visual acuity (BCVA) and contrast sensitivity, and has shown favorable safety outcomes, with no evidence of phototoxicity. In LIGHTSITE II, PBM-treated eyes showed a mean ~4-letter gain in BCVA at 9 months, with approximately one-third of patients achieving ≥5-letter improvement, while sham-treated eyes showed minimal change. Earlier studies also reported improvements in contrast sensitivity, microperimetry, and reductions in drusen burden, suggesting both functional and anatomical benefits .
More recent data from the pivotal LIGHTSITE III trial further support these findings, demonstrating statistically significant gains in visual acuity compared with sham treatment, with mean improvements exceeding 5 letters and a substantial proportion of patients achieving clinically meaningful gains. At 24 months, PBM-treated eyes showed sustained visual improvement (+6.2 letters) and a reduced progression to geographic atrophy (6.8% vs 24.0% in controls), suggesting potential disease-modifying effects.
However, despite these encouraging results, meta-analyses indicate that overall effect sizes remain modest and that variability across studies, small sample sizes, and protocol heterogeneity limit definitive conclusions regarding long-term clinical benefit. Thus, while PBM represents a promising and biologically plausible therapy for dry AMD, its role in routine clinical practice continues to evolve, with ongoing studies needed to confirm durability, optimal patient selection, and real-world effectiveness.
DME
Photobiomodulation (PBM) has been explored as a non-invasive adjunctive or alternative therapy for Diabetic Macular Edema, targeting key pathogenic mechanisms, including mitochondrial dysfunction, oxidative stress, and chronic inflammation.
Unlike anti-VEGF therapy, which primarily addresses vascular permeability, PBM aims to restore retinal metabolic balance through light-induced activation of mitochondrial pathways. Early clinical studies using red-to-near-infrared wavelengths (typically ~630–850 nm) have demonstrated reductions in central retinal thickness (CRT) and improvements in retinal morphology on OCT, alongside stabilization or modest gains in best-corrected visual acuity (BCVA). These effects are particularly notable in mild-to-moderate DME and in patients with non-center-involving edema, where PBM may reduce inflammatory signaling and improve fluid homeostasis.
Clinical data, although still limited compared to age-related macular degeneration, suggest that PBM may have value as an adjunct to standard of care, potentially reducing treatment burden in patients requiring repeated intravitreal injections. Some studies report decreased intraretinal fluid and improvement in OCT biomarkers following PBM treatment cycles, with a favorable safety profile and no evidence of retinal damage.
However, results remain heterogeneous, with variability in treatment protocols, patient populations, and outcome measures. Importantly, PBM has not yet demonstrated efficacy comparable to anti-VEGF therapy in center-involving DME, and its role is best considered complementary rather than substitutive at this stage. Larger randomized controlled trials are needed to define optimal dosing strategies, identify responder phenotypes, and clarify long-term functional benefits in DME management.
Why is OCT Critical for PBM Monitoring?
Optical Coherence Tomography (OCT) is essential for monitoring photobiomodulation (PBM) therapy because PBM aims to induce subtle, progressive structural and functional changes in the retina—especially in conditions like dry AMD progression.
Unlike anti-VEGF treatments, where effects can be more immediate, PBM outcomes are gradual and microstructural, such as changes in drusen volume, RPE integrity, outer retinal layers, and choriocapillaris perfusion. These changes are often invisible on fundus photography or visual acuity alone, making OCT the only practical tool for objective, layer-by-layer tracking over time.
Serial OCT scans allow clinicians to detect early signals of response (e.g., drusen regression or stabilization) and differentiate them from natural disease progression, which is critical for validating PBM efficacy in real-world practice.
AI-Assisted OCT Analysis
Artificial intelligence–driven analysis of optical coherence tomography (OCT) enables automated, quantitative assessment of retinal biomarkers critical to PBM monitoring. Machine learning and deep learning algorithms can segment retinal layers and identify pathological features such as drusen, hyperreflective foci (HRF), and fluid compartments with high accuracy.
In the context of PBM, AI tools provide:
- Automated retinal layer segmentation, including ellipsoid zone (EZ) and retinal pigment epithelium (RPE)
- Quantification of drusen volume and distribution, particularly relevant in Age-related Macular Degeneration
- Detection and tracking of subtle structural changes over time that may not be apparent on qualitative review
These capabilities are especially important because PBM effects are often incremental rather than dramatic, requiring sensitive longitudinal comparison.
Longitudinal Tracking and Progression Analysis
Digital platforms enable time-series analysis of OCT data, allowing clinicians to monitor the disease trajectory before, during, and after PBM therapy. Automated registration of sequential scans ensures that the same retinal locations are compared over time.
Key advantages include:
- Change detection algorithms for early identification of treatment response
- Trend analysis of biomarkers such as central retinal thickness, EZ integrity, and HRF density
- Objective progression metrics, reducing inter-observer variability
Such tools are critical for distinguishing true therapeutic effects from natural fluctuations in disease, particularly in slowly progressing conditions like dry AMD progression.
Digital tools and AI are transforming PBM monitoring by enabling precise, quantitative, and longitudinal assessment of retinal biomarkers. From automated OCT analysis to AI-driven decision support and remote monitoring, these technologies address the key challenge of PBM therapy—detecting subtle, progressive changes over time, like in dry AMD progression, etc. Their continued development will be essential for standardizing PBM protocols and optimizing patient outcomes in retinal disease management.
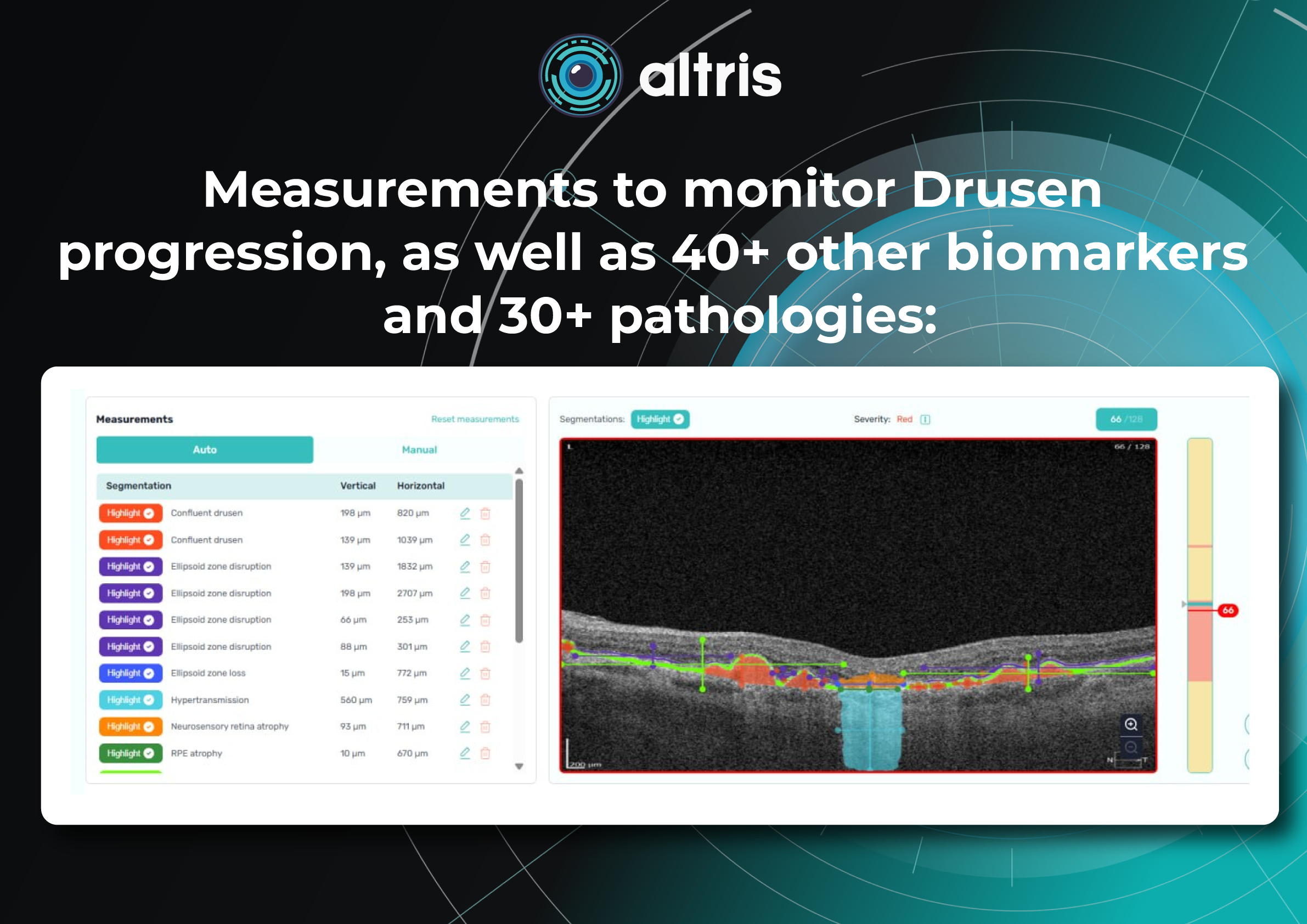
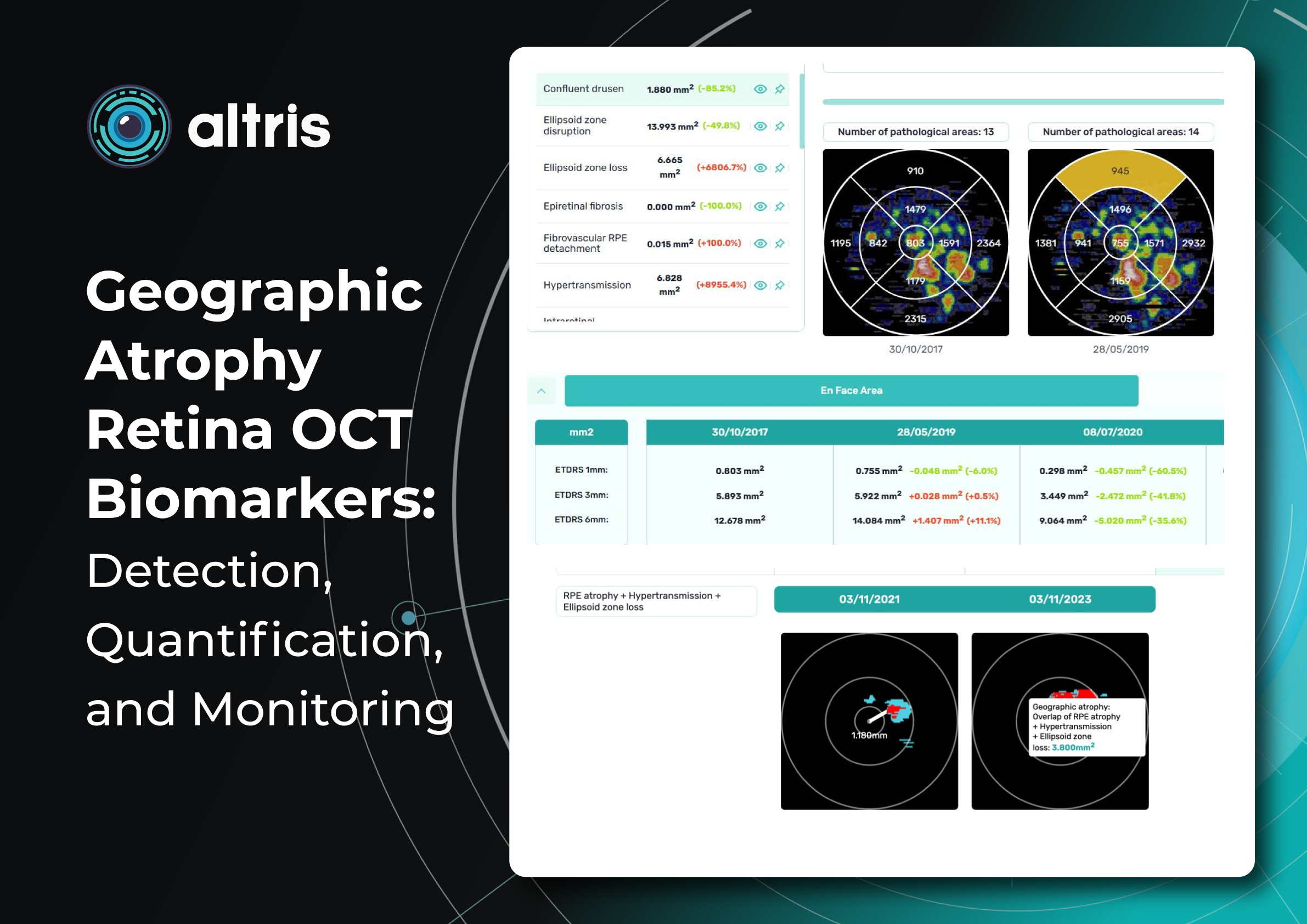
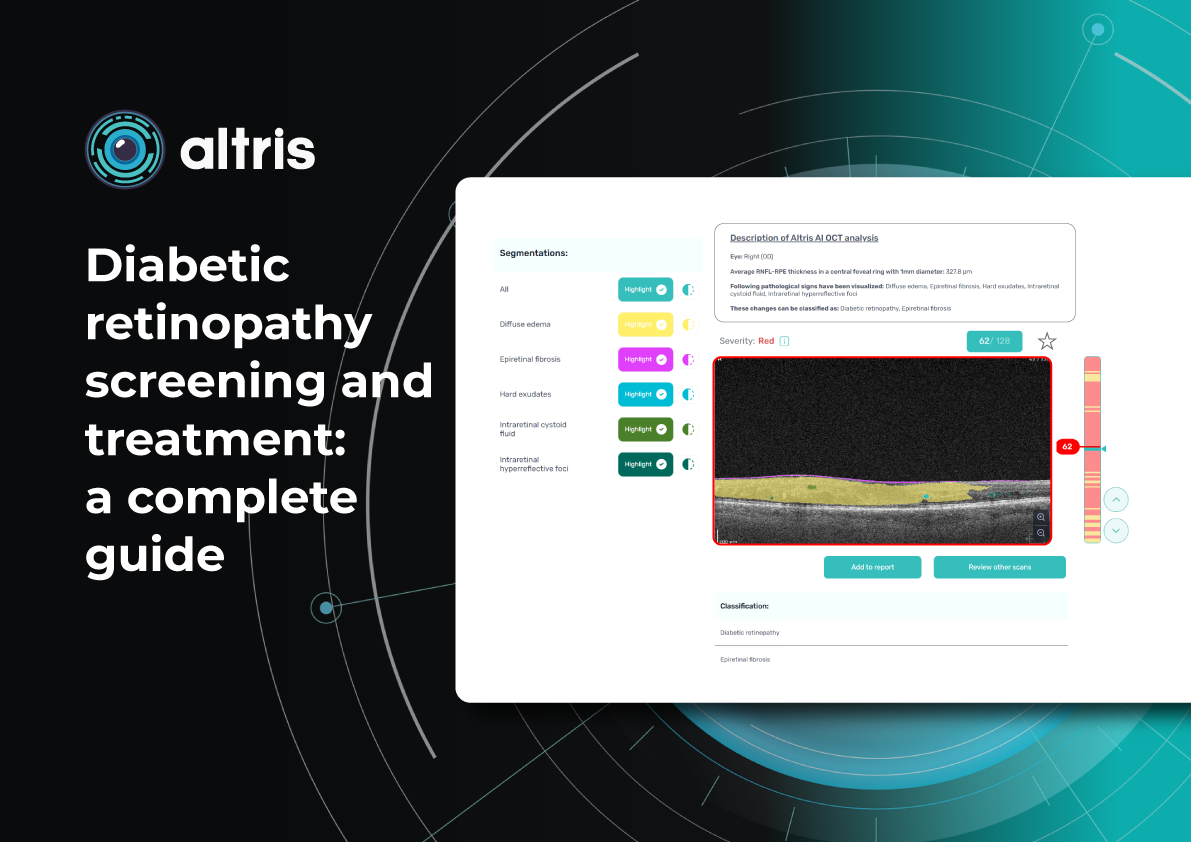
Altris AI for PBM monitoring on OCT: Drusen Progression +40 biomarkers
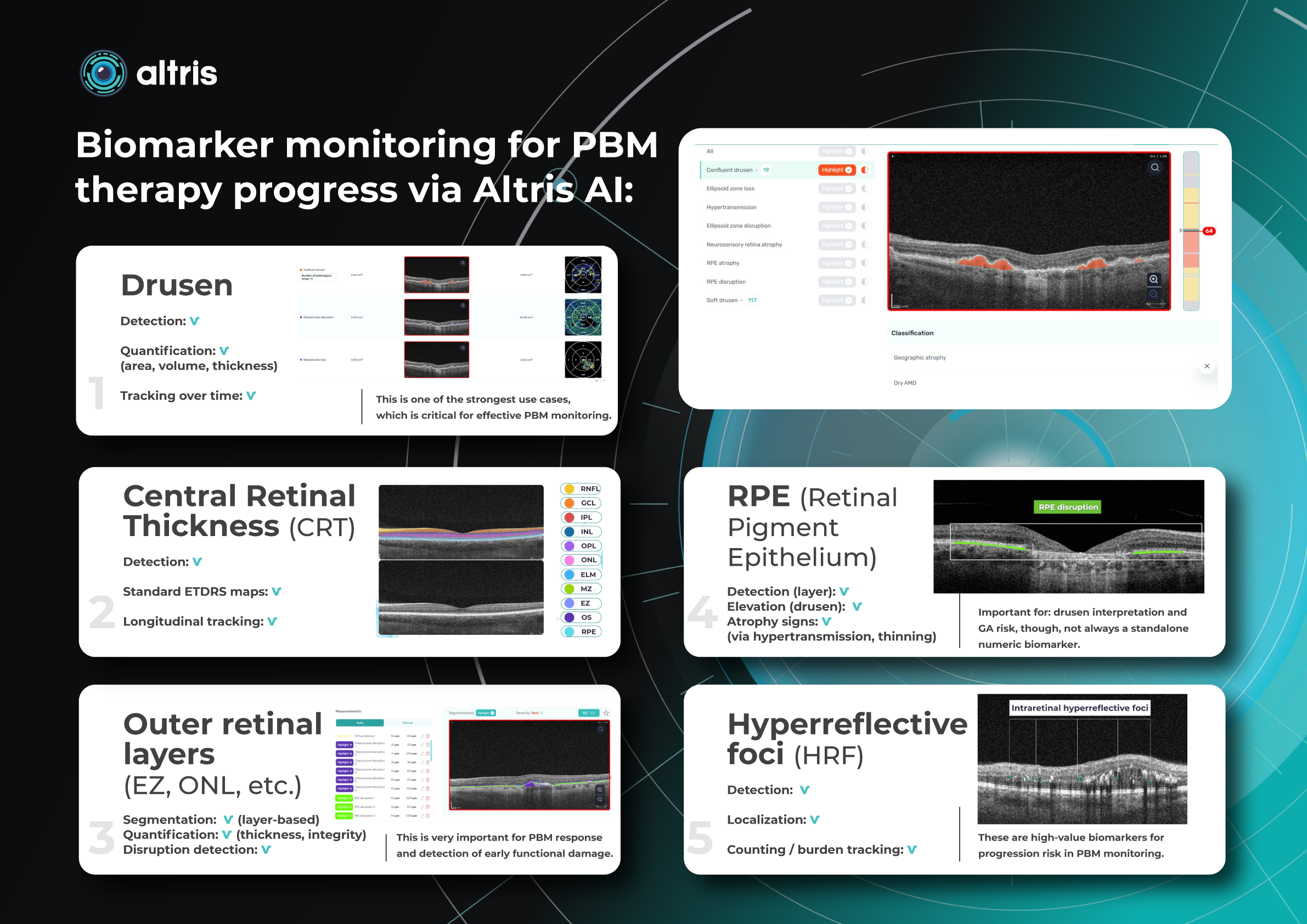
Altris AI has contributed to PBM monitoring on OCT: Drusen progression, as well as 40+ other biomarkers and 30+ pathologies, which may be monitored with the system. Altris AI enhances this process by turning OCT into a quantitative, standardized monitoring system rather than a subjective review. It automatically segments retinal layers and biomarkers (e.g., drusen, hyperreflective foci, fluid), calculates precise volumetric metrics, and enables longitudinal comparison across visits with high reproducibility. Here are the examples of biomarker monitoring cases via Altris AI:

- Drusen
Detection: ✔️
Quantification: ✔️ (area, volume, thickness)
Tracking over time: ✔️
This is one of the strongest use cases, which is critical for effective PBM monitoring.
- Central Retinal Thickness (CRT)
Detection: ✔️
Standard ETDRS maps: ✔️
Longitudinal tracking: ✔️
- Outer retinal layers (EZ, ONL, etc.)
Segmentation: ✔️ (layer-based)
Quantification: ✔️ (thickness, integrity)
Disruption detection: ✔️
This is very important for PBM response and detection of early functional damage.
- RPE (Retinal Pigment Epithelium)
Detection (layer): ✔️
Elevation (drusen): ✔️
Atrophy signs: ✔️ (via hypertransmission, thinning)
Important for: drusen interpretation and GA risk, though, not always a standalone numeric biomarker.
- Hyperreflective foci (HRF)
Detection: ✔️
Localization: ✔️
Counting / burden tracking: ✔️
These are high-value biomarkers for progression risk in PBM monitoring.
Assistance like this allows clinicians to track PBM response objectively, identify responders vs non-responders earlier, and generate consistent reports for clinical decision-making or research. In short, while OCT provides the necessary imaging depth, Altris AI unlocks its full value for PBM by making subtle retinal changes measurable, comparable, and clinically actionable.
Conclusion
PBM represents a novel and biologically plausible therapeutic modality that targets key pathological mechanisms in retinal disease. By enhancing mitochondrial function, reducing oxidative stress, and modulating inflammation, PBM holds significant potential to complement existing treatment strategies and improve outcomes in retinal disease management. However, further research is required to fully define its role in routine clinical practice.
Despite the promising findings, the clinical integration of PBM remains in an evolving stage. Variability in treatment parameters—including wavelength, dose, and treatment protocols—has limited standardization and comparability across studies. Moreover, much of the current evidence is derived from small-scale clinical trials and preclinical models, underscoring the need for large, randomized controlled trials to establish optimal treatment regimens for dry AMD progression and to assess long-term efficacy in other eye pathologies.
In this context, OCT—especially when enhanced with AI-driven analysis—plays a critical role in advancing PBM adoption. Quantitative OCT biomarkers such as drusen volume, outer retinal integrity, and subtle structural changes provide objective endpoints for assessing therapeutic response. AI-based platforms further enable precise, reproducible, and longitudinal analysis of these changes, helping to standardize evaluation, identify responders earlier, and strengthen the clinical evidence base for PBM.
FAQ Section
1. How do I objectively measure response to PBM therapy?
Clinicians look for quantifiable OCT biomarkers, not just visual acuity:
- Drusen volume (regression or stabilization)
- Outer retinal layer integrity (EZ, RPE)
- Hypertransmission / atrophy areas
The challenge: changes are subtle → require precise, longitudinal OCT comparison.
2. Which OCT biomarkers are most relevant for PBM monitoring?
The most discussed and clinically relevant:
- Drusen volume/area
- RPE atrophy
- Hypertransmission
- Ellipsoid Zone (EZ) disruption/loss
- Hyperreflective foci (secondary)
For GA specifically:
Overlap of RPE atrophy + hypertransmission + EZ loss = key composite metric.
3. How often should I monitor patients on PBM?
Typical real-world patterns:
- Baseline OCT before starting PBM
- Follow up every 3–6 months
- More frequent (monthly) in studies.
4. How do I distinguish PBM effect from natural AMD progression?
Distinguishing the effect of PBM from the natural progression of AMD remains one of the key clinical challenges. AMD typically progresses slowly and can show natural fluctuations, while PBM-related changes tend to be gradual and relatively modest. To differentiate between the two, clinicians rely on consistent OCT metrics tracked over time, comparing trends rather than single visits. Bilateral analysis—evaluating treated versus untreated eyes—can provide additional context, while assessing the rate of change, such as slowing of drusen growth or stabilization of atrophic areas, helps determine whether observed changes are likely treatment-related rather than part of the disease’s natural course.
5.Do I need AI/software for PBM monitoring, or is manual OCT review enough?
Whether AI/software is needed for PBM monitoring versus manual OCT review is an increasingly important question in clinical practice. While manual assessment can provide a general, qualitative understanding, it is often variable, time-consuming, and limited in its ability to detect subtle changes. PBM, however, requires identification of micron-level structural differences and high reproducibility across visits to accurately assess treatment response. AI-based OCT analysis addresses these challenges by enabling automated segmentation of key biomarkers, delivering precise volumetric measurements, and supporting reliable longitudinal tracking in standardized units such as mm², mm³, and percentage change. This level of consistency also helps clinicians more confidently distinguish responders from non-responders, making monitoring more objective and clinically actionable.
References:
https://pmc.ncbi.nlm.nih.gov/articles/PMC11488463/
https://link.springer.com/article/10.1007/s40135-025-00340-x
https://d-nb.info/136218389X/34
https://www.frontiersin.org/journals/ophthalmology/articles/10.3389/fopht.2024.1388602/full
https://retinatoday.com/articles/2020-may-june/photobiomodulation-as-a-treatment-in-dry-amd
https://espansionegroup.it/it/
-

Geographic Atrophy Retina OCT Biomarkers: Detection, Quantification, and Monitoring
 Maria Znamenska
5 min.
Maria Znamenska
5 min.Introduction. Overview of Geographic Atrophy (GA) as a Late Stage of Dry AMD
Geographic atrophy (GA) is a chronic progressive retinal degeneration that represents part of the late stage of age-related macular degeneration (AMD). It is characterized by gradual and irreversible atrophy of photoreceptors, the retinal pigment epithelium (RPE), and the choriocapillaris. As a result, a persistent defect of neurosensory tissue develops, which clinically manifests as central vision loss, the appearance of central scotomas, and reduced contrast sensitivity.
Atrophic lesions typically originate in the outer retinal layers and gradually expand, involving the macula and fovea. Over time, this leads to irreversible visual impairment and a significant decline in quality of life. In the early stages, patients may not experience noticeable changes in visual acuity. However, involvement of the central foveal region may lead to a sudden functional deterioration, with patients reporting difficulties in reading, recognizing faces, and working with fine details.
GA is considered one of the leading causes of clinically significant central blindness among people over the age of 60 in developed countries. With the aging population, the prevalence of this condition continues to increase, creating a substantial social and economic burden. In addition to reduced visual acuity, GA significantly affects patients’ quality of life.
Geographic atrophy retina OCT, together with modern digital image analysis algorithms, has become a key tool in the diagnosis, monitoring, and evaluation of OCT biomarkers predicting GA progression. OCT provides cross-sectional imaging of the retina with microscopic resolution, enabling detailed assessment of individual retinal structures—from the inner retinal layers to the RPE–Bruch’s membrane–choriocapillaris complex. This technology has enabled the transition from subjective ophthalmoscopic assessment to objective structural analysis.
The advantages of OCT in the diagnosis and monitoring of GA include its non-invasive nature, high reproducibility, ability to detect early structural changes, and accurate quantitative measurements. Structural alterations at the level of photoreceptors and the RPE often occur long before they become visible on ophthalmoscopy or fundus photography. Proper recognition of OCT biomarkers of GA is essential not only for disease diagnosis but also for personalizing treatment strategies, predicting the risk of progression, and evaluating therapeutic outcomes.
The purpose of this article is to summarize current scientific evidence on OCT biomarkers of geographic atrophy, including their morphological definition, quantitative parameters, prognostic significance, and role in monitoring disease progression. Particular attention will be given to the practical aspects of OCT in clinical practice, interpretation of longitudinal changes, and effective communication with patients regarding the expected course of the disease.
2. Main OCT Biomarkers of Geographic Atrophy
Modern understanding of GA morphology has been largely shaped by the work of international expert groups, particularly the Classification of Atrophy Meetings (CAM) Group. The CAM group proposed standardized terminology and clear OCT-based criteria for retinal atrophy, enabling harmonization of diagnostic approaches in both clinical practice and multicenter studies.
The CAM group recommends spectral-domain OCT (SD-OCT) as the preferred imaging modality for detecting GA-related changes, as it allows identification of the earliest signs of developing atrophy.
2.1 OCT Signs of Geographic Atrophy
The following three features form the basis for the standardized OCT definition of GA:
- Loss of the outer retina
- Loss of the retinal pigment epithelium (RPE) ≥250 µm in diameter
- Choroidal hypertransmission ≥250 µm in diameter
1. Loss of the Outer Retinal Layers
On OCT B-scans this manifests as:
- disruption or loss of the ellipsoid zone (EZ)
- absence of the interdigitation zone
- thinning or complete loss of the outer nuclear layer (ONL)
- thinning (atrophic changes) of the neuroepithelium above the lesion
This feature reflects the loss of photoreceptors, which are the primary functional elements responsible for central vision.
2. Loss of the Retinal Pigment Epithelium (RPE)
The CAM group established a threshold of 250 µm in the largest horizontal dimension to define clinically significant atrophy.
AI detection of RPE atrophy OCT appears as:
- absence or severe thinning of the hyperreflective RPE band
- a well-defined border between preserved and atrophic RPE
3. Choroidal Hypertransmission
Due to the loss of the RPE, light penetrates more deeply into the underlying layers, resulting in increased visualization of the choroid.
On OCT this appears as:
- Increased visibility of the choriocapillaris layer
- Clear correspondence with the area of RPE defect
Classification of Outer Retinal Atrophy Associated with AMD
- Complete RPE and outer retinal atrophy (cRORA)
- Incomplete RPE and outer retinal atrophy (iRORA)
- Complete outer retinal atrophy (cORA)
- Incomplete outer retinal atrophy (iORA)
2.2 OCT Parameters for Monitoring Geographic Atrophy
Once the diagnosis is established, OCT biomarkers predicting GA progression and quantitative monitoring of disease progression becomes critical.
1. Morphological Triad
RPE atrophy, choroidal hypertransmission, and neuroepithelial atrophy represent the hallmarks of complete retinal atrophy.
This triad defines retinal atrophy within the lesion area and allows differentiation between complete and incomplete atrophy using structural criteria.
2. Area of Geographic Atrophy (mm²)
Quantitative measurement of GA area is a key parameter in both clinical practice and research.
OCT segmentation enables highly reproducible calculation of the affected area. Modern OCT systems allow:
- automatic segmentation of atrophy boundaries
- calculation of the GA area in mm²
- comparison of measurements between visits
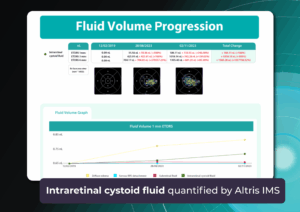
The annual enlargement rate of the GA area is an objective marker of disease progression and correlates with functional visual outcomes. Importantly, the GA area may increase even when visual acuity remains stable.
The area of GA served as the primary endpoint in clinical trials evaluating the intravitreal therapies Syfovre and Izervay, which were recently approved by the FDA as treatments to slow GA lesion growth.
AI-based algorithms further improve the precision and reproducibility of measurements, which is particularly important for long-term monitoring.
Modern OCT systems provide GA area measurements in mm², and comparisons between visits provide an objective measure of disease dynamics. Even when patients do not perceive changes, increasing lesion area confirms disease progression.
3. Distance Between GA Lesions and the Fovea
An important quantitative parameter is the distance between the foveal center and the nearest border of the atrophic lesion.
This parameter has direct functional significance. Decreasing distance over time correlates with declining visual function: the closer GA approaches the fovea, the higher the risk of sudden vision loss.
Patients with GA lesions approaching the fovea have a poorer prognosis and often require more intensive monitoring and therapeutic interventions.
This parameter also allows objective risk prediction and supports:
- early referral to specialized ophthalmology centers
- discussion of potential vision loss with patients
2.3 Predictors of GA Development and Progression
GA frequently develops as a consequence of drusen involution or structural alterations of the RPE.
GA lesions in AMD may arise in association with:
- certain drusen types (large or confluent drusen, reticular pseudodrusen)
- previous choroidal neovascularization
- RPE detachment or RPE tear
- refractile deposits
- vitelliform lesions
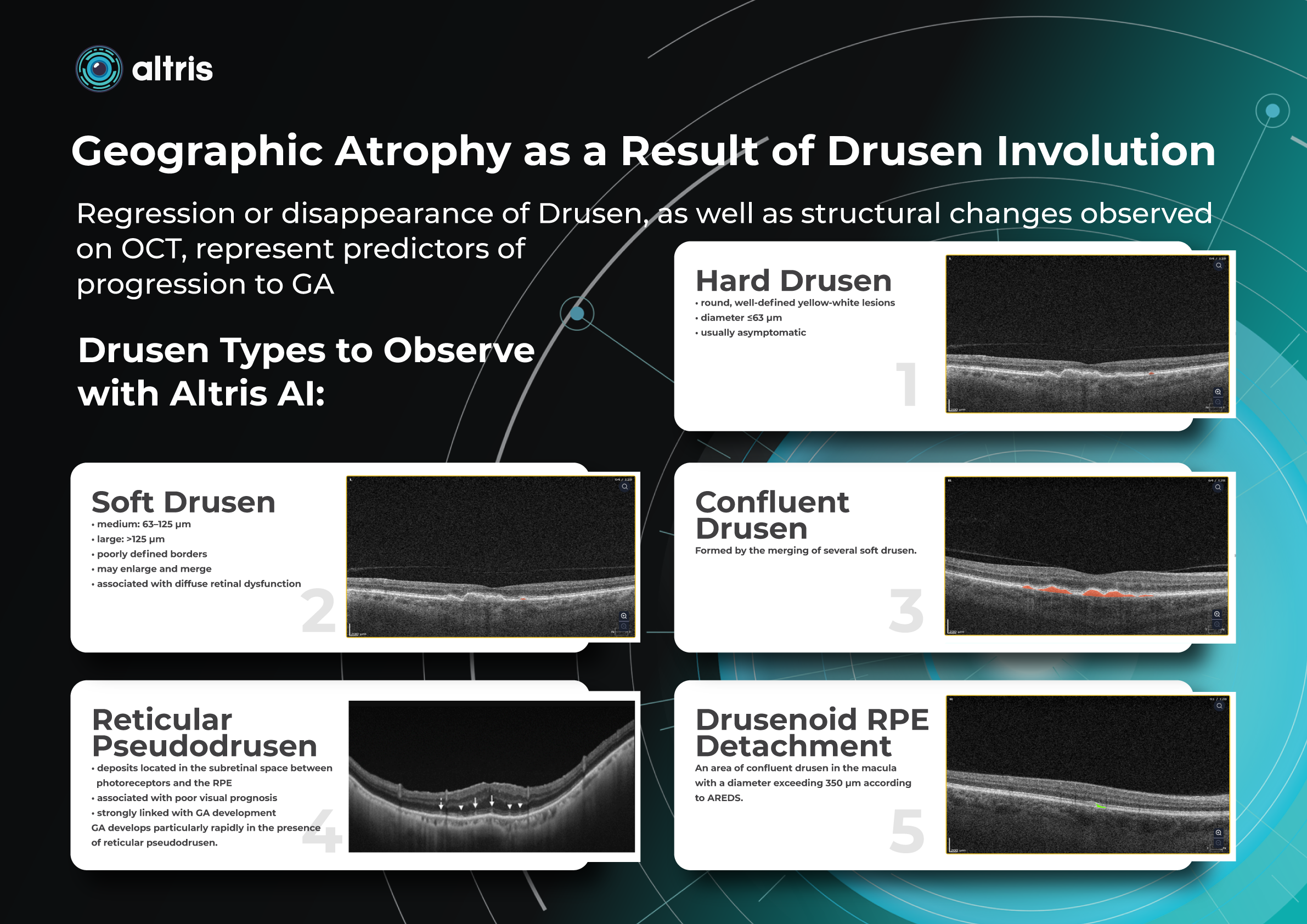
Geographic Atrophy as a Result of Drusen Involution
Drusen are localized accumulations of pathological material (photoreceptor metabolic by-products) between the RPE and Bruch’s membrane. They may change in number, size, and morphology.
Regression or disappearance of drusen, as well as structural changes observed on OCT, represent predictors of progression to GA. Regular monitoring allows early detection of potentially dangerous changes.
Types of Drusen

1. Hard Drusen
- round, well-defined yellow-white lesions
- diameter ≤63 µm
- usually asymptomatic
2. Soft Drusen
- medium: 63–125 µm
- large: >125 µm
- poorly defined borders
- may enlarge and merge
- associated with diffuse retinal dysfunction
3. Confluent Drusen
Formed by the merging of several soft drusen.
4. Drusenoid RPE Detachment
An area of confluent drusen in the macula with a diameter exceeding 350 µm according to AREDS.
5. Cuticular Drusen
- located between the RPE and Bruch’s membrane
- small in diameter but numerous
- often confluent
- steep, sloping sides (“saw-tooth” appearance)
- may disrupt RPE structure
- represent a risk factor for progression to GA
6. Reticular Pseudodrusen
- deposits located in the subretinal space between photoreceptors and the RPE
- associated with poor visual prognosis
- strongly linked with GA development
GA develops particularly rapidly in the presence of reticular pseudodrusen.
Predictors of GA Development in Eyes with Drusen
- large numbers of drusen, particularly in the central macula
- regression of drusen
- structural changes such as heterogeneous internal reflectivity
These predictors help identify patients at high risk for GA development and are valuable for optimizing monitoring intervals and potential preventive strategies.
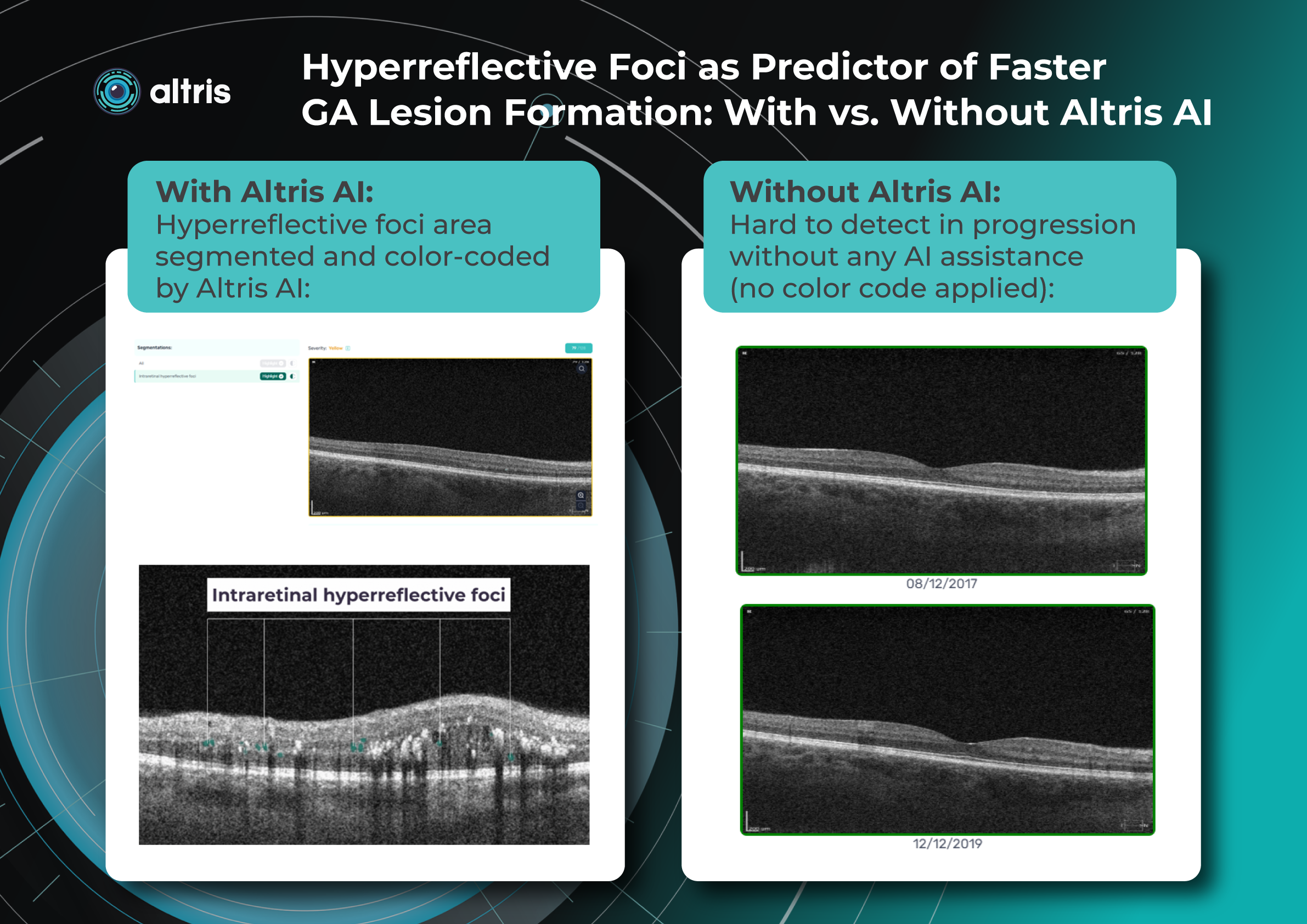
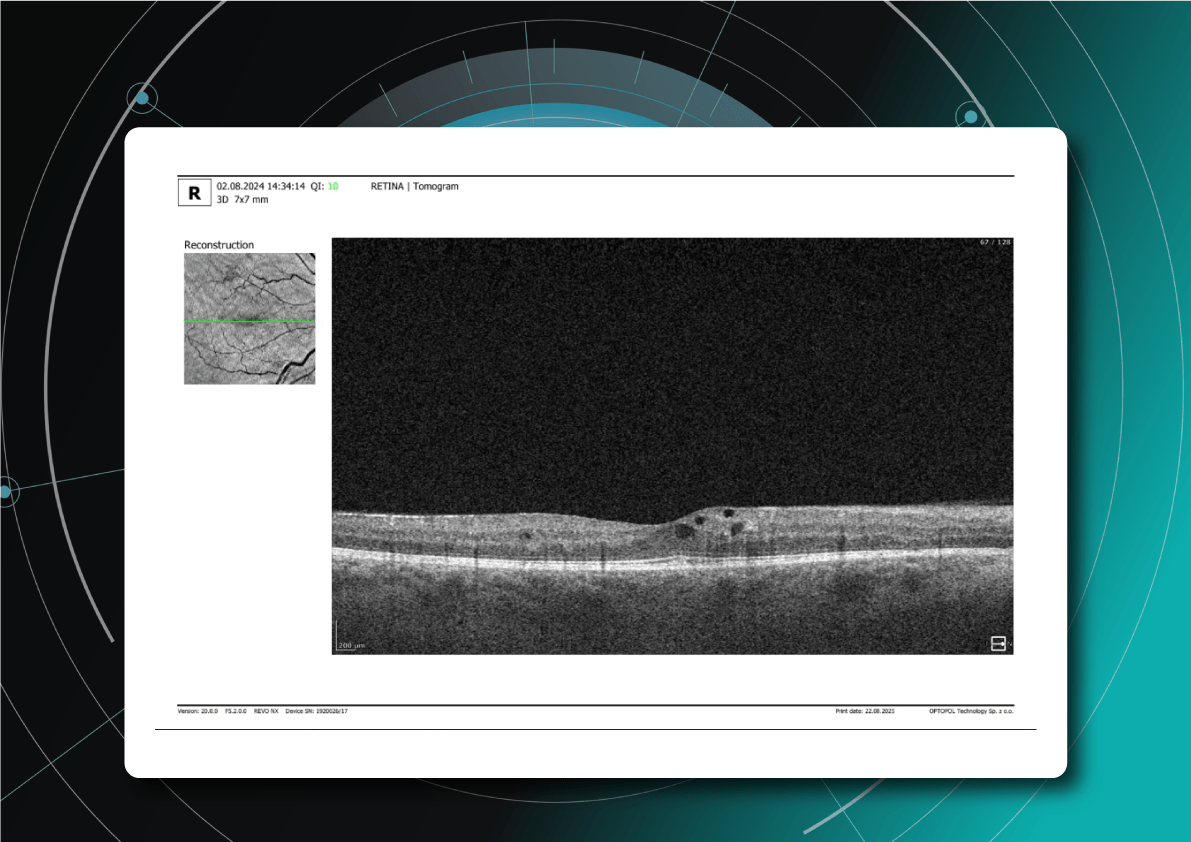
Another predictor of faster GA lesion formation is hyperreflective foci. These are small intraretinal hyperreflective dots, often located above drusen and typically associated with local disruption of the RPE structure. They likely represent migrating RPE cells and activated microglia. A tiny blue spot is a hyperreflective foci area detected by Altris automated GA segmentation OCT:

Their presence significantly increases the risk of GA development within the next few years (in some studies up to five-fold within two years).
Clinical Importance of Predictors
Identifying high-risk patients allows clinicians to:
- individualize OCT monitoring frequency
- initiate treatment earlier
- predict functional vision loss
- discuss expected disease progression with patients in a timely manner.
Management of Geographic Atrophy and Patient Education
Management of patients with GA today extends far beyond simple observation. It involves an active, structured strategy that combines regular OCT monitoring, timely initiation of therapy, risk-factor modification, and comprehensive patient education.
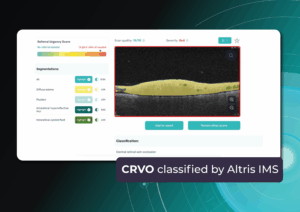
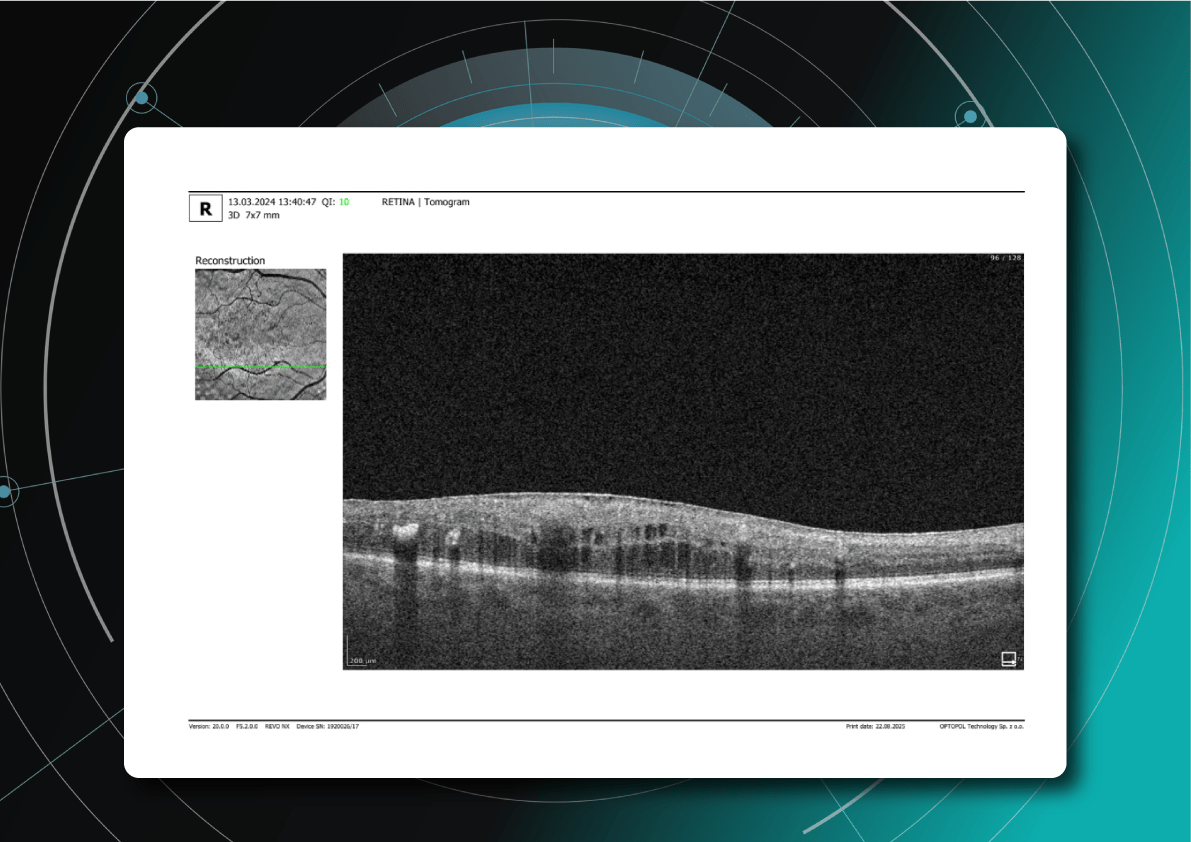
The main goal is to slow disease progression and reduce the rate of atrophy expansion while preserving the central fovea for as long as possible. GA Progression quantified via Altris:

The Role of OCT
Effective GA management is impossible without high-quality OCT monitoring.
OCT enables clinicians to:
- quantify the area of atrophy
- determine the rate of lesion expansion
- measure the distance to the fovea
- analyze outer retinal layer integrity
- identify predictors of rapid progression
Monitoring is recommended every 3–6 months, and when intravitreal therapy is used, OCT should be performed before each injection to assess disease activity and lesion growth rate.
OCT also serves as a powerful motivational tool: showing patients the dynamics of structural changes helps them better understand the need for treatment and regular follow-up visits.
Patients should be informed that GA may progress without sudden visual deterioration. Structural OCT changes often precede functional vision loss, making regular examinations essential even when visual acuity appears stable.
Current Treatment Options

Intravitreal Therapy
- Izervay (avacincaptad pegol)
- Syfovre (pegcetacoplan)
For the first time in decades, FDA-approved treatments are available that slow the expansion rate of GA lesions. Although they do not restore lost vision, slowing visual decline is an important clinical goal.
Patients should clearly understand that treatment slows progression but does not restore vision. Proper expectation management improves treatment adherence and reduces disappointment.
Nutritional Supplements
Formulations based on AREDS / AREDS2 have been shown to reduce the risk of progression from intermediate AMD to advanced stages.
Patients should be informed that these supplements do not treat GA, but may have preventive value at earlier stages.
What Patients Must Understand
1. Progressive Nature of GA
GA is a chronic progressive disease. The area of atrophy almost always increases over time. The rate of progression varies depending on morphological characteristics.
Patients should understand that treatment aims to slow, not completely stop, disease progression.
2. Importance of Lifestyle
Although lifestyle modification has limited influence once GA is established, recommendations remain relevant:
- smoking cessation
- blood pressure and lipid control
- antioxidant-rich diet and omega-3 fatty acids
- regular physical activity
These factors improve overall vascular health and may reduce systemic inflammation.
3. Psychological Adaptation
Progressive central vision loss often leads to anxiety, fear of blindness, and reduced social activity.
It is important to discuss:
- low-vision aids (magnifiers, telescopic glasses, electronic magnifiers)
- support resources for people with low vision
Psychological support significantly improves adaptation and quality of life.
Patient Partnership: The Foundation of Success
Modern management of dry AMD is no longer hopeless. With approved therapies and evidence-based preventive strategies, clinicians can meaningfully influence the rate of disease progression.
However, the effectiveness of any strategy depends on collaboration between the physician and the patient.
Patient education regarding:
- the nature of the disease
- the role of regular OCT monitoring
- treatment possibilities and limitations
- the importance of lifestyle modification
is an essential component of modern GA management.
FAQs
Which OCT biomarkers are predictive of GA progression to look for?
Key biomarkers include hypertransmission defects, RPE atrophy, photoreceptor loss, ellipsoid zone disruption, hyperreflective foci, and reticular pseudodrusen. These structural changes are strongly associated with GA development and progression in AMD.
How does AI OCT help prioritize patients at risk of GA progression?
AI for GA systems identifies high-risk biomarkers and calculates progression rates, enabling clinicians to triage patients for closer monitoring or treatment.
Can AI detect multiple retinal pathologies in addition to GA?
Many platforms detect 70+ retinal pathologies and biomarkers simultaneously on OCT scans. Altris detects and quantifies 40+ retina biomarkers and 40+ pathologies.
How can AI quantify geographic atrophy on OCT scans?
AI algorithms automatically segment GA lesions and calculate lesion area, retinal layer loss, and biomarker overlap, providing objective measurements in millimeters or percentages.
Can AI OCT support treatment decisions for GA therapies?
AI can measure structural parameters such as EZ loss or RPE integrity, which may help evaluate treatment response or disease activity. Altris applies Flags to filter out the eligible patients then.
Can AI detect early GA before it becomes clinically visible?
Yes. AI models can identify subtle structural abnormalities on OCT, such as EZ disruption or early hypertransmission, enabling earlier detection of atrophy.
Which OCT metrics should be monitored to track GA progression?
Clinically relevant metrics include: GA lesion area (mm²), rate of lesion growth, distance from lesion margin to the fovea, percentage of macular involvement. AI can automatically calculate and track these parameters over time.
How to efficiently measure geographic atrophy on OCT?
To efficiently measure Geographic Atrophy on Optical Coherence Tomography (OCT), clinicians should identify key biomarkers such as RPE loss, outer retinal thinning, and choroidal hypertransmission, then quantify the atrophy area (mm²) using en-face OCT or automated segmentation tools. Tracking lesion size and its distance to the fovea over time allows accurate monitoring of disease progression. AI-assisted OCT platforms can automate detection and measurements, making longitudinal assessment faster and more consistent.
References
- Guymer RH, Rosenfeld PJ, Curcio CA, et al.
Incomplete retinal pigment epithelium and outer retinal atrophy in age-related macular degeneration: Classification of Atrophy Meeting report.
Ophthalmology.
Available at: https://pubmed.ncbi.nlm.nih.gov/38387826/ - Natural history and progression of geographic atrophy in AMD.
ScienceDirect.
Available at: https://www.sciencedirect.com/science/article/pii/S2468653023006681 - OCT Spotlight: Characterizing Geographic Atrophy Development and Progression.
Retina Today.
Available at: https://retinatoday.com/articles/2025-apr/oct-spotlight-characterizing-ga-development-and-progression - Automated monitoring of geographic atrophy using OCT imaging.
Scientific Reports.
Available at: https://www.nature.com/articles/s41598-023-34139-2 - Classification of Atrophy Meeting (CAM) consensus for OCT-based atrophy classification in AMD.
American Academy of Ophthalmology Journal.
Available at: https://www.aaojournal.org/article/S0161-6420(17)31703-7/abstract - Identifying Geographic Atrophy Biomarkers.
Optometric Management.
Available at: https://www.optometricmanagement.com/issues/2025/october/identifying-geographic-atrophy-biomarkers/ - FDA Approval Announcement for Izervay (avacincaptad pegol).
Astellas Pharma Newsroom.
Available at:
https://newsroom.astellas.com/2023-08-05-Iveric-Bio-Receives-U-S-FDA-Approval-for-IZERVAY-TM-avacincaptad-pegol-intravitreal-solution-,-a-New-Treatment-for-Geographic-Atrophy
-

Altris AI Receives Health Canada Approval
 Altris Inc.
Altris Inc.
Altris AI Receives Health Canada Approval, Reinforcing Its Position as a Globally Trusted AI Decision Support Platform for OCT Analysis
Regulatory clearance marks a pivotal milestone in Altris AI’s international expansion and its mission to bring clinical-grade AI to eye care worldwide.
10 March 2026 — Altris AI, a leading provider of AI-powered decision support for optical coherence tomography (OCT) analysis, today announced it has received approval from Health Canada | Santé Canada, Canada’s federal health regulatory authority. This clearance represents a significant step forward in the company’s global growth strategy and its commitment to meeting the highest standards of medical device safety and clinical reliability.
For a company scaling across international markets, regulatory approvals are far more than administrative milestones — they are foundational growth enablers. Health Canada approval strengthens Altris AI’s international positioning, opens new pathways for future regulatory submissions across key markets, and delivers a clear signal to the global healthcare community: Altris AI is built for real-world clinical practice.
The approval confirms that Altris AI’s clinical and technical validation withstands the rigorous scrutiny of Health Canada’s regulatory review process, which evaluated the platform across five critical dimensions: clinical evidence supporting efficacy and safety claims; risk management protocols; cybersecurity safeguards; quality management systems; and intended use claims. Each of these pillars reflects the standard that modern AI-driven medical devices must meet before being trusted in clinical settings.
Altris AI’s platform serves optometrists, ophthalmologists, and pharmaceutical organizations by providing intelligent, reliable support for interpreting OCT scans — one of the most widely used diagnostic tools in eye care. By surfacing clinically relevant findings with speed and precision, Altris AI empowers clinicians to make more informed decisions, improve patient outcomes, and increase the efficiency of ophthalmic workflows.

“This approval is external confirmation that our platform meets the standards required for medical-grade AI,” said Maria Znamenska, Chief Medical Officer at Altris AI. “Health Canada’s review is thorough, evidence-based, and internationally respected. Receiving this clearance validates not just our technology, but the entire approach we’ve taken — building AI that clinicians can trust, in environments where accuracy is not optional.”
The Health Canada clearance follows a broader regulatory strategy that positions Altris AI as a compliant, audit-ready platform for healthcare systems worldwide. As global regulators increasingly scrutinize AI in medical devices, early and consistent compliance across multiple jurisdictions will become a decisive competitive differentiator.
Altris AI’s mission is to accelerate the transition of AI in eye care from innovation to infrastructure — not as a replacement for clinical expertise, but as a trusted decision-support layer that elevates the standard of care across every point of practice.
About Altris AI Altris AI is an AI Decision Support platform for OCT analysis, designed to support optometry, ophthalmology, and pharma with clinically validated, regulatory-compliant technology. The company is committed to expanding access to intelligent eye care diagnostics globally.
-

AI in Optometry: 5 Real Applications
 Maria Znamenska
9 min.
Maria Znamenska
9 min.Highlights: AI in optometry is revolutionizing clinical decision-making by allowing eye care professionals to analyse B-scans with greater precision, consistency, and confidence right at the point of care.
- AI for fundus analysis is another way to automatically evaluate fundus scans in an optimized way throughout all devices.
- AI-driven deep learning algorithms can detect and quantify retinal and optic nerve pathologies in OCT images, enabling earlier identification of diseases such as glaucoma, age-related macular degeneration (AMD), and other retinal conditions.
- AI-assisted analysis enhances clinical efficiency by helping clinicians triage scans, monitor disease progression over time, and focus on clinically significant findings, rather than relying solely on manual scan reviews.
- Other AI-powered tools provide objective visual insights for clinicians and patients, improving the accuracy of triage and treatment monitoring and enhancing patients’ understanding of retinal health.
- AI enables optometrists to manage more complex ocular cases in primary care, facilitating earlier detection, risk stratification, and informed referral decisions based on objective insights.
- AI chatbots in optometry inform about eye symptoms, guide whether to seek care (urgent vs. routine), suggest possible conditions, support patients and help decide next steps, etc. Or manage eye care specialists’ daily routine.
Introduction
Artificial intelligence is increasingly shaping healthcare by enhancing clinicians’ ability to interpret complex medical data and make earlier, more informed decisions. AI in optometry is especially important in OCT imaging, where it is essential to correctly interpret subtle structural changes to identify eye disorders early.
AI for optometrists can therefore more reliably and consistently detect and track retinal and optic nerve disorders by integrating deep learning into OCT data. In routine optometric practice, risk management and referral decision-making are enhanced by converting OCT images into understandable, actionable findings.
Discover how optometry AI tools redefine optometry by improving diagnostic accuracy, clinical efficiency, and the quality of patient care in 5 real cases.
1. AI Decision Support for OCT Analysis
One of the most effective AI applications in optometry is AI decision support for OCT analysis.
AI decision-support systems are increasingly applied to OCT imaging to assist optometrists in interpreting complex retinal and optic nerve data. Here’s one of the real cases when AI brings ultimate use to practitioners:
Thus, AI-powered platforms like Altris use deep learning algorithms to automatically detect and quantify structural changes, highlight areas of concern, and track progression over time via an AI OCT pathology detection tool.
By analysing patterns across large datasets of retinal scans, the system can flag subtle abnormalities that may be difficult to identify manually, segment them automatically, and provide structured, visual insights that help clinicians make more informed, consistent decisions while monitoring patient eye health. It does everything eye care specialists do, but faster, error-free, and unbiased.
In particular, Altris AI also applies deep learning to OCT scans to automatically detect and highlight complex retinal and optic nerve changes.
The system quantifies abnormalities, tracks progression over time, and provides visual insights that help optometrists interpret scans more accurately and consistently, again supporting far more informed clinical decisions.
2. AI for Fundus Analysis
AI for Fundus Analysis is another way to automatically evaluate fundus scans in an optimized way throughout all OCT devices. Among the top 3 AI software solutions for fundus imaging, there are:
Auroraa AI (Optomed) is an advanced artificial intelligence platform integrated with Optomed’s handheld and tabletop fundus cameras, designed to detect multiple retinal abnormalities including diabetic retinopathy, glaucoma, and age‑related macular degeneration; it provides immediate, automated screening results to support clinicians and improve early disease detection.
Beammed’s AI‑powered fundus cameras pair intelligent image analysis with portable retinal imaging to enable early detection of diabetic retinopathy and other retinal conditions, leveraging deep learning algorithms to highlight pathology and help streamline screening programs.
Cybersight AI (Orbis) offers an AI‑driven diagnostic support tool focused on interpreting fundus images to assist eye care providers in low‑resource settings and telemedicine programs, combining machine learning with expert clinical guidance to improve access to retinal disease screening globally. Here’s how fundus tool may look like:
Such systems provide severity scores, highlight areas of concern, and track changes over time, giving clinicians objective, reproducible insights to support their decisions. Since AI can detect early signs of conditions like glaucoma, diabetic retinopathy, and age-related macular degeneration, it often spots subtle changes that are hard to see with the naked eye.
3. AI for Automated Visit Scheduling
AI‑powered appointment scheduling systems are digital tools that, alongside any modern AI OCT pathology detection tool or similar, use artificial intelligence — including natural language processing (NLP), predictive analytics, machine learning, and automated communication — to handle clinical scheduling tasks usually done manually by staff. These tools can:
- let patients self‑schedule online, by chat, voice, or text at any time,
- automatically confirm, remind, reschedule, or cancel appointments,
- optimize schedules based on provider availability and patient needs, and
- predict and prevent clinic inefficiencies such as no‑shows.
In essence, the system acts as a digital receptionist and smart scheduler, integrating with clinic practice management software, EHRs, and CRM systems to manage workflows seamlessly. Best EHR systems include:

Elation EHR is a cloud-based electronic health record designed primarily for independent primary care practices. It focuses on simplifying clinical workflows, documentation, and patient engagement, with strong charting tools and longitudinal patient records. It’s used to help physicians deliver personalized care while reducing administrative burden.
Epic is one of the most widely used enterprise EHR systems globally, typically implemented by large hospitals and health systems. It integrates clinical, administrative, and billing functions into a single platform, supporting everything from patient records to population health management. It’s used to coordinate care at scale and improve interoperability across departments and organizations.
Tebra combines electronic health records with practice management, billing, and patient communication tools, targeting small to mid-sized medical practices. It streamlines front-office and clinical operations in one system, helping practices manage scheduling, documentation, and revenue cycle efficiently.
Nextech EHR is a specialty-focused EHR designed for fields like ophthalmology, dermatology, and plastic surgery. It includes tailored templates, imaging integration, and workflow tools specific to these specialties. It’s used to enhance clinical efficiency and documentation accuracy in specialized practices.
InSync EHR is a cloud-based EHR built for behavioral health and therapy practices. It supports telehealth, scheduling, documentation, and billing, with features tailored to mental health workflows. It’s used to improve care coordination and streamline operations for therapists and behavioral health providers.
These tools are designed to improve operational efficiency, reduce administrative burden, and enhance the patient journey in healthcare settings. They offer 24/7 availability, personalized booking flows, and real-time updates, making them a powerful part of your workflow.
ModMed EMA (Electronic Medical Assistant) is an AI-driven, specialty-specific EHR developed by Modernizing Medicine. It uses structured data and adaptive templates to support clinical decision-making and documentation. It’s used by specialists to increase efficiency, improve outcomes, and reduce time spent on manual data entry.
“Up to 71% of U.S. hospitals now use predictive AI technologies for scheduling and related automation.”

Indeed, AI in optometry for appointments, self-scheduling, and other administrative tasks can optimize routine workflows and offer a range of benefits for both opticians and their visitors. They:
- Remove Administrative Burden
AI takes over repetitive scheduling tasks — booking, confirmations, cancellations, preregistration, and follow‑ups — freeing front‑desk staff and nurses to focus on patient care rather than paperwork.
Historically, nurse managers and front‑desk staff can spend up to 40% of their day on scheduling tasks, and automating this saves hours of labour.
- Provide 24/7 Booking & Patient Flexibility
Patients no longer have to call during office hours. AI scheduling tools enable self‑service booking, changes, and cancellations at any time via chatbots, voice interfaces, or online portals.
This has become especially crucial, as 40% of healthcare appointments are requested outside normal business hours — times when traditional phone booking is impossible.
- Reduce No‑Shows & Better Attendance
Automated reminders — via SMS, email, or voice — consistently reduce no‑show rates, which can otherwise waste clinic time and revenue. Clinics using these tools have reported:
-
No‑shows dropped from 20% to as low as 7% with automated reminders.
-
In some settings, AI virtual assistants reduced missed visits by up to ~73%.
AI can also predict patients likely to skip visits and prompt engagement before issues arise.
- Enhance Resource Use
Instead of manually guessing who should be booked and where, such AI in optometry:
✔ matches patients with the right clinician,
✔ avoids double bookings and schedule conflicts,
✔ spreads appointments evenly to reduce bottlenecks, and
✔ improves utilization of staff time and rooms.
AI scheduling can increase provider utilization by 15–25% and reduce wait times by 15–30%.
- Improve Patient Experience
Patients appreciate convenience. Access to online or chatbot booking correlates with improved satisfaction — one study showed satisfaction scores can rise by over 20% when patients can self‑manage appointments.
AI also reduces inbound phone volume by 25–40%, allowing clinics to serve patients more efficiently.
For instance, Tele-optometry decision support that offers
- AI pre-analyses remote exams
- Flags cases requiring in-person referral
- Supports non-specialist reviewers
has the following workflow impact:
- Scales remote care
- Consistent quality
- Faster review cycles
Used in:
- Large optometry chains
- Retail vision centres
- Franchise-based practices
- Rural clinics
- Community health centres
- Mobile eye clinics, etc.
4. AI Workflow & Practice Optimization
So, AI-assisted OCT analysis has become helpful in retina & glaucoma screening, in follow-ups, progression tracking, and other workflows. Meaning, what happens with OCT with the help of an AI OCT pathology detection tool is helpful in many ways:
- OCT scans are automatically analysed
- Pathologies are flagged (fluid, thinning, progression risk, etc.)
- Clinicians review AI output before raw scans.
So, they get
- Faster exam review
- Fewer missed subtle findings
- Consistent interpretation across doctors, etc.
But real-world AI applications in optometry for clinic management do not stop there: scheduling, reminders, patient triage, administrative automation, analytics, and beyond may also be supported by specific optometry AI tools. Here are a few examples.
AI triage tools for urgent eye issues
- AI pre-screens OCT/fundus images
- Exams are auto-prioritized by severity
- High-risk patients are flagged before the visit
Workflow impact:
- Smarter scheduling
- Faster routing to specialists
- Less cognitive load on staff
Used in:
- High-volume optometry chains
- Tele-optometry services
An example of AI diagnostic software for optometry and ophthalmology can be IDx-DR AI Diagnostic System for Detecting Diabetic Retinopathy.
Automated appointment reminders
Automated appointment reminders are AI- or rules-based systems, not yet independent chatbots or AI assistants, that automatically notify patients about upcoming eye exams via SMS, email, WhatsApp, or voice calls, without staff involvement.
They usually trigger:
- 7 days before (prep + reschedule window)
- 48–72 hours before (confirmation)
- 24 hours or same day (final reminder)
which makes them still a new generation of automation tools for eye care. Well-designed systems, like the majority of AI in optometry tools, support:
- HIPAA / GDPR-compliant messaging
- No clinical advice in reminders
- Audit logs of sent communications
- Opt-out controls
This makes them safe for both routine and medical eye care appointments.
For example: EyeCloudPro

But why do no-shows happen in optometry? Like in any other service sphere, there are real reasons why:
- Routine exams feel “non-urgent.”
- Long booking lead times (2–6 weeks)
- Patients forget dilation/prep requirements
- Elderly patients miss calls or misremember dates
- Parents forget pediatric appointments
- No easy way to confirm or reschedule
Across outpatient care (including optometry), automated reminders typically achieve:
- 20–40% reduction in no-shows
- 5–10% increase in appointment confirmations
- Up to 30% fewer last-minute cancellations
- 1–2 hours/day staff time saved (no manual calls)
In optometry specifically, clinics with long routine exam cycles often see results closer to the upper end of those ranges. What automated reminders do here is directly target forgetfulness + reduce friction. No ordinary staff can do that to such an extent. But AI can.
Therefore, by keeping patients aware, ready, and involved, automated appointment reminders help optometry clinics reduce no-show rates. Practices may increase patient flow, maximize chair time, and enhance attendance by providing timely, customized notifications through preferred channels—all without adding to the administrative burden.
Patient communication and optometrists’ education appsPatient communication, as well as optometrist education systems and applications, for mobile and desktop usage, support clearer understanding, better engagement, and more consistent care delivery.
For example: Chatbots in Healthcare from Capacity

These tools help patients understand their eye health and treatment plans, while enabling optometrists to stay informed through structured education, clinical updates, and decision-support resources—improving outcomes without increasing chair time:
- AI translates OCT findings into plain language
- Visual overlays show “what changed” and “why it matters.”
- Used chairside or via patient portal
Workflow impact:
- Better patient understanding
- Higher treatment acceptance
- Shorter explanation time per visit
Used in
- Routine eye exams
- OCT review appointments
- Retina & glaucoma visits
- Anti-VEGF treatment discussions
- Glaucoma therapy initiation
- Long-term monitoring plans, etc.
Altris Education application, as an example of a unique tool specifically designed for eye care specialists’ training:
5. Chatbots for Consultation
A separate category is AI chatbots and virtual assistants that help with patient follow-up, education, and communication, improving day-to-day patient communication and more. With the AI Help Assistant feature, you can create an AI chatbot trained on your specific content from any platform you like. Chatbots for consultation in optometry offer clear, practical benefits for both clinics and patients:
- 24/7 Q&A
- Absolutely personalized follow-up instructions
- Higher patients satisfaction rate
Furthermore, they provide instant, 24/7 responses to the most common eye-care questions, helping patients understand symptoms, prepare for visits, and follow post-exam instructions without even waiting for staff availability.
Chatbots can assist with pre-consultation triage by gathering symptoms, visual complaints, and medical history, allowing optometrists to focus on higher-value clinical tasks.
They also improve patient engagement and adherence by delivering personalized education, reminders, and care instructions in simple, easy-to-understand language.
Overall, optometry chatbots dramatically reduce administrative workload, shorten response times, and support more efficient, patient-centered care while maintaining consistent communication quality.
Types of Chatbots in Eye Care & Optometry

1. Symptom & Triage Chatbots
These ask users about eye symptoms, guide whether to seek care (urgent vs. routine), suggest possible conditions, and help decide next steps. Example: Ada Health
A study published in Eye showed that large‑language‑model‑based chatbots (e.g., ChatGPT) could answer common ophthalmology questions with high accuracy and clarity — scoring higher than alternative generative models for diagnostic and triage‑related queries (accuracy and comprehensiveness metrics showed ChatGPT did well on standard patient questions).
Another clinical evaluation found that AI (GPT‑4) correctly identified the appropriate diagnosis among the top three options in up to 93% of ophthalmology cases and correctly assessed urgency levels in most cases — performance comparable to that of trainees.
2. Real‑World Chatbots for Eye Care
The patient describes eye symptoms and uploads images (e.g., photos or OCT scans).
An AI assistant organizes symptoms and images for review, then a real ophthalmologist chats with the patient to guide care. Key inputs here:
- Collect symptoms in natural language
- Translate or clarify reports
- Help the clinician interpret visuals and decide next steps
It combines AI symptom intake with real human consultation, making remote triage efficient.
AI in optometry real chatbot examples:
- WebEyeClinic / Visio — symptom intake & remote clinician chat
- VisionCare Assistant — eye health guidance
- DocsBot AI — practice support
- Custom AI agents via Voiceflow — administrative automation, etc.
3. DocsBot AI for Optometry Services
Practice‑focused chatbot helping clinics answer FAQs, provide pre‑appointment instructions, and automate patient engagement. Benefits:
• Instant responses to common patient queries
• 24/7 availability for basic information
• Can free up staff time by handling routine communication, such as allowing self-booking, etc.“Patients express high satisfaction with AI self‑booking capabilities (up to 85% positive ratings).”

Here are some more real AI chatbot applications in eye care:
Tool / Platform Primary Focus DocsBot AI Patient FAQs & practice engagement ThriveDesk AI AI customer support for optometry Voiceflow AI Agent Custom appointment/scheduling chatbot MedReception AI Manage eye exams, contact lens orders, and optical retail coordination OptoAI AI Assistant Knowledge and clinical support agent Pod AI AI phone/communication agent As highlighted, there is a wide array of chatbot types in optometry, ranging from patient-facing virtual assistants to AI-powered communication platforms.
With features such as automated appointment scheduling, pre-visit coaching, FAQ handling, 24/7 patient engagement, and basic clinical decision support, these optometry AI systems offer substantial value.
By streamlining administrative tasks and improving patient education, these AI chatbots free up clinicians’ time to focus on direct care.
Overall, the integration of AI-driven chatbots is revolutionizing the delivery of eye care. They enhance operational efficiency, reduce missed appointments, support timely patient triage, and improve adherence to care plans. By combining automation with intelligent decision support, AI not only optimizes clinic workflows but also elevates patient outcomes and satisfaction, marking a transformative shift in modern optometry practice.
Conclusion
AI is rapidly becoming a practical and valuable tool in optometry, particularly for analysing OCT imaging. By enabling more consistent interpretation of complex retinal and optic nerve data, AI in optometry supports earlier identification of disease-related changes, more efficient triage, and improved longitudinal monitoring.
Beyond clinical efficiency, AI enhances patient communication by translating OCT findings into clear visual insights, supporting better understanding and engagement. As a result, optometrists are better equipped to manage more complex cases in primary care, make informed referral decisions, and deliver higher-quality, data-informed eye care—positioning AI as a meaningful complement to clinical expertise rather than a replacement.
FAQs
Is AI for optometry safe?
AI in optometry is generally safe when it is properly validated, regulated, and used as a clinical support tool rather than a replacement for professional judgment. It can improve screening accuracy, enable earlier detection of eye diseases, and streamline workflows, but it also carries risks such as diagnostic errors, data privacy concerns, and bias if systems are poorly trained or over-relied upon. For safe use, optometrists must remain responsible for final decisions; patients should be informed when AI is involved; and tools should comply with medical regulations (such as FDA or CE approval) and data protection standards, such as GDPR or similar, depending on the region.
What’s an AI OCT pathology detection tool?
An AI OCT pathology detection tool is a software system that uses artificial intelligence to analyse optical coherence tomography (OCT) images of the eye and automatically identify signs of disease, such as macular degeneration, glaucoma, or diabetic retinopathy; it assists clinicians by highlighting abnormalities and suggesting potential diagnoses, but it is designed to support—rather than replace—professional interpretation, and its safety and effectiveness depend on proper validation, regulatory approval, and clinician oversight.
Can AI help reduce no-shows and long wait times in an optometry practice?
Yes. AI applications in optometry are limitless. AI can help reduce no-shows and long wait times in optometry practices by supporting appointment scheduling, reminders, and workflow optimization—such as identifying scheduling patterns, highlighting bottlenecks, and enabling more efficient patient flow—while assisting staff with better resource planning and communication.
What can chatbots and AI assistants do for an optometry practice?
Chatbots and AI assistants can help an optometry practice automate patient communication, streamline scheduling, and improve clinical efficiency by answering FAQs 24/7, booking and confirming appointments, sending reminders, pre-screening patients with symptom checkers, collecting medical history before visits, and triaging urgent cases. They can also support front-desk staff by handling insurance questions, guiding patients to the right services (e.g., OCT, glaucoma screening, contact lens exams), following up after visits, and reactivating inactive patients through personalized messaging. Internally, AI in optometry assistants can summarize patient data, flag high-risk cases, analyse trends in no-shows or referrals, and help with marketing automation — ultimately reducing administrative workload, improving patient satisfaction, and increasing practice revenue.
What kinds of clinical or diagnostic support can AI provide in eye exams?
AI can support eye exams by assisting with image review, pattern recognition, and data organization—such as highlighting features in retinal images, supporting consistency in exam review, and providing quantitative reference information—while remaining a complement to clinician judgment rather than a replacement for clinical decision-making.
How can AI increase optical revenue and overall patient satisfaction?
AI in optometry can increase optical revenue and patient satisfaction by helping practices streamline workflows, reduce wait times, support personalized patient communication, and enhance the in-practice experience through clearer visualization and education tools—leading to more efficient operations, improved engagement, and higher-quality service delivery.
References:
Luhmann, U. F. O. (2015). Innate immunity in age-related retinal degeneration. Acta Ophthalmologica. https://doi.org/10.1111/j.1755-3768.2015.0144
https://remidio.us/solutions/teleophthalmology-telehealth/
https://medpick.in/product/idx-dr-ai-diagnostic-system-for-detecting-diabetic-retinopathy/
https://www.rcophth.ac.uk/academic-and-research/eye-journal/
https://webeyeclinic.com/how-it-works/
https://docsbot.ai/industry/optometry-services
https://www.voiceflow.com/ai/optometrists
https://healthus.ai/service/ai-chatbot-appointment-module/
https://arxiv.org/abs/2511.09394
https://ajbsr.net/data/uploads/6387.pdf
-

Altris becomes the winner of VSP Vision Challenge at Vision Expo
 Altris In
Altris In
ORLANDO, FL — Altris, an IMS for OCT analysis with ROU AI models, has been named the Judge’s Winner of the 2026 VSP Vision Innovation Challenge, one of the eyecare industry’s most prestigious startup competitions. The award was presented live on March 13, 2026, on the Innovation Stage at Vision Expo inside the Orange County Convention Center in Orlando, Florida.
Produced by RX and The Vision Council in collaboration with VSP Vision™, the 2026 VSP Vision Innovation Challenge drew applications from companies around the globe — more than half of which were venture-backed, collectively representing over $300 million in funding. After a rigorous selection process, Altris AI was chosen as one of four finalists and participated in an intensive four-week startup bootcamp before delivering a live pitch to a distinguished panel of industry judges.
Judges recognized Altris for its clinical impact, technological sophistication, and potential to fundamentally transform the eyecare experience. The judging panel represented a diverse cross-section of industry leaders, including executives, clinicians, and investors.
“Winning the VSP Vision Innovation Challenge is a powerful validation of what we’re building at Altris AI. Our mission is to put the most advanced retinal intelligence in the hands of every eye care professional — and this recognition from a world-class panel of judges confirms that the industry is ready for this transformation.”
Altris AI CEO, Andrey Kuropyatnyk
About Altris
Altris AI serves as a “second set of eyes” for eye care specialists, identifying more than 70 retinal biomarkers from optical coherence tomography (OCT) scans ( AI models are used for Research Use Only). The platform enables providers to match patients to the most appropriate treatments, devices, and clinical trials with objective, data-driven precision. Altris system is FDA-cleared and HIPAA-compliant, and integrates seamlessly with OCT scanners from nine major manufacturers.
By combining advanced deep learning algorithms with a clinically intuitive interface, Altris AI reduces diagnostic variability, supports earlier detection of sight-threatening conditions, and frees eye care professionals to focus on what matters most: patient outcomes.
About the VSP Vision Innovation Challenge
The VSP Vision Innovation Challenge is a global competition designed to source, support, and accelerate early-stage startups and technologies advancing the eyecare experience. This year’s finalists represented a broad spectrum of innovation — from AI-driven diagnostics and exam automation to digital education and accessibility-focused smart solutions.
In addition to pitching live, finalists exhibited at Vision Expo’s Innovation Center, a dedicated emerging-technology destination featuring more than 20 next-generation startups spanning AI, augmented and virtual reality, and advanced diagnostics.
“Innovation in eyecare is accelerating, which is why it’s crucial for the industry to stay actively engaged. We launched the VSP Vision Innovation Challenge to connect emerging technologies with the stakeholders they aim to serve, and this year is no exception. We’re proud to support solutions that advance care for both providers and patients.”
— Will Flanagan, Head of VSP Global Innovation Center Programs and Partnerships
Looking Ahead
With this recognition, Altris AI continues to accelerate its mission of making retinal AI accessible to every eye care professional. The company will leverage the visibility and industry connections gained from the challenge to deepen partnerships with providers, expand clinical integrations, and advance its platform’s capabilities.
The next VSP Vision Innovation Challenge will take place at Vision Expo 2027, scheduled for March 10–13 in Las Vegas. Applications are expected to open later this fall.
About Altris Inc.
Altris AI is a clinical-grade SaaS platform that empowers eye care specialists with AI-driven retinal analysis. By identifying 70+ retinal biomarkers from OCT scans, Altris AI helps providers deliver more precise, evidence-based care. The platform is FDA-approved, HIPAA-compliant, and compatible with OCT devices from nine leading manufacturers.
-

 Altris Inc.
Altris Inc.
Altris AI Receives Health Canada Approval, Reinforcing Its Position as a Globally Trusted AI Decision Support Platform for OCT Analysis
Regulatory clearance marks a pivotal milestone in Altris AI’s international expansion and its mission to bring clinical-grade AI to eye care worldwide.
10 March 2026 — Altris AI, a leading provider of AI-powered decision support for optical coherence tomography (OCT) analysis, today announced it has received approval from Health Canada | Santé Canada, Canada’s federal health regulatory authority. This clearance represents a significant step forward in the company’s global growth strategy and its commitment to meeting the highest standards of medical device safety and clinical reliability.
For a company scaling across international markets, regulatory approvals are far more than administrative milestones — they are foundational growth enablers. Health Canada approval strengthens Altris AI’s international positioning, opens new pathways for future regulatory submissions across key markets, and delivers a clear signal to the global healthcare community: Altris AI is built for real-world clinical practice.
The approval confirms that Altris AI’s clinical and technical validation withstands the rigorous scrutiny of Health Canada’s regulatory review process, which evaluated the platform across five critical dimensions: clinical evidence supporting efficacy and safety claims; risk management protocols; cybersecurity safeguards; quality management systems; and intended use claims. Each of these pillars reflects the standard that modern AI-driven medical devices must meet before being trusted in clinical settings.
Altris AI’s platform serves optometrists, ophthalmologists, and pharmaceutical organizations by providing intelligent, reliable support for interpreting OCT scans — one of the most widely used diagnostic tools in eye care. By surfacing clinically relevant findings with speed and precision, Altris AI empowers clinicians to make more informed decisions, improve patient outcomes, and increase the efficiency of ophthalmic workflows.
“This approval is external confirmation that our platform meets the standards required for medical-grade AI,” said Maria Znamenska, Chief Medical Officer at Altris AI. “Health Canada’s review is thorough, evidence-based, and internationally respected. Receiving this clearance validates not just our technology, but the entire approach we’ve taken — building AI that clinicians can trust, in environments where accuracy is not optional.”
The Health Canada clearance follows a broader regulatory strategy that positions Altris AI as a compliant, audit-ready platform for healthcare systems worldwide. As global regulators increasingly scrutinize AI in medical devices, early and consistent compliance across multiple jurisdictions will become a decisive competitive differentiator.
Altris AI’s mission is to accelerate the transition of AI in eye care from innovation to infrastructure — not as a replacement for clinical expertise, but as a trusted decision-support layer that elevates the standard of care across every point of practice.
About Altris AI Altris AI is an AI Decision Support platform for OCT analysis, designed to support optometry, ophthalmology, and pharma with clinically validated, regulatory-compliant technology. The company is committed to expanding access to intelligent eye care diagnostics globally.